Abstract
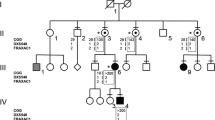
Five folate-sensitive fragile sites have been characterized at the molecular level (FRAXA, FRAXE, FRAXF, FRA16A and FRA11B)1–8. Three of them (FRAXA, FRAXE and FRA11B) are associated with clinical problems, and two of the genes (FMR1 in FRAXA2 and CBL2 in FRA11B8) have been identified. All of these fragile sites are associated with (CCG)n/(CGG)n triplet expansions which are hypermethylated beyond a critical size. FRAXE is a rare folate sensitive fragile site only recently recognized9. Its cytogenetic expression was found to involve the amplification of a (CCG)n repeat adjacent to a CpG island5. Normal alleles vary from 6 to 25 copies5. Expansions of greater than 200 copies were found in FRAXE expressing males and their FRAXE associated CpG island was fully methylated5. An association of FRAXE expression with concurrent methylation of the CpG island and mild non-specific mental handicap in males has been reported by several groups10–12. We now report the cloning and characterization of a gene (FMR2) adjacent to FRAXE. Elements of FMR2 were initially identified from sequences deleted from a developmental delayed boy13. We correlate loss of FMR2 expression with (CCG)n expansion at FRAXE, demonstrating that this is a gene associated with the CpG island adjacent to FRAXE and contributes to FRAXE-associated mild mental retardation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Yu, S. et al. Fragile X genotype characterized by an unstable region of DNA. Science 252, 1179–1181 (1991).
Verkerk, A.J.M.H. et al. Identification of a gene (FMR-1) containing a CCG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell 65, 905–914 (1991).
Kremer, E.J. et al. Mapping of DNA instability at the fragile X to a trinucleotide repeat sequence p(CCG)n. Science 252, 1711–1714 (1991).
Fu, Y.H. et al. Variation of the CCG repeat at the fragile X site results in genetic instability: resolution of the Sherman paradox. Cell 67, 1047–1058 (1991).
Knight, S.J. et al. Trinucleotide repeat amplification and hypermethylation of a CpG island in FRAXE mental retardation. Cell 74, 127–134, (1993).
Parrish, J.E. et al. Isolation of a GCC repeat showing expansion in FRAXF, a fragile site distal to FRAXA and FRAXE. Nature Genet. 8, 229–235 (1994).
Nancarrow, J.K. et al. Implications of FRA16A structure for the mechanism of chromosomal fragile site genesis. Science 264, 1938–1941 (1994).
Jones, C. et al. Association of a chromosome deletion syndrome with fragile site within the proto-oncogene CBL2. Nature 376, 145–149 (1995).
Sutherland, G.R. & Baker, E. Characterization of a new rare fragile site easily confused with the fragile X. Hum. Mol. Genet. 1, 111–113 (1992).
Knight, S.J.L. et al. Triplet repeat expansion at the FRAXE locus and X-linked mild mental handicap. Am. J. Hum. Genet. 55, 81–86 (1994).
Hamel, B.C.J. et al. Segregation of FRAXE in a large family: Clinical, psychometric, cytogenetic, and molecular data. Am. J. Hum. Genet. 55, 923–931 (1994).
Mulley, J.C. et al. FRAXE and mental retardation. J. Med. Genet. 32, 162–169 (1995).
Gedeon, A.K. Overlapping submicroscopic deletions in Xq28 in two unrelated boys with developmental disorders: Identification of a gene near FRAXE. Am. J. Hum. Genet. 56, 907–914 (1995).
Brown, T. et al. Identification of a FRAXE microdeletion associated with a complex mosaic FRAXA mutation. 6th X Chromosome Workshop, S13 (1995).
Riggins, G.J. et al. Human genes containing polymorphic trinucleotide repeats. Nature Genet. 2, 186–191 (1992).
Blake, T.J., Shapiro, M., Morse, H.C. & Langdon, W.Y. The sequence of the human and mouse c-cbl proto-oncogenes shows proline -rich domain and leucine zipper-like motif. Oncogene. 6, 653–657 (1991).
Kozak, M. Point mutation define a sequence flanking the AUG initiation codon that modulates translation by eucaryotic ribosomes. Cell 44, 283–292 (1986).
Altchul, D. et al. Basic local alignment tool. J Mol. Biol. 215, 403–10 (1990).
Nakamura, T. et al. Genes on chromosomes 4, 9 and 19 involved in 11q23 abnormalities in acute leukemias share sequence sequence homology and/or common motifs. Proc. Natl. Acad. Sci. USA 90, 4631–4635 (1993).
Morrissey, J. et al. A serine/proline-rich protein is fused to HRX in t(4;11) acute leukemias. Blood 81, 1124–1131 (1993).
Mermod, N., O'Neil, E.A., Kelly, T.J. & Tjian, R. The proline-rich transcription activator of CTF/NF-I is distinct from the replication and DNA binding domain. Cell 58, 741–753 (1989).
Lehrke, R.G. A theory of X-linkage of major intellectual traits. Am. J. Med. Defic. 76, 611–619 (1972).
Gedeon, A.K., Donnelly, A.J., Bronwyn, K., Turner, G. & Mulley, J.C. How many X-linked genes for non-specific mental retardation (MRX) are there. Am. J. Med. Genet, (in the press).
Chomczynski, P. & Sacci, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162, 156–159 (1987).
Lee, E.Y. & Lee, W.H. Molecular cloning of the human esterase D gene, a genetic marker of retinoblastoma. Proc. Natl. Acad. Sci. USA 83, 6337–6341 (1986).
Kalatzis, V. Mapping of contiguous segments of DNA using yeast artificial chromosomes in the region distal to the fragile X. Honours thesis (The University of Adelaide, Adelaide, South Australia, 1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gecz, J., Gedeon, A., Sutherland, G. et al. Identification of the gene FMR2, associated with FRAXE mental retardation. Nat Genet 13, 105–108 (1996). https://doi.org/10.1038/ng0596-105
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0596-105