Abstract
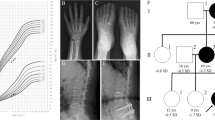
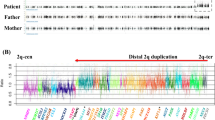
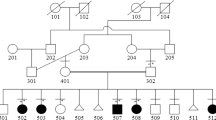
Achondroplasia is the most common type of genetic dwarfism. It is characterized by disproportionate short stature and other skeletal anomalies resulting from a defect in the maturation of the chondrocytes in the growth plate of the cartilage. We have now mapped the achondroplasia gene near the telomere of the short arm of chromosome 4 (4p16.3), by family linkage studies using 14 pedigrees. A positive lod score of z=3.35 with no recombinants was obtained with an intragenic marker for IDUA. This localization will facilitate the positional cloning of the disease gene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Scott, Jr., C. Dwarfism. CIBA-GEIGY Clinical Symposia 40, 3–7 (1988).
Hecht, J., Francomano, C., Horton, W. & Annegers, J. Mortality in achondroplasia. Am. J. hum. Genet. 41, 454–464 (1987).
Pauli, R. et al. Homozygous achondroplasia with survival beyond infancy. Am. J. med. Genet. 16, 459–473 (1983).
Orioli, I., Castilla, E. & Barbosa-Neto, J. The birth prevalence rates for skeletal dysplasias. J. med. Genet. 23, 328–332 (1986).
Stanescu, R., Stanescu, V. & Maroteaux, P. Homozygous achondroplasia: morphologic and biochemical study of cartilage. Am. J. med. Genet. 37, 412–421 (1990).
Pedrini-Mille, A. & Pedrini, V. Proteoglycans and glycosaminoglycans of human achondroplastic cartilage. J. Bone Joint Surg. 64-A, 39–46 (1982).
Finkelstein, J. et al. Analysis of the chondroitin sulfate proteoglycan core protein (CSPGCP) gene in achondroplasia and pseudoachondroplasia. Am. J. hum. Genet. 48, 97–102 (1991).
Ogilvie, D., Wordsworth, P., Thompson, E. & Sykes, B. Evidence against the structural gene encoding type II collagen (COL2A1) as the mutant locus in achondroplasia. J. med. Genet. 23, 19–22 (1986).
Francomano, C. & Pyeritz, R. Achondroplasia is not caused by mutation in the gene for type II collagen. Am. J. med. Genet. 29, 955–961 (1988).
Sweetman, W. et al. SSCP and segregation analysis of the human type X collagen gens (COL10A1) in heritable forms of chondrodysplasia. Am. J. hum. Genet. 51, 841–849 (1992).
Weiasenbach, J. et al. A second-generation linkage map of the human genome. Nature 359, 794–801 (1992).
Scott, H. et al. An 86-bp VNTR within IDUA is the basis of the D4S111 polymorphic locus. Genomics 14, 1118–1120 (1992).
Scott, H. et al. Chromosomal localization of the human α-L-lduronidase gene (IDUA) to 4p 16.3. Am. J. hum. Genet. 47, 802–807 (1990).
Magenis, R. et al. Huntington disease-linked restriction fragment length polymorphism localized within band p16.1 of chromosome 4 by in situ hybridization. Am. J. hum. Genet. 39, 383–391 (1986).
Wang, H. et al. Subregional assignment of the linked marker G8 (D4S1O) for Huntington disease to chromosome 4p16.1–16.3. Am. J. hum. Genet. 39, 393–396 (1986).
Scott, H. et al. Identification of mutations in the a-L-Iduronidase gene (IDUA) that causes Hurier and Scheie syndromes. Am. J. hum. Genet. 53, 973–986 (1993).
Cordone, M. et al. Ultrasonographlc features in a case of heterozygous achondroplasia at 25 weeks gestation. Pren. Diagnosis 13, 395–401 (1993).
McKusick, V.A., Kelly, T.E. & Dorst, J.P. Observations suggesting allelism of the achondroplasia and hypochondroplasia genes. J. med. Genet. 10, 11–16 (1973).
Schinzel, A. Catalogue of unbalanced chromosome aberrations in man. 161–164 (Walter de Gruyter, Berlin, 1984).
Gandelman, K-Y., Gibson, L., Meyn, M. & Yang-Feng, T.L. Molecular definition of the smallest region of deletion overlap in the Wolf–Hirschhom syndrome. Am. J. hum. Genet. 51, 571–578 (1992).
Lin, C. et al. New DNA markers in the Huntington's disease gene candidate region. Som. Cell molec. Genet. 17, 481–488 (1991).
Weber, B. et al. Isolation and characterization of new highly polymorphic DNA markers from the Huntington disease region. Am. J. hum. Genet. 47, 382–393 (1992).
Tommerup, N. et al. A Zinc-finger gene ZNF141 mapping at 4p16.3/D4S90 is a candidate gene for the Wolf-Hirschhom (4p-) syndrome. Hum. molec. Genet. 2, 1571–1576 (1993).
Taylor, S. et al. Cloning of the a-adducin gene from the Huntington's disease candidate region of chromosome 4 by exon amplification. Nature Genet. 2, 223–227 (1992).
Ambrose, C. et al. A novel G protein-coupled receptor kinase gene cloned from 4p16.3. Hum. molec. Genet. 1, 697–703 (1992).
Duyao, M. et al. A gene from chromosome 4p16.3 with similarity to a superfamily of transporter proteins. Hum. molec. Genet. 2, 673–676 (1993).
Collins, C., Schappert, K. & Hayden, M. The genomic organization of a novel regulatory myosin light chain gene (MYL5) that maps to chromosome 4p16.3 and shows different patterns of exxpression between primates. Hum. molec. Genet. 1, 727–733 (1992).
Weber, B. et al. Genomic organization and complete sequence of the human gene encoding the β-subunit of the cGMP phosphodiesterase and its localization to 4p16.3. Nucl. Acids Res. 19, 6263–6268 (1991).
Padanilam, B. et al. Characterization of the human HOX7 cDNA and identification of polymorphic markers. Hum. molec. Genet. 1, 407–410 (1992).
Stadler, H. et al. Identification and genetic mapping of a homeobox gene to the 4p16.1 region of human chromosome 4. Proc. natn. Acad. Sci. U.S.A. 89, 11579–11583 (1992).
Thompson, L. et al. A gene encoding a flbroblast growth factor receptor isolated from the Huntington disease gene region of human chromosome 4. Genomics 11, 1133–1142 (1991).
Peters, K., Ornitz, D., Werner, S. & Williams, L. Unique expression pattern of the FGF receptor 3 gene during mouse organogenesis. Dev. Biol. 155, 423–430 (1993).
Langer, Jr., L., Schaefer, B. & Wadswoith, D. Patient with double heterozygosity for achondroplasia and pseudoachondroplasia, with comments on these conditions and the relationship between pseudoachondroplasia and multiple epiphyseal dysplasia, Fairbank type. Am. J. med. Genet. 47, 772–781 (1993).
Vignal, A. et al. Nonradioactive multiplex procedure for genotyping of microsatellite markers. Meth. molec. Genet. 1, 211–221 (1993).
Alitto, B. et al. Assay by polymerase chain reaction (PCR) of multi-allele polymorphisms in the Huntington's disease region of chromosome 4. Molec. cell. Probes 6, 513–520 (1992).
Taylor, S., Barnes, G., MacDonald, M. & Gusella, J. A dinucleotide repeat polymorphism at the D4S127 locus. Hum. molec. Genet. 1, 142 (1992).
Lathrop, G., Lalouel, J., Julier, C. & Ott, J. Strategies for multi-point linkage analysis in humans. Proc. natn. Acad. Sci. U.S.A. 81, 3443–3446 (1984).
Locke, P. et al. A genetic linkage map of the chromosome 4 short arm. Som. Cell molec. Genet. 19, 95–101 (1993).
Murray, J. et al. Cooperative Human Linkage Center Report 1 (1) 1993.
Lander, E. & Green, P. Construction of multi-locus genetic linkage maps in humans. Proc. natn. Acad. Sci. U.S.A. 85, 2363–2367 (1987).
MacDonald, M. et al. Huntington disease-linked locus D4S111 exposed as the α-L-lduronidase gene. Som. Cell molec. Genet. 17, 421–425 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Velinov, M., Slaugenhaupt, S., Stoilov, I. et al. The gene for achondroplasia maps to the telomeric region of chromosome 4p. Nat Genet 6, 314–317 (1994). https://doi.org/10.1038/ng0394-314
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0394-314
This article is cited by
-
A single nucleotide mutation in Nppc is associated with a long bone abnormality in lbab mice
BMC Genetics (2007)
-
Genetic analysis of skeletal dysplasia: recent advances and perspectives in the post-genome-sequence era
Journal of Human Genetics (2006)
-
Rapid detection of FGFR3 gene mutation in achondroplasia by DHPLC system-coupling heteroduplex and fluorescence-enhanced primer-extension analysis
Journal of Human Genetics (2004)
-
A recurrent mutation in the tyrosine kinase domain of fibroblast growth factor receptor 3 causes hypochondroplasia
Nature Genetics (1995)