Abstract
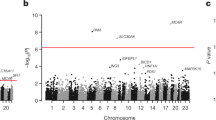
Metformin is the most commonly used pharmacological therapy for type 2 diabetes. We report a genome-wide association study for glycemic response to metformin in 1,024 Scottish individuals with type 2 diabetes with replication in two cohorts including 1,783 Scottish individuals and 1,113 individuals from the UK Prospective Diabetes Study. In a combined meta-analysis, we identified a SNP, rs11212617, associated with treatment success (n = 3,920, P = 2.9 × 10−9, odds ratio = 1.35, 95% CI 1.22–1.49) at a locus containing ATM, the ataxia telangiectasia mutated gene. In a rat hepatoma cell line, inhibition of ATM with KU-55933 attenuated the phosphorylation and activation of AMP-activated protein kinase in response to metformin. We conclude that ATM, a gene known to be involved in DNA repair and cell cycle control, plays a role in the effect of metformin upstream of AMP-activated protein kinase, and variation in this gene alters glycemic response to metformin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Nathan, D.M. et al. Medical management of hyperglycaemia in type 2 diabetes mellitus: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 52, 17–30 (2009).
NICE clinical guideline 87. Type 2 diabetes: the management of type 2 diabetes. (National Institute for Health and Clinical Excellence, London, UK, 2009).
Zhou, G. et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108, 1167–1174 (2001).
Owen, M.R., Doran, E. & Halestrap, A.P. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem. J. 348, 607–614 (2000).
Hawley, S.A. et al. Use of cells expressing gamma subunit variants to identify diverse mechanisms of AMPK activation. Cell Metab. 11, 554–565 (2010).
Donnelly, L.A., Doney, A.S., Hattersley, A.T., Morris, A.D. & Pearson, E.R. The effect of obesity on glycaemic response to metformin or sulphonylureas in Type 2 diabetes. Diabet. Med. 23, 128–133 (2006).
Dupuis, J. et al. New genetic loci implicated in fasting glucose homeostasis and their impact on type 2 diabetes risk. Nat. Genet. 42, 105–116 (2010).
Boder, E. Ataxia-telangiectasia: an overview. Kroc Found. Ser. 19, 1–63 (1985).
Schalch, D.S., McFarlin, D.E. & Barlow, M.H. An unusual form of diabetes mellitus in ataxia telangiectasia. N. Engl. J. Med. 282, 1396–1402 (1970).
Bar, R.S. et al. Extreme insulin resistance in ataxia telangiectasia: defect in affinity of insulin receptors. N. Engl. J. Med. 298, 1164–1171 (1978).
Sun, Y., Connors, K.E. & Yang, D.Q. AICAR induces phosphorylation of AMPK in an ATM-dependent, LKB1-independent manner. Mol. Cell. Biochem. 306, 239–245 (2007).
Fu, X., Wan, S., Lyu, Y.L., Liu, L.F. & Qi, H. Etoposide induces ATM-dependent mitochondrial biogenesis through AMPK activation. PLoS One 3, e2009 (2008).
Sanli, T. et al. Ionizing radiation activates AMP-activated kinase (AMPK): a target for radiosensitization of human cancer cells. Int. J. Radiat. Oncol. Biol. Phys. 78, 221–229 (2010).
Lavin, M.F. Ataxia-telangiectasia: from a rare disorder to a paradigm for cell signalling and cancer. Nat. Rev. Mol. Cell Biol. 9, 759–769 (2008).
Schneider, J.G. et al. ATM-dependent suppression of stress signaling reduces vascular disease in metabolic syndrome. Cell Metab. 4, 377–389 (2006).
Miles, P.D., Treuner, K., Latronica, M., Olefsky, J.M. & Barlow, C. Impaired insulin secretion in a mouse model of ataxia telangiectasia. Am. J. Physiol. Endocrinol. Metab. 293, E70–E74 (2007).
Trinklein, N.D., Aldred, S.J., Saldanha, A.J. & Myers, R.M. Identification and functional analysis of human transcriptional promoters. Genome Res. 13, 308–312 (2003).
Fukao, T. et al. ATM is upregulated during the mitogenic response in peripheral blood mononuclear cells. Blood 94, 1998–2006 (1999).
Savitsky, K. et al. Ataxia-telangiectasia: structural diversity of untranslated sequences suggests complex post-transcriptional regulation of ATM gene expression. Nucleic Acids Res. 25, 1678–1684 (1997).
Gudmundsson, J. et al. Two variants on chromosome 17 confer prostate cancer risk, and the one in TCF2 protects against type 2 diabetes. Nat. Genet. 39, 977–983 (2007).
Libby, G. et al. New users of metformin are at low risk of incident cancer: a cohort study among people with type 2 diabetes. Diabetes Care 32, 1620–1625 (2009).
Huang, X. et al. Important role of the LKB1-AMPK pathway in suppressing tumorigenesis in PTEN-deficient mice. Biochem. J. 412, 211–221 (2008).
Morris, A.D. et al. The diabetes audit and research in Tayside Scotland (DARTS) study: electronic record linkage to create a diabetes register. DARTS/MEMO Collaboration. Br. Med. J. 315, 524–528 (1997).
Anonymous. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352, 854–865 (1998).
Kahn, S.E. et al. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N. Engl. J. Med. 355, 2427–2443 (2006).
Barrett, J.C. et al. Genome-wide association study of ulcerative colitis identifies three new susceptibility loci, including the HNF4A region. Nat. Genet. 41, 1330–1334 (2009).
Zeggini, E. et al. Replication of genome-wide association signals in UK samples reveals risk loci for type 2 diabetes. Science 316, 1336–1341 (2007).
Marchini, J., Howie, B., Myers, S., McVean, G. & Donnelly, P. A new multipoint method for genome-wide association studies by imputation of genotypes. Nat. Genet. 39, 906–913 (2007).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Howie, B.N., Donnelly, P. & Marchini, J. A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet. 5, e1000529 (2009).
Aulchenko, Y.S., Ripke, S., Isaacs, A. & van Duijn, C.M. GenABEL: an R library for genome-wide association analysis. Bioinformatics 23, 1294–1296 (2007).
Hickson, I. et al. Identification and characterization of a novel and specific inhibitor of the ataxia-telangiectasia mutated kinase ATM. Cancer Res. 64, 9152–9159 (2004).
Eaton, J.S., Lin, Z.P., Sartorelli, A.C., Bonawitz, N.D. & Shadel, G.S. Ataxia-telangiectasia mutated kinase regulates ribonucleotide reductase and mitochondrial homeostasis. J. Clin. Invest. 117, 2723–2734 (2007).
Crescenzi, E., Palumbo, G., de Boer, J. & Brady, H.J. Ataxia telangiectasia mutated and p21CIP1 modulate cell survival of drug-induced senescent tumor cells: implications for chemotherapy. Clin. Cancer Res. 14, 1877–1887 (2008).
Hardie, D.G., Salt, I.P. & Davies, S.P. Analysis of the role of the AMP-activated protein kinase in the response to cellular stress. Methods Mol. Biol. 99, 63–74 (2000).
Dale, S., Wilson, W.A., Edelman, A.M. & Hardie, D.G. Similar substrate recognition motifs for mammalian AMP-activated protein kinase, higher plant HMG-CoA reductase kinase-A, yeast SNF1, and mammalian calmodulin-dependent protein kinase I. FEBS Lett. 361, 191–195 (1995).
Acknowledgements
We are grateful to all the participants who took part in this study, to the general practitioners, to the Scottish School of Primary Care for their help in recruiting the participants, and to the whole team, which includes interviewers, computer and laboratory technicians, clerical workers, research scientists, volunteers, managers, receptionists and nurses. The Wellcome Trust provides support for the Wellcome Trust United Kingdom Type 2 Diabetes Case Control Collection (GoDARTS) and informatics support was provided by the Chief Scientist Office. The Wellcome Trust funds the Scottish Health Informatics Programme, provides core support for the Wellcome Trust Centre for Human Genetics in Oxford and funds the Wellcome Trust Case Control Consortium 2. This research was specifically funded by Diabetes UK (07/0003525), MRC (G0601261) and the Wellcome Trust (084726/Z/08/Z, 085475/Z/08/Z, 085475/B/08/Z). We also acknowledge support from the National Institute for Health Research award to Moorfields Eye Hospital National Health Service Foundation Trust and University College London Institute of Ophthalmology for a Specialist Biomedical Research Centre for Ophthalmology (to A.C.V.). P. Donnelly was supported in part by a Wolfson-Royal Society Merit Award. K.Z. holds a Henry Wellcome Post-Doctoral Fellowship. S.A.H. and D.G.H. were supported by the EXGENESIS consortium (LSHM-CT-2004-005272) funded by the European Commission.
Author information
Authors and Affiliations
Consortia
Contributions
A.D.M., C.N.A.P., E.R.P., A.S.F.D. H.C., A.T.H. and M.I.M. oversaw cohort collection for GoDARTS. R.R.H., M.I.M., R.L.C. and C.J.G. oversaw cohort collection for the UKPDS. The WTCCC2 DNA, genotyping, data quality control and informatics group (S.D., S.E., E.G., S.H. and C.L.) executed GWAS sample handling, genotyping and quality control. A.J.B., R. Tavendale, L.B., C.J.G. and F.C. performed the replication genotyping. The WTCCC2 Management Committee (P. Donnelly, J.M.B., E.B., M.A.B., J.P.C., A.C., N.C., P. Deloukas, A.D., J.J., H.S.M., C.G.M., R.P., A.R., S.J.S., N.J.S., R. Trembath, A.C.V., L.P. and N.W.W.) monitored the execution of the GWAS. K.Z., C.B., C.C.A.S., L.A.D., A.S. and C.F. performed statistical analyses. K.Z. and L.W.H. performed bioinformatic analyses. S.A.H., D.G.H., C. Schofield and C. Sutherland performed the functional studies. MAGIC investigators provided summary data on glycemic quantitative trait association. K.Z., C.B., C.C.A.S., C.N.P., A.D.M., C. Sutherland, D.G.H., R.R.H., M.I.M., P. Donnelly and E.R.P. contributed to writing the manuscript. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The author declare no competing financial interests.
Additional information
A full list of authors and affiliations is provided at the end of the paper. A full list of members is provided in the Supplementary Note.
A full list of members is provided in the Supplementary Note.
A full list of members is provided in the Supplementary Note.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–6, Supplementary Figures 1–3 and Supplementary Note. (PDF 598 kb)
Rights and permissions
About this article
Cite this article
The GoDARTS and UKPDS Diabetes Pharmacogenetics Study Group., The Wellcome Trust Case Control Consortium 2. Common variants near ATM are associated with glycemic response to metformin in type 2 diabetes. Nat Genet 43, 117–120 (2011). https://doi.org/10.1038/ng.735
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.735
This article is cited by
-
Mapping novel QTL and fine mapping of previously identified QTL associated with glucose tolerance using the collaborative cross mice
Mammalian Genome (2024)
-
Pharmacogenomics: current status and future perspectives
Nature Reviews Genetics (2023)
-
Präzisionstherapie bei Diabetes mellitus Typ 2
Die Diabetologie (2023)
-
Neoadjuvant docetaxel, epirubicin, and cyclophosphamide with or without metformin in breast cancer patients with metabolic abnormality: results from the randomized Phase II NeoMET trial
Breast Cancer Research and Treatment (2023)
-
Genome-wide association analysis identifies ancestry-specific genetic variation associated with acute response to metformin and glipizide in SUGAR-MGH
Diabetologia (2023)