Abstract
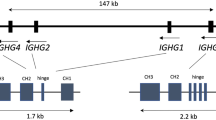
We have constructed the physical map of the 0.8 megabase DNA fragment which contains the 3′ 64 variable region (V) gene segments of the human immunoglobulin heavy chain (H) locus. The organization of the VH locus showed several features that indicate dynamic reshuffling of this locus. The sequenced 64 VH segments include 31 pseudogenes, of which 24 are highly conserved except for a few point mutations. Comparison of the 64 germline VH sequences shows that each VH family has conserved sequences, suggesting that there might be some genetic or selection mechanisms involved in maintenance of each family. The total number of the human VH segments was estimated to be about 120, including at least 7 orphons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Tonegawa, S. Somatic generation of antibody diversity. Nature 302, 575–581 (1983).
Honjo, T. & Habu, S. Origin of immune diversity: genetic variation and selection. Ann. Rev. Biochem. 54, 803–830 (1985).
Croce, C.M. et al. Chromosomal location of the genes for human immunoglobulin heavy chains. Proc. natn. Acad. Sci. U.S.A. 76, 3416–3419 (1979).
Cherif, D. & Berger, R. New localization of VH sequences by in situ hybridization with biotinylated probes. Genes Chromosomes Cancer 2, 103–108 (1990).
Matsuda, F. et al. Organization of variable region segments of the human immunoglobulin heavy chain: duplication of the D5 cluster within the locus and interchromosomal translocation of variable region segments. EMBO J. 9, 2501–2506 (1990).
Matsuda, F. et al. Dispersed localization of D segments in the human immunoglobulin heavy-chain locus. EMBO J. 7, 1047–1051 (1988).
Berman, J.E. et al. Content and organization of the human Ig VH locus: definition of three new VH families and linkage to the Ig CH locus. EMBO J. 7, 723–733 (1988).
Kodaira, M. et al. Organization and evolution of variable region genes of the human immunoglobulin heavy chain. J. molec. Biol. 190, 529–541 (1986).
Brodeur, P.H., Osman, G.E., Mackle, J.J. & Lalor, T.M. The organization of the mouse Igh-V locus. J. exp. Med. 168, 2261–2278 (1988).
Yang, P.H. et al. Possible deletion of a developmental regulated heavy-chain variable region gene in autoimmune diseases. Proc. natn. Acad. Sci. U.S.A. 88, 7907–7911 (1990).
Walter, M.A., Gibson, W.T., Ebers, G.C. & Cox, D.W. Susceptibility to multiple sclerosis is associated with the proximal immunoglobulin heavy chain variable region. J. clin. Invest. 87, 1266–1273 (1991).
Yancopoulos, G.D. et al. Preferential utilization of the most JH proximal VH gene segments in pre-B cell lines. Nature 311, 727–733 (1984).
Schroeder, H.W. Jr., Hillson, J.L. & Perlmutter, R.M. Early restriction of the human antibody repertoire. Science 238, 791–793 (1987).
Humphries, C.G. et al. A new human immunoglobulin VH family preferentially rearranged in immature B-cell tumors. Nature 331, 446–449 (1988).
Shin, E.K. et al. Physical map of the 3′ region of the human immunoglobulin heavy chain locus: clustering of autoantibody-related variable segments in one haplotype. EMBO J. 10, 3641–3645 (1991).
Walter, M.A., Surti, U., Hofker, M.H. & Cox, D.W. The physical organization of the human immunoglobulin heavy chain gene complex. EMBO J. 9, 3303–3313 (1990).
Lorenz, W., Straubinger, B. & Zachau, H.G. Physical map of the human immunoglobulin k locus and its implication for the mechanisms of Vκ-Jκ rearrangement. Nucl. Acids Res. 15, 9667–9676 (1987).
Lee, K.H., Matsuda, F., Kinashi, T., Kodaira, M. & Honjo, T. A novel family of variable region genes of the human immunoglobulin heavy chain. J. molec. Biol. 195, 761–768 (1987).
Schroeder, H.W. Jr., & Wang, J.Y. Preferential utilization of conserved immunoglobulin heavy chain variable gene segments during human fetal life. Proc. natn. Acad. Sci. U.S.A. 87, 6146–6150 (1990).
Pascual, V. et al. The complete nucleotide sequences of the heavy chain variable regions of six monospecific rheumatoid factors derived from Epstein-Barr virus-transformed B cells isolated from the synovial tissue of patients with rheumatoid arthritis. J. clin. Invest. 86, 1320–1328 (1990).
Cairns, E. et al. Analysis of variable region genes encoding a human anti-DNA antibody of normal origin. J. Immunol. 143, 685–691 (1989).
Sanz, I., Dang, H., Takei, M., Talal, N. & Capra, J.D. VH sequence of a human anti-Sm autoantibody. J. Immunol. 142, 883–887 (1989).
Dersimonian, H., Schwartz, R.S., Barrett, K.J. & Stollar, B.D. Relationship of human variable region heavy chain germ-line genes to genes encoding anti-DNA autoantibodies. J. Immunol. 139, 2496–2501 (1987).
Imai, T. & Olson, M.V. Second-generation approach to the construction of yeast artificial-chromosome libraries. Genomics 8, 297–303 (1990).
Anand, R., Villasante, A. & Tyler-Smith, C. Construction of yeast artificial chromosome libraries with large inserts using fractionation by pulsed-field gel electrophoresis. Nucl. Acids Res. 17, 3425–3433 (1989).
Green, E.D. & Olson, M.V. Systematic screening of yeast artificial chromosome libraries by use of the polymerase chain reaction. Proc. natn. Acad. Sci. U.S.A. 89, 1213–1217 (1990).
Sambrook, J., Fritsch, F.F. & Maniatis, T. Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Laboratory Press, New York, 1989).
Gibson, T.J., Coulson, A.R., Sulston, J.E. & Little, P.F.R. Lorist 2, a cosmid with transcriptional terminators insulating vector genes from interference by promoters within the insert: effect on DMA yield and cloned insert frequency. Gene 53, 275–281 (1987).
Nishida, Y., Miki, T., Hisajima, H. & Honjo, T. Cloning of human immunoglobulin epsilon chain genes: evidence for multiple C-epsilon genes. Proc. natn. Acad. Sci. U.S.A. 79, 3833–3837 (1982).
Takahashi, N., Noma, T. & Honjo, T. Rearranged immunoglobulin heavy chain variable region (VH) pseudogene that delete the second complementarity-determining region. Proc. natn. Acad. Sci. U.S.A. 81, 5194–5198 (1984).
Southern, E.M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J. molec. Biol. 98, 503–517 (1975).
Burke, D.T., Carle, G.F. & Olson, M.V. Cloning of large segments of exogenous DMA into yeast by means of artificial chromosome vectors. Science 236, 806–812 (1987).
Kabat, E.A., Wu, T.T., Perry, H.M., Gottesman, K.S. & Foeller, C. Sequences of Proteins of Immunological Interest (NIH publications, Washington D. C., 1991).
Ebeling, S.B. et al. Expression of members of the immunoglobulin VH3 gene families is not restricted at the level of individual genes in human chronic lymphocytic leukemia. Int. Immunol. 4, 313–320 (1992).
Dersimonian, H., Mcadam, K.P.W.J., Mackworth-Young, C. & Stollar, B.D. The recurrent expression of variable region segments in human IgM anti-DNA autoantibodies. J. Immunol. 142, 4027–4033 (1989).
Felgenhauer, M., Kohl, J. & Ruker, F. Nucleotide sequences of the cDNAs encoding the V-regions of H- and L-chains of a human monoclonal antibody specific to HIV-gp41. Nucl. Acids Res. 18, 4927 (1990).
Mortari, F., Newton, J.A., Wang, J.Y. & Schroeder, Jr., H.W. The human cord blood antibody repertoire. Frequent usage of the VH7 gene family. Eur. J. Immunol. 22, 241–245 (1992).
Adderson, E.E., Shackelford, P.G., Quinn, A. & Carroll, W.L. Restricted IgH chain V gene usage in the human antibody response to Haemophilus influenzae type b capsular polysaccharide. J. Immunol. 147, 1667–1674 (1991).
Friedman, D.F. et al. The role of clonal selection in the pathogenesis of an autoreactive human B cell lymphoma. J. exp. Med. 174, 525–537 (1991).
Kishimoto, T., Okajima, H., Okumoto, T. & Taniguchi, M. Nucleotide sequences of the cDNAs encoding the V-regions of H- and L-chains of a human monoclonal antibody with broad reactivity to malignant tumor. Nucl. Acids Res. 17, 4385 (1989).
Matthyssens, G. & Rabbits, T.H. Structure and multiplicity of genes for the human immunoglobulin heavy chain variable region. Proc. natn. Acad. Sci. U.S.A. 77, 6561–6565 (1980).
Sanz, I., Casali, P., Thomas, J.W., Notkins, A.L. & Capra, J.D. Nucleotide sequences of eight human natural autoantibody VH regions reveals apparent restricted use of VH families. J. Immunol. 142, 4054–4061 (1989).
Sanz, I. et al. The smaller human VH gene families display remarkably little polymorphism. EMBO J. 8, 3471–3478 (1989).
Nickerson, K.G., Berman, J., Clickman, E., Chess, L. & Alt, F.W. Early human IgH gene assembly in Epstein-Barr virus-transfected fetal B cell lines. J. exp. Med. 169, 1391–1403 (1989).
Chen, P.P. Scand. Structural analyses of human developmentally regulated Vh3 genes. J. Immunol. 31, 257–267 (1990).
Akahori, Y., Kurosawa, Y., Kamachi, Y., Torii, S. & Matsuoka, H. Presence of immunoglobulin (Ig) M and IgG double isotype-bearing cells and defect of switch recombination in hyper IgM immunodeficiency. J. clin. Invest. 85, 1722–1727 (1990).
Silverstein, L.E., Litwin, S. & Carmack, C.E. Relationship of variable region genes expressed by a human B cell lymphoma secreting pathologic anti-Pr2 erythrocyte autoantibodies. J. exp. Med. 169, 1631–1643 (1989).
Harindranath, N. et al. Complete sequence of the genes encoding the VH and VL regions of low- and high-affinity monoclonal IgM and lgA1 rheumatoid factors produced by CD5+ B cells from a rheumatoid arthritis patient. Int. Immunol. 3, 865–875 (1991).
Milili, M., Fougereau, M., Guglielmi, P. & Schiff, C. Early occurrence of immunoglobulin isotype switching in human fetal liver. Molec. Immunol. 28, 753–761 (1991).
Rechavi, G., Ram, D., Glazer, L., Zakut, R. & Givol, D. Evolutionary aspects of immunoglobulin heavy chain variable region (VH) gene subgroups. Proc. natn. Acad. Sci. U.S.A. 80, 855–859 (1983).
Shen, A., Humphries, C., Tucker, P. & Blattner, F. Human heavy-chain variable region gene family nonrandomly rearranged in familial chronic lymphocytic leukemia. Proc. natn. Acad. Sci. U.S.A. 84, 8563–8567 (1987).
Rechavi, G. et al. Organization and evolution of immunoglobulin V-H gene subgroups. Proc. natn. Acad. Sci. U.S.A. 79, 4405–4409 (1982).
Alexander, A., Anicito, I. & Buxbaum, J. Gamma heavy chain disease in man. J. clin. Invest. 82, 1244–1252 (1988).
Hughes-Jones, N.C., Bye, J.M., Beale, D. & Coadwell, J. Nucleotide sequences and three-dimensional modeling of the Vh and V1 domains of two human monoclonal antibodies specific for the D antigen of the human Rh-blood-group system. Biochem. J. 268, 135–140 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Matsuda, F., Kyun Shin, E., Nagaoka, H. et al. Structure and physical map of 64 variable segments in the 3′ 0.8–megabase region of the human immunoglobulin heavy–chain locus. Nat Genet 3, 88–94 (1993). https://doi.org/10.1038/ng0193-88
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0193-88
This article is cited by
-
Association between a common immunoglobulin heavy chain allele and rheumatic heart disease risk in Oceania
Nature Communications (2017)
-
Human Antibody Production in Transgenic Animals
Archivum Immunologiae et Therapiae Experimentalis (2015)
-
Exploration of Factors Affecting the Onset and Maturation Course of Follicular Lymphoma through Simulations of the Germinal Center
Bulletin of Mathematical Biology (2009)
-
Determination of gene organization in the human IGHV region on single chromosomes
Genes & Immunity (2005)
-
Association between telomere length and VH gene mutation status in chronic lymphocytic leukaemia: clinical and biological implications
British Journal of Cancer (2003)