Abstract
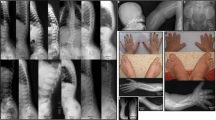
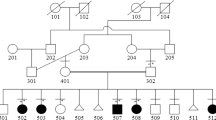
Achondroplasia (ACH) is a frequent condition of unknown origin characterized by short–limbed dwarfism and macrocephaly. Milder forms, termed hypochondroplasias (HCH) result in short stature with radiological features similar to those observed in ACH. We report on the mapping of a gene causing ACH/HCH to human chromosome 4p16.3, by linkage to the iduronidase A (IDUA) locus, in 15 informative families (Z max = 3.01 at θ = 0 for ACH; Z max = 4.71 at θ = 0 for ACH/HCH). Multipoint linkage analysis provides evidence for mapping the disease locus telomeric to D4S412 (location score in log 10 = 4.60). Moreover, this study supports the view that ACH and HCH are genetically homogeneous in our series.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Parrot, J.M. Sur les malformations achondroplasiques et le dieu Ptah. Bull. Anthropol. (Paris) 1, 296 (1878).
Maroteaux, P. & Lamy, M. Achondroplasia in man and animals. Clin. Orthop. 33, 91–103 (1964).
Ravenna. Achondroplasie et chondrohypoplasie. Nou. Iconog. Sapel. 26, 157–184 (1913).
Levy. Hypochondroplasie. Bull. Soc. Med. Hôp. Paris 48, 1780–1787 (1924).
Maroteaux, P. & Falzon, P. Hypochondroplasie. Revue de 80 cas. Arch. Fr. Pediat. 45, 105–109 (1988).
Oberklaid, F., Danks, D.M., Jensen, F., Stace, L. & Rosshandler, S. Achondroplasia and hypochondroplasia. J. med. Genet. 16, 140–146 (1979).
Wynne-Davies, T. et al. Achondroplasia and hypochondroplasia: clinical variation and spinal stenosis. J. bone joint Surg. 63B, 508–515 (1981).
McKusick, V.A., Kelly, T.E. & Dorst, J.P. Observations suggesting allelism of the achondroplasia and hypochondroplasia genes. J. med. Genet. 10, 11–16 (1973).
Sommer, A., Young-Wee, T. & Frye, T. Achondroplasia-Hypochondroplasia complex. Am. J. med. Genet. 26, 949–957 (1987).
Stanescu, V., Stanescu, R. & Maroteaux, P. Etude morphologique et blochimique du cartilage de croissance dans les astéochondrodysplasis. Arch. Fr. Pediat. XXXIV 3 (suppl) (1977).
Stanescu, R., Stanescu, V. & Maroteaux, P. Homozygous achondroplasia: morphologic and biochemical study of cartilage. Am. J. med. Genet. 37, 412–421 (1990).
Ponseti, I.V. Skeletal growth in achondroplasia. J. bone joint Surg. 52A, 701–716 (1970).
Murdoch, J.L. et al. Achondroplasia-a genetic and statistical survey. Ann. hum. Genet. 33, 227–244 (1970).
Orioli, I.M., Castilla, E.E. & Barbosa-Neto, J.G. The birth prevalence rates for the skeletal dysplasia. J. med. Genet. 23, 328–332 (1986).
Stoll, C., Dott, B., Roth, M.P. & Alembik, Y. Birth prevalence rates of skeletal dysplasias. Clin. Genet. 35, 88–92 (1989).
Bouvet, J.P., Maroteaux, P. & Felngold, J. Etude génétique de I'achondroplasie. Ann. Génét. 14, 127–131 (1971).
Hall, B.D. & Spranger, J. Hypochondroplasia: clinical and radiological aspects in 39 cases. Radiology 133, 95–100 (1979).
Gardner, R.J.M. A new estimate of the achondroplasia mutation rate. Clin. Genet. 11, 31–38 (1977).
Pulst, S.M. et al. The achondroplasia gene is not linked to the locus for neurofibromatosis 1 on chromosome 17. Hum. Genet. 85, 12–14 (1990).
Pulst, S.M. et al. Molecular analysis of a patient with neurofibromatosis 1 and achondroplasia. Am. J. med. Genet. 10, 84–87 (1991).
Ogilvie, D., Wordsworth, P., Thompson, E. & Sykes, B. Evidence against the structural gene encoding type II collagen (COL2A1) as the mutant locus in achondroplasia. J. med. Genet. 23, 19–22 (1986).
Carter, W.A. et al. SSCP and segregation analysis of the human type X collagen gene (COL10A1) in heritable forms of cnondrodysplasia. Am. J. hum. Genet. 51, 841–849 (1992).
Weissenbach, J. et al. A second generation linkage map of the human genome. Nature 359, 794–801 (1992).
Gusella, J.F. et al. Sequence tagged sits (STSs) spanning up 4p16.3 and the Huntington disease candidate region. Genomics 13, 75–80 (1992).
Scott, H.S., Nelson, P.V., Hopwood, J.J. & Morris, C.P. PCR of a VNTR linked to mucopolysaccharidosls type I and Huntington disease. Nuc. Acids Res. 19, 6348 (1991).
Maynard, J.A. et al. Histochemistry and ultrastructure of the growth plate in achondroplasia. J. bone Joint. Surg. 63A, 969–979 (1981).
Mullis, P.E., Patell, M.S., Brickell, P.M., Hindmarsh, P.C. & Brook, C. Growth characteristics and response to growth hormone therapy In patients with hypochondroplasia: genetic linkage of the insulin-like growth factor 1 gene at chromosome 12q23 to the disease in a subgroup of these patients. Clin. Endocrinol. 14, 265–274 (1991).
Wilson, H.G. Genetic and clinical studies in 13 patients with the Wolf-Hirschhorn syndrome (del 4p). Hum. Genet. 59, 397–307 (1981).
Tommerup, M. et al. A zinc finger gene ZNF141 mapping at 4p16.3/D4S90 is a candidate gene for the Wolf-Hirschhorn (4p-) syndrome. Hum. molec. Genet. 2, 1571–1575 (1993).
Weber, B. et al. Genomic organization and complete sequence of the human gene encoding the B-subunit of the cGMP phosphodiesterase and its localisation to 4p16.3. Nucl. Acid. Res. 19, 6263–6268 (1991).
Padanilam, B.J. et al. Characterization of the human HOX 7 cDNA and identification of polymorphic markers. Hum. molec. Genet. 1, 104–410 (1992).
Keegan, K., Johnson, D.E., Williams, L.T. & Hayman, M.J. Isolation of an additional member of the fibroblast growth factor receptor family FGFR3. Proc. natn. Acac. Sci. U.S.A. 88, 1095–1099 (1991).
Thompson, L.M. et al. A gene encoding a fibroblast growth factor receptor isolated from the Huntington disease gene region of human chromosome 4. Genomics 11, 1133–1142 (1991).
Burgess, W.H. & Maciag, T. The heparin-binding (fibroblast) growth factor family of proteins. Ann. Rev. Biochem. 58, 575–606 (1989).
Kato, Y. & Iwamoto, M. Fibroblast growth factor is an inhibitor of chondrocyte terminal differentiation. J. biol. Chem. 365, 5903–5909 (1990).
Iwamoto, M., Shimazu, A., Nakasnima, K., Susuki, F. & Kato, Y. Reduction in basic fibroblast growth factor receptor is coupled with terminal differentiation of chrondrocytes. J. biol. Chem. 266, 461–467 (1991).
Vignal, A. et al. Non radioactive multiplex procedure for genotyping of microsatellite markers. In Methods in Molecular Genetics: chromosome and gene analysis 1 (ed. Adolph, K.W.) 211–221 (Academic Press, Orlando, 1993).
Lathrop, G.M. & Lalouel, J.M. Easy calculation of lod scores and genetic risk on small computers. Am. J. hum. Genet. 36, 460–465 (1984).
Lathrop, G.M., Lalouel, J.M., Julier, C. & Ott, J. Multllocus linkage analysis in human. Detection of linkage and estimation of recombination. Am. J. hum. Genet. 37, 482–498 (1985).
Ott, J. Variability of the recombination fraction: analysis of human genetic linkage 112–115 (John Hopkins University Press, Baltimore, 1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Merrer, M., Rousseau, F., Legeai-Mallet, L. et al. A gene for achondroplasia–hypochondroplasia maps to chromosome 4p. Nat Genet 6, 318–321 (1994). https://doi.org/10.1038/ng0394-318
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0394-318
This article is cited by
-
Molecular modeling study of the induced-fit effect on kinase inhibition: the case of fibroblast growth factor receptor 3 (FGFR3)
Journal of Computer-Aided Molecular Design (2015)
-
Cenni di genetica e alterazioni morfologiche nello scheletro dell’acondroplasico
Archivio di Ortopedia e Reumatologia (2009)
-
Mutation analysis in Indian children with achondroplasia — utility of molecular diagnosis
The Indian Journal of Pediatrics (2009)
-
Genetic analysis of skeletal dysplasia: recent advances and perspectives in the post-genome-sequence era
Journal of Human Genetics (2006)
-
Rapid detection of FGFR3 gene mutation in achondroplasia by DHPLC system-coupling heteroduplex and fluorescence-enhanced primer-extension analysis
Journal of Human Genetics (2004)