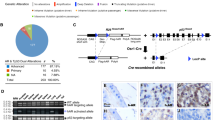
Abstract
The genetic bases underlying prostate tumorigenesis are poorly understood. Inactivation of the tumor-suppressor gene PTEN and lack of p27KIP1 expression have been detected in most advanced prostate cancers1,2. But mice deficient for Cdkn1b (encoding p27Kip1) do not develop prostate cancer3,4,5. PTEN activity leads to the induction of p27KIP1 expression, which in turn can negatively regulate the transition through the cell cycle6. Thus, the inactivation of p27KIP1 may be epistatic to PTEN in the control of the cell cycle. Here we show that the concomitant inactivation of one Pten allele and one or both Cdkn1b alleles accelerates spontaneous neoplastic transformation and incidence of tumors of various histological origins. Cell proliferation, but not cell survival, is increased in Pten+/−/Cdkn1b−/− mice. Moreover, Pten+/−/Cdkn1b−/− mice develop prostate carcinoma at complete penetrance within three months from birth. These cancers recapitulate the natural history and pathological features of human prostate cancer. Our findings reveal the crucial relevance of the combined tumor-suppressive activity of Pten and p27Kip1 through the control of cell-cycle progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cairns, P. et al. Frequent inactivation of PTEN/MMAC1 in primary prostate cancer. Cancer Res. 57, 4997–5000 (1997).
Macri, E. & Loda, M. Role of p27 in prostate carcinogenesis. Cancer Metastasis Rev. 17, 337–344 (1998).
Fero, M.L. et al. A syndrome of multiorgan hyperplasia with features of gigantism, tumorigenesis, and female sterility in p27(Kip1)-deficient mice. Cell 85, 733–744 (1996).
Kiyokawa, H. et al. Enhanced growth of mice lacking the cyclin-dependent kinase inhibitor function of p27(Kip1). Cell 85, 721–732 (1996).
Nakayama, K. et al. Mice lacking p27(Kip1) display increased body size, multiple organ hyperplasia, retinal dysplasia, and pituitary tumors. Cell 85, 707–720 (1996).
Li, D.M. & Sun, H. PTEN/MMAC1/TEP1 suppresses the tumorigenicity and induces G1 cell cycle arrest in human glioblastoma cells. Proc. Natl. Acad. Sci. USA 95, 15406–15411 (1998).
Landis, S.H., Murray, T., Bolden, S. & Wingo, P.A. Cancer statistics, 1998. CA Cancer J. Clin. 48, 6–29 (1998).
Verma, R.S., Manikal, M., Conte, R.A. & Godec, C.J. Chromosomal basis of adenocarcinoma of the prostate. Cancer Invest. 17, 441–447 (1999).
Di Cristofano, A., Pesce, B., Cordon-Cardo, C. & Pandolfi, P.P. Pten is essential for embryonic development and tumour suppression. Nature Genet. 19, 348–355 (1998).
Podsypanina, K. et al. Mutation of Pten/Mmac1 in mice causes neoplasia in multiple organ systems. Proc. Natl. Acad. Sci. USA 96, 1563–1568 (1999).
Cordon-Cardo, C. et al. Distinct altered patterns of p27KIP1 gene expression in benign prostatic hyperplasia and prostatic carcinoma. J. Natl. Cancer Inst. 90, 1284–1291 (1998).
Di Cristofano, A. et al. Impaired Fas response and autoimmunity in Pten+/− mice. Science 285, 2122–2125 (1999).
Kurman, R.J., Kaminski, P.F. & Norris, H.J. The behavior of endometrial hyperplasia. A long-term study of “untreated” hyperplasia in 170 patients. Cancer 56, 403–412 (1985).
Di Cristofano, A. & Pandolfi, P.P. The multiple roles of PTEN in tumor suppression. Cell 100, 387–390 (2000).
Price, D. Comparative aspects of development and structure in the prostate. in Biology of the Prostate and Related Tissues (eds. Vollmer, E.P. & Kauffman, G.) 1–28 (US Government Printing Office, Washington, DC, 1963).
McNeal, J.E. Origin and development of carcinoma in the prostate. Cancer 23, 24–34 (1969).
Green, J.E. et al. Workgroup 3: transgenic and reconstitution models of prostate cancer. Prostate 36, 59–63 (1998).
Osman, I. et al. Alterations affecting the p53 control pathway in bilharzial-related bladder cancer. Clin. Cancer Res. 3, 531–536 (1997).
Acknowledgements
We thank C. Abate-Shen, M. Shen, P. Scardino and H.I. Scher for advice and discussions; M. Jiao for the preparation of pathological samples; and S. Kim for critical suggestions concerning the Pten immunohistochemical analysis. P.P.P. is a Scholar of the Leukemia & Lymphoma Society of America (formerly known as the Leukemia Society of America). This work was supported by the NCI and by the I.T. Hirschl/M. Weill-Caulier Foundation to P.P.P.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Cristofano, A., De Acetis, M., Koff, A. et al. Pten and p27KIP1 cooperate in prostate cancer tumor suppression in the mouse. Nat Genet 27, 222–224 (2001). https://doi.org/10.1038/84879
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/84879
This article is cited by
-
Lineage tracing of mutant granulosa cells reveals in vivo protective mechanisms that prevent granulosa cell tumorigenesis
Cell Death & Differentiation (2023)
-
Reciprocal negative feedback regulation of ATF6α and PTEN promotes prostate cancer progression
Cellular and Molecular Life Sciences (2023)
-
Akt: a key transducer in cancer
Journal of Biomedical Science (2022)
-
The equilibrium of tumor suppression: DUBs as active regulators of PTEN
Experimental & Molecular Medicine (2022)
-
Identification of a Cowden syndrome patient with a novel PTEN mutation and establishment of patient-derived induced pluripotent stem cells
In Vitro Cellular & Developmental Biology - Animal (2022)