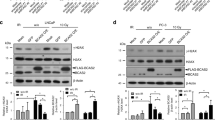
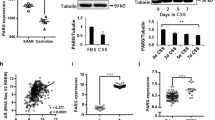
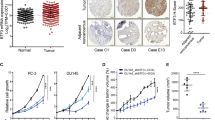
Abstract
DNA double-strand breaks (DSBs) can lead to the development of genomic rearrangements, which are hallmarks of cancer. Fusions between TMPRSS2, encoding the transmembrane serine protease isoform 2, and ERG, encoding the v-ets erythroblastosis virus E26 oncogene homolog, are among the most common oncogenic rearrangements observed in human cancer. We show that androgen signaling promotes co-recruitment of androgen receptor and topoisomerase II beta (TOP2B) to sites of TMPRSS2-ERG genomic breakpoints, triggering recombinogenic TOP2B-mediated DSBs. Furthermore, androgen stimulation resulted in de novo production of TMPRSS2-ERG fusion transcripts in a process that required TOP2B and components of the DSB repair machinery. Finally, unlike normal prostate epithelium, prostatic intraepithelial neoplasia cells showed strong coexpression of androgen receptor and TOP2B. These findings implicate androgen-induced TOP2B-mediated DSBs in generating TMPRSS2-ERG rearrangements.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
Change history
18 July 2010
In the version of this article initially published online, there were four sentences (in the Results on pages 2 and 3, in the legend to Figure 3 and in the Online Methods) containing minor errors. These errors have been corrected for the print, PDF and HTML versions of this article.
References
Rowley, J.D. Chromosomal translocations: revisited yet again. Blood 112, 2183–2189 (2008).
Mitelman, F., Johansson, B. & Mertens, F. The impact of translocations and gene fusions on cancer causation. Nat. Rev. Cancer 7, 233–245 (2007).
Morgan, W.F. et al. DNA double-strand breaks, chromosomal rearrangements, and genomic instability. Mutat. Res. 404, 125–128 (1998).
Richardson, C. & Jasin, M. Frequent chromosomal translocations induced by DNA double-strand breaks. Nature 405, 697–700 (2000).
Kumar-Sinha, C., Tomlins, S.A. & Chinnaiyan, A.M. Recurrent gene fusions in prostate cancer. Nat. Rev. Cancer 8, 497–511 (2008).
Tomlins, S.A. et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310, 644–648 (2005).
Jemal, A. et al. Cancer statistics, 2009. CA Cancer J. Clin. 59, 225–249 (2009).
Pedersen-Bjergaard, J., Andersen, M.K. & Johansson, B. Balanced chromosome aberrations in leukemias following chemotherapy with DNA-topoisomerase II inhibitors. J. Clin. Oncol. 16, 1897–1898 (1998).
Smith, M.A., McCaffrey, R.P. & Karp, J.E. The secondary leukemias: challenges and research directions. J. Natl. Cancer Inst. 88, 407–418 (1996).
Zhang, Y. & Rowley, J.D. Chromatin structural elements and chromosomal translocations in leukemia. DNA Repair (Amst.) 5, 1282–1297 (2006).
Strick, R., Strissel, P.L., Borgers, S., Smith, S.L. & Rowley, J.D. Dietary bioflavonoids induce cleavage in the MLL gene and may contribute to infant leukemia. Proc. Natl. Acad. Sci. USA 97, 4790–4795 (2000).
Ju, B.G. et al. A topoisomerase IIβ-mediated dsDNA break required for regulated transcription. Science 312, 1798–1802 (2006).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
Irizarry, R.A., Wang, C., Zhou, Y. & Speed, T.P. Gene Set Enrichment Analysis Made Simple (The Berkeley Electronic Press, 2009).
Nelson, W.G., Liu, L.F. & Coffey, D.S. Newly replicated DNA is associated with DNA topoisomerase II in cultured rat prostatic adenocarcinoma cells. Nature 322, 187–189 (1986).
Liu, W. et al. Copy number analysis indicates monoclonal origin of lethal metastatic prostate cancer. Nat. Med. 15, 559–565 (2009).
Demichelis, F. et al. Distinct genomic aberrations associated with ERG rearranged prostate cancer. Genes Chromosom. Cancer 48, 366–380 (2009).
Liu, W. et al. Multiple genomic alterations on 21q22 predict various TMPRSS2/ERG fusion transcripts in human prostate cancers. Genes Chromosom. Cancer 46, 972–980 (2007).
Wang, Q. et al. A hierarchical network of transcription factors governs androgen receptor-dependent prostate cancer growth. Mol. Cell 27, 380–392 (2007).
Dekker, J., Rippe, K., Dekker, M. & Kleckner, N. Capturing chromosome conformation. Science 295, 1306–1311 (2002).
Kinner, A., Wu, W., Staudt, C. & Iliakis, G. Gamma-H2AX in recognition and signaling of DNA double-strand breaks in the context of chromatin. Nucleic Acids Res. 36, 5678–5694 (2008).
Berkovich, E., Monnat, R.J. Jr. & Kastan, M.B. Roles of ATM and NBS1 in chromatin structure modulation and DNA double-strand break repair. Nat. Cell Biol. 9, 683–690 (2007).
Lavin, M.F. Ataxia-telangiectasia: from a rare disorder to a paradigm for cell signalling and cancer. Nat. Rev. Mol. Cell Biol. 9, 759–769 (2008).
Clark, J. et al. Complex patterns of ETS gene alteration arise during cancer development in the human prostate. Oncogene 27, 1993–2003 (2008).
Perner, S. et al. TMPRSS2-ERG fusion prostate cancer: an early molecular event associated with invasion. Am. J. Surg. Pathol. 31, 882–888 (2007).
Lin, C. et al. Nuclear receptor-induced chromosomal proximity and DNA breaks underlie specific translocations in cancer. Cell 139, 1069–1083 (2009).
Ripple, M.O., Henry, W.F., Rago, R.P. & Wilding, G. Prooxidant-antioxidant shift induced by androgen treatment of human prostate carcinoma cells. J. Natl. Cancer Inst. 89, 40–48 (1997).
De Marzo, A.M. et al. Inflammation in prostate carcinogenesis. Nat. Rev. Cancer 7, 256–269 (2007).
Li, T.K. et al. Activation of topoisomerase II-mediated excision of chromosomal DNA loops during oxidative stress. Genes Dev. 13, 1553–1560 (1999).
Azarova, A.M. et al. Roles of DNA topoisomerase II isozymes in chemotherapy and secondary malignancies. Proc. Natl. Acad. Sci. USA 104, 11014–11019 (2007).
Zhang, H., D'Arpa, P. & Liu, L.F. A model for tumor cell killing by topoisomerase poisons. Cancer Cells 2, 23–27 (1990).
Nelson, W.G., De Marzo, A.M. & Isaacs, W.B. Prostate cancer. N. Engl. J. Med. 349, 366–381 (2003).
Denmeade, S.R., Lin, X.S. & Isaacs, J.T. Role of programmed (apoptotic) cell death during the progression and therapy for prostate cancer. Prostate 28, 251–265 (1996).
Marker, P.C. Does prostate cancer co-opt the developmental program? Differentiation 76, 736–744 (2008).
Litvinov, I.V., De Marzo, A.M. & Isaacs, J.T. Is the Achilles' heel for prostate cancer therapy a gain of function in androgen receptor signaling? J. Clin. Endocrinol. Metab. 88, 2972–2982 (2003).
Hällström, T.M. & Laiho, M. Genetic changes and DNA damage responses in the prostate. Prostate 68, 902–918 (2008).
Carver, B.S. et al. Aberrant ERG expression cooperates with loss of PTEN to promote cancer progression in the prostate. Nat. Genet. 41, 619–624 (2009).
King, J.C. et al. Cooperativity of TMPRSS2-ERG with PI3-kinase pathway activation in prostate oncogenesis. Nat. Genet. 41, 524–526 (2009).
Klezovitch, O. et al. A causal role for ERG in neoplastic transformation of prostate epithelium. Proc. Natl. Acad. Sci. USA 105, 2105–2110 (2008).
Hermans, K.G. et al. Two unique novel prostate-specific and androgen-regulated fusion partners of ETV4 in prostate cancer. Cancer Res. 68, 3094–3098 (2008).
Mani, R.S. et al. Induced chromosomal proximity and gene fusions in prostate cancer. Science 326, 1230 (2009).
Aguilera, A. & Gomez-Gonzalez, B. Genome instability: a mechanistic view of its causes and consequences. Nat. Rev. Genet. 9, 204–217 (2008).
Boonyaratanakornkit, V. et al. High-mobility group chromatin proteins 1 and 2 functionally interact with steroid hormone receptors to enhance their DNA binding in vitro and transcriptional activity in mammalian cells. Mol. Cell. Biol. 18, 4471–4487 (1998).
Wang, Q., Carroll, J.S. & Brown, M. Spatial and temporal recruitment of androgen receptor and its coactivators involves chromosomal looping and polymerase tracking. Mol. Cell 19, 631–642 (2005).
Splinter, E., Grosveld, F. & de Laat, W. 3C technology: analyzing the spatial organization of genomic loci in vivo. Methods Enzymol. 375, 493–507 (2004).
Spitzner, J.R., Chung, I.K. & Muller, M.T. Eukaryotic topoisomerase II preferentially cleaves alternating purine-pyrimidine repeats. Nucleic Acids Res. 18, 1–11 (1990).
DeWeese, T.L. et al. Human papillomavirus E6 and E7 oncoproteins alter cell cycle progression but not radiosensitivity of carcinoma cells treated with low-dose-rate radiation. Int. J. Radiat. Oncol. Biol. Phys. 37, 145–154 (1997).
Gurel, B. et al. Nuclear MYC protein overexpression is an early alteration in human prostate carcinogenesis. Mod. Pathol. 21, 1156–1167 (2008).
Bolstad, B.M., Irizarry, R.A., Astrand, M. & Speed, T.P. A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19, 185–193 (2003).
Gentleman, R.C. et al. Bioconductor: open software development for computational biology and bioinformatics. Genome Biol. 5, R80 (2004).
Haffner, M.C. et al. Interaction and functional interference of glucocorticoid receptor and SOCS1. J. Biol. Chem. 283, 22089–22096 (2008).
Padilla, P.I. et al. Association of guanine nucleotide-exchange protein BIG1 in HepG2 cell nuclei with nucleolin, U3 snoRNA, and fibrillarin. Proc. Natl. Acad. Sci. USA 105, 3357–3361 (2008).
Acknowledgements
We thank D. Coffey for helpful comments and C. Heaphy, H. Zhang and L. Dasko-Vincent from the SKCCC Cell Imaging Core Facility for technical support. We also thank the Brady Urological Research Institute Prostate Specimen Repository for providing TMA sections. This work was supported by funding from the NIH/NCI, Department of Defense PCRP, Prostate Cancer Foundation, Maryland Cigarette Restitution Fund and the Patrick C. Walsh Prostate Cancer Research Fund/Dr. and Mrs. Peter S. Bing Scholarship.
Author information
Authors and Affiliations
Contributions
M.C.H. executed and analyzed all experiments and assisted in writing the manuscript. M.J.A. analyzed microarray data and assisted with statistical analysis of data. A.T., R.A., B.G., A.K.M., G.N. and A.M.D.M. assisted with execution and analysis of FISH, immunostaining and pathology experiments. D.M.E. assisted with execution of experiments. W.B.I., G.S.B., W.L. and J.X. contributed to analysis of microarray data in determination of prostate cancer TMPRSS2-ERG genomic breakpoints. W.G.N. assisted in experimental design and analysis and contributed to writing the manuscript. S.Y. conceived the study together with W.G.N., assisted in experimental design, execution and analysis and wrote the manuscript. All authors assisted in editing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–19 and Supplementary Tables 1–4. (PDF 7983 kb)
Rights and permissions
About this article
Cite this article
Haffner, M., Aryee, M., Toubaji, A. et al. Androgen-induced TOP2B-mediated double-strand breaks and prostate cancer gene rearrangements. Nat Genet 42, 668–675 (2010). https://doi.org/10.1038/ng.613
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.613
This article is cited by
-
Bipolar androgen therapy plus nivolumab for patients with metastatic castration-resistant prostate cancer: the COMBAT phase II trial
Nature Communications (2024)
-
Induction of double-strand breaks with the non-steroidal androgen receptor ligand flutamide in patients on androgen suppression: a study protocol for a randomized, double-blind prospective trial
Trials (2023)
-
The testosterone paradox of advanced prostate cancer: mechanistic insights and clinical implications
Nature Reviews Urology (2023)
-
Calcium signalling pathways in prostate cancer initiation and progression
Nature Reviews Urology (2023)
-
The potential role of the microbiota in prostate cancer pathogenesis and treatment
Nature Reviews Urology (2023)