Abstract
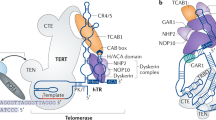
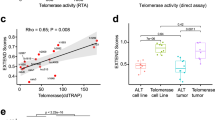
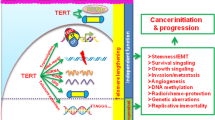
Telomeres consist of short repeated sequences that are synthesized by telomerase, a ribonucleo-protein DMA polymerase1–4. Telomerase activity is present in many tumours and not detected in many normal tissues5–7. Telomere shortening in human and mouse tissues and primary cell cultures may be due to the absence of telomerase activity8–11. To determine when telomerase is activated during tumour development and progression, we examined telomerase activity and expression of the recently cloned mouse telomerase RNA component (mTR)12 in two different transgenic mouse models of multi-stage tumorigenesis. These mouse models allow examination of many independent tumours from genetically identical individuals. These mice reproducibly develop pancreatic islet cell carcinoma13 and squamous cell carcinoma of the skin14. In both carcinoma types, we detected telomerase activity only in late-stage tumours; in contrast, we found mTR levels were upregulated in the early pre-neoplastic stages, and further increased during progression. Surprisingly, mTR levels did not parallel the amount of telomerase activity detected and a subset of tumours lacked telomerase activity and yet expressed telomerase RNA. Regulation of telomerase activity may therefore be separable from expression of its RNA component. These results clearly demonstrate telomerase is activated in late stages of tumour progression, and show for the first time that the initial up regulation of telomerase RNA is an early event, concurrent with the hyperproliferation elicited by viral oncogenes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Blackburn, E.H. Structure and function of telomeres. Nature 350, 569–573 (1991).
Greider, C.W. & Blackburn, E.H. Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 43, 405–413 (1985).
Greider, C.W. & Blackburn, E.H. A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature 337, 331–337 (1989).
Collins, K., Kobayashi, R. & Greider, C.W. Purification of Tetrahymena telomerase and cloning of the genes for the two protein components of the enzyme. Cell 81, 677–686 (1995).
Counter, G.M., Hirte, H.W., Bacchetti, S. & Harley, C.B. Telomerase activity in human ovarian carcinoma. Proc. Natl. Acad. Sci. USA 91, 2900–2904 (1994).
Kim, N.W. et al. Specific association of human telomerase activity with immortal cells and cancer. Science 266, 2011–2014 (1994).
Chadeneau, C., Siegel, P., Harley, C.B., Muller, W. & Bacchetti, S. Telomerase activity in normal and malignant murine tissues. Oncogene 11, 893–898 (1995).
Harley, C.B., Futcher, A.B. & Greider, C.W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990).
Hastie, N.D. et al. Telomere reduction in human colorectal carcinoma and with ageing. Nature 346, 866–868 (1990).
de Lange, T. et al. Structure and variability of human chromosome ends. Mol. Cell. Bid. 10, 518–527 (1990).
Counter, C.M. et al. Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. EMBO J. 11, 1921–1929 (1992).
Blasco, M., Funk, W., Villeponteau, B. & Greider, C.W. Functional characterization and developmental regulation of mouse telomerase RNA. Science 269, 1267–1270 (1995).
Hanahan, D. Heritable formation of pancreatic β–cell tumors in transgenic mice expressing recombinant insuline/simian virus 40 oncogenes. Nature 315, 115–122 (1985).
Arbeit, J.M., Munger, K., Howley, P.M. & Hanahan, D. Progressive squamous epithelial neoplasia in K14-human papillomavirus type 16 transgenic mice. J. Virol. 68, 4358–4368 (1994).
Hurlin, P.J. et al. Myc and mad are inversely regulated during epidermal differentiation and HPV–associated tumorigenesis. Oncogene(in the press).
Broccoli, D., Young, J.W. & de Lange, T. Telomerase activity in normal and malignant heamatopoietic cells. Proc. Natl. Acad. Sci. USA 92, 9082–9082 (1995).
Counter, C.M., Gupta, J., Harley, C.B., Leber, B. & Bacchetti, S. Telomerase activity in normal leukocytes and in hematologic malignancies. Blood 85, 2315–2320 (1995).
Hiyama, K. et al. Activation of telomerase in human lymphocytes and hematopoietic progenitor cells. J. Immun.(in the press).
Christofori, G., Naik, P. & Hanahan, D. A second signal supplied by insuline–like growth factor II in oncogene–induced tumorigenesis. Nature 369, 414–418 (1994).
Folkman, J., Watson, J., Ingber, K. & Hanahan, D. Induction of angiogenesis during the transition from hyperplasia to neoplasia. Nature 339, 58–61 (1989).
Bacchetti, S. & Counter, C.M. Telomeres and telomerase in human cancer (Review). Intl. J. Oncd. 7, 423–432 (1995).
Harley, C.B. & Villeponteau, B. Telomeres and telomerase in aging and cancer. Curr. Opin. Genet. Dev. 5, 249–255 (1995).
Shay, J.W. & Wright, W.E. Telomerase activity in human cancer. Curr Opin. Oncol. (in the press).
Counter, C.M., Botelho, F.M., Wang, P., Harley, C.B. & Bacchetti, S. Stabilization of short telomeres and telomerase activity accompany immortalization of Epstein–Barr virus transfromed human B lymphocytes. J. Virol. 68, 3410–3414 (1994).
Feng, J. et al. The human telomerase RNA component. Science 269, 1236–1241 (1995).
Avilion, A.A. et al. Human telomerase activity and RNA component levels in immortal cell lines and tumor tissues. Cancer Res.(in the press).
Gotoh, M., Maki, T., Kiyoizumi, T., Satomi, S & Monaco, A.P. An improved method for isolation of mouse pancreatic islets. Transplantation 40, 437–438 (1985).
Ausubel, P.M. et al. Current Protocols in Molecular Biology (Wiley and Sons, Inc., New York, 1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blasco, M., Rizen, M., Greider, C. et al. Differential regulation of telomerase activity and telomerase RNA during multi-stage tumorigenesis. Nat Genet 12, 200–204 (1996). https://doi.org/10.1038/ng0296-200
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0296-200
This article is cited by
-
Telomerase deficiency and dysfunctional telomeres in the lung tumor microenvironment impair tumor progression in NSCLC mouse models and patient-derived xenografts
Cell Death & Differentiation (2023)
-
TERC suppresses PD-L1 expression by downregulating RNA binding protein HuR
Science China Life Sciences (2022)
-
Translational research in neuroendocrine tumors: pitfalls and opportunities
Oncogene (2017)
-
Genetic and epigenetic trends in telomere research: a novel way in immunoepigenetics
Cellular and Molecular Life Sciences (2015)
-
A non-canonical function of telomerase RNA in the regulation of developmental myelopoiesis in zebrafish
Nature Communications (2014)