Abstract
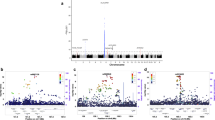
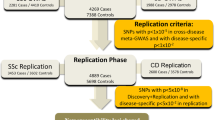
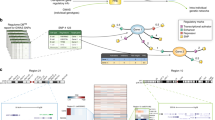
We report the results of a meta-analysis of genome-wide association scans for multiple sclerosis (MS) susceptibility that includes 2,624 subjects with MS and 7,220 control subjects. Replication in an independent set of 2,215 subjects with MS and 2,116 control subjects validates new MS susceptibility loci at TNFRSF1A (combined P = 1.59 × 10−11), IRF8 (P = 3.73 × 10−9) and CD6 (P = 3.79 × 10−9). TNFRSF1A harbors two independent susceptibility alleles: rs1800693 is a common variant with modest effect (odds ratio = 1.2), whereas rs4149584 is a nonsynonymous coding polymorphism of low frequency but with stronger effect (allele frequency = 0.02; odds ratio = 1.6). We also report that the susceptibility allele near IRF8, which encodes a transcription factor known to function in type I interferon signaling, is associated with higher mRNA expression of interferon-response pathway genes in subjects with MS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hauser, S.L. & Oksenberg, J.R. The neurobiology of multiple sclerosis: genes, inflammation, and neurodegeneration. Neuron 52, 61–76 (2006).
Barcellos, L.F. et al. Heterogeneity at the HLA-DRB1 locus and risk for multiple sclerosis. Hum. Mol. Genet. 15, 2813–2824 (2006).
Yeo, T.W. et al. A second major histocompatibility complex susceptibility locus for multiple sclerosis. Ann. Neurol. 61, 228–236 (2007).
Hafler, D.A. et al. Risk alleles for multiple sclerosis identified by a genomewide study. N. Engl. J. Med. 357, 851–862 (2007).
Rubio, J.P. et al. Replication of KIAA0350, IL2RA, RPL5 and CD58 as multiple sclerosis susceptibility genes in Australians. Genes Immun. 9, 624–630 (2008).
International Multiple Sclerosis Genetics Consortium. Refining genetic associations in multiple sclerosis. Lancet Neurol. 7, 567–569 (2008).
Ramagopalan, S.V., Anderson, C., Sadovnick, A.D. & Ebers, G.C. Genomewide study of multiple sclerosis. N. Engl. J. Med. 357, 2199–2200 (2007).
Baranzini, S.E. et al. Genome-wide association analysis of susceptibility and clinical phenotype in multiple sclerosis. Hum. Mol. Genet. 18, 767–778 (2009).
Frazer, K.A. et al. A second generation human haplotype map of over 3.1 million SNPs. Nature 449, 851–861 (2007).
Li, Y. & Abecasis, G.R. Rapid haplotype reconstruction and missing genotype inference. Am. J. Hum. Genet. S79, 2290 (2006).
de Bakker, P.I. et al. Practical aspects of imputation-driven meta-analysis of genome-wide association studies. Hum. Mol. Genet. 17, R122–R128 (2008).
Burton, P.R. et al. Association scan of 14,500 nonsynonymous SNPs in four diseases identifies autoimmunity variants. Nat. Genet. 39, 1329–1337 (2007).
Aulchenko, Y.S. et al. Genetic variation in the KIF1B locus influences susceptibility to multiple sclerosis. Nat. Genet. 40, 1402–1403 (2008).
de Bakker, P.I. et al. A high-resolution HLA and SNP haplotype map for disease association studies in the extended human MHC. Nat. Genet. 38, 1166–1172 (2006).
De Jager, P.L. et al. The role of the CD58 locus in multiple sclerosis. Proc. Natl. Acad. Sci. USA 106, 5264–5269 (2009).
Hoffmann, L.A. et al. TNFRSF1A R92Q mutation in association with a multiple sclerosis-like demyelinating syndrome. Neurology 70, 1155–1156 (2008).
Kumpfel, T. et al. Late-onset tumor necrosis factor receptor-associated periodic syndrome in multiple sclerosis patients carrying the TNFRSF1A R92Q mutation. Arthritis Rheum. 56, 2774–2783 (2007).
Aksentijevich, I. et al. The tumor-necrosis-factor receptor-associated periodic syndrome: new mutations in TNFRSF1A, ancestral origins, genotype-phenotype studies, and evidence for further genetic heterogeneity of periodic fevers. Am. J. Hum. Genet. 69, 301–314 (2001).
Wildemann, B. et al. The tumor-necrosis-factor-associated periodic syndrome, the brain, and tumor-necrosis-factor-alpha antagonists. Neurology 68, 1742–1744 (2007).
Jenne, D.E. et al. The low-penetrance R92Q mutation of the tumour necrosis factor superfamily 1A gene is neither a major risk factor for Wegener's granulomatosis nor multiple sclerosis. Ann. Rheum. Dis. 66, 1266–1267 (2007).
Pedchenko, T.V., Park, G.Y., Joo, M., Blackwell, T.S. & Christman, J.W. Inducible binding of PU.1 and interacting proteins to the Toll-like receptor 4 promoter during endotoxemia. Am. J. Physiol. Lung Cell. Mol. Physiol. 289, L429–L437 (2005).
Lee, C.H. et al. Regulation of the germinal center gene program by interferon (IFN) regulatory factor 8/IFN consensus sequence-binding protein. J. Exp. Med. 203, 63–72 (2006).
Hassan, N.J. et al. CD6 regulates T-cell responses through activation-dependent recruitment of the positive regulator SLP-76. Mol. Cell. Biol. 26, 6727–6738 (2006).
Castro, M.A. et al. Extracellular isoforms of CD6 generated by alternative splicing regulate targeting of CD6 to the immunological synapse. J. Immunol. 178, 4351–4361 (2007).
Hafler, D.A. et al. Immunologic responses of progressive multiple sclerosis patients treated with an anti-T-cell monoclonal antibody, anti-T12. Neurology 36, 777–784 (1986).
Sarrias, M.R. et al. CD6 binds to pathogen-associated molecular patterns and protects from LPS-induced septic shock. Proc. Natl. Acad. Sci. USA 104, 11724–11729 (2007).
Hunt, K.A. et al. Newly identified genetic risk variants for celiac disease related to the immune response. Nat. Genet. 40, 395–402 (2008).
Barrett, J.C. et al. Genome-wide association defines more than 30 distinct susceptibility loci for Crohn's disease. Nat. Genet. 40, 955–962 (2008).
Graham, R.R. et al. Genetic variants near TNFAIP3 on 6q23 are associated with systemic lupus erythematosus. Nat. Genet. 40, 1059–1061 (2008).
Raychaudhuri, S. et al. Common variants at CD40 and other loci confer risk of rheumatoid arthritis. Nat. Genet. 40, 1216–1223 (2008).
Musone, S.L. et al. Multiple polymorphisms in the TNFAIP3 region are independently associated with systemic lupus erythematosus. Nat. Genet. 40, 1062–1064 (2008).
Nair, R.P. et al. Genome-wide scan reveals association of psoriasis with IL-23 and NF-κB pathways. Nat. Genet. 41, 199–204 (2009).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
van Baarsen, L.G. et al. A subtype of multiple sclerosis defined by an activated immune defense program. Genes Immun. 7, 522–531 (2006).
Degre, M., Dahl, H. & Vandvik, B. Interferon in the serum and cerebrospinal fluid in patients with multiple sclerosis and other neurological disorders. Acta Neurol. Scand. 53, 152–160 (1976).
Stranger, B.E. et al. Genome-wide associations of gene expression variation in humans. PLoS Genet. 1, e78 (2005).
Greenberg, S.A. et al. Interferon-α/β-mediated innate immune mechanisms in dermatomyositis. Ann. Neurol. 57, 664–678 (2005).
van der Pouw Kraan, T.C. et al. Rheumatoid arthritis subtypes identified by genomic profiling of peripheral blood cells: assignment of a type I interferon signature in a subpopulation of patients. Ann. Rheum. Dis. 66, 1008–1014 (2007).
Baechler, E.C. et al. Interferon-inducible gene expression signature in peripheral blood cells of patients with severe lupus. Proc. Natl. Acad. Sci. USA 100, 2610–2615 (2003).
van Oosten, B.W. et al. Increased MRI activity and immune activation in two multiple sclerosis patients treated with the monoclonal anti-tumor necrosis factor antibody cA2. Neurology 47, 1531–1534 (1996).
The Lenercept Multiple Sclerosis Study Group and The University of British Columbia MS/MRI Analysis Group. TNF neutralization in MS: results of a randomized, placebo-controlled multicenter study. Neurology 53, 457–465 (1999).
Siddiqui, M.A. & Scott, L.J. Spotlight on infliximab in Crohn disease and rheumatoid arthritis. BioDrugs 20, 67–70 (2006).
De Jager, P.L. et al. Integrating risk factors: HLA-DRB1*1501 and Epstein-Barr virus in multiple sclerosis. Neurology 70, 1113–1118 (2008).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
McDonald, W.I. et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann. Neurol. 50, 121–127 (2001).
Price, A.L. et al. Principal components analysis corrects for stratification in genome-wide association studies. Nat. Genet. 38, 904–909 (2006).
Bienias, J.L., Beckett, L.A., Bennett, D.A., Wilson, R.S. & Evans, D.A. Design of the Chicago Health and Aging Project (CHAP). J. Alzheimers Dis. 5, 349–355 (2003).
Gauthier, S.A., Glanz, B.I., Mandel, M. & Weiner, H.L. A model for the comprehensive investigation of a chronic autoimmune disease: the multiple sclerosis CLIMB study. Autoimmun. Rev. 5, 532–536 (2006).
Miller, D., Barkhof, F., Montalban, X., Thompson, A. & Filippi, M. Clinically isolated syndromes suggestive of multiple sclerosis, part 2: non-conventional MRI, recovery processes, and management. Lancet Neurol. 4, 341–348 (2005).
Acknowledgements
P.L.D. is a Harry Weaver Neuroscience Scholar Award of the National MS Society (NMSS); he is also a William C. Fowler Scholar in Multiple Sclerosis Research and is supported by a National Institute of Neurological Disorders and Stroke (NINDS) K08 grant, NS46341. D.A.H. is a Jacob Javits Scholar of the US National Institutes of Health; he is also supported by NINDS P01 AI039671, R01 NS049477, R01NS046630, NMSS Collaborative MS Research Award and NMSS RG3567A. The International MS Genetics Consortium is supported by R01NS049477. L.P. is supported by an NMSS fellowship grant (FG1665-A-1). The genome-wide data on the BWH subjects and the RNA data on MS and CIS subjects from the CLIMB study were generated as part of a collaboration with Affymetrix, Inc. We thank the Myocardial Infarction Genetics Consortium (MIGen) study for the use of their genotype data as control data in our study. The MIGen study was funded by the US National Institutes of Health and National Heart, Lung, and Blood Institute's STAMPEED genomics research program and a grant from the National Center for Research Resources. We acknowledge use of genotype data from the British 1958 Birth Cohort DNA collection, funded by the Medical Research Council grant G0000934 and the Wellcome Trust grant 068545/Z/02. We thank R. Lincoln and R. Gomez for expert specimen management at UCSF as well as A. Santaniello for database management. We thank the Accelerated Cure Project for its work in collecting samples from subjects with MS and for making these samples available to MS investigators. We also thank the following clinicians for contributing to sample collection efforts: Accelerated Cure project, E. Frohman, B. Greenberg, P. Riskind, S. Sadiq, B. Thrower and T. Vollmer; Washington University, B.J. Parks and R.T. Naismith. Finally, we thank the Brigham & Women's Hospital PhenoGenetic Project for providing DNA samples from healthy subjects that were used in the replication effort of this study.
Author information
Authors and Affiliations
Consortia
Contributions
P.L.D., D.A.H., S.L.H., P.M.M. and J.R.O. designed the study. P.L.D. and J.R.O. wrote the manuscript. P.I.W.d.B., P.L.D., S.R., M.J.D., D.T., J.W., S.E.B. and X.J. performed analytical work. P.I.W.d.B., X.J. and M.J.D. developed the meta-analysis method while S.R. developed the subject matching algorithm. L.O. and P.L.D. performed the quality control analysis and quantitative trait analysis of the RNA from MS PBMC samples. C.A. generated and processed genotype data for analysis. P.L.D., N.T.A., L.P., R.B., R.A.G., P.M.M., Y.N., L.K., B.U., C.P., W.L.M., D.P.S., D.E., A.H.C., A.C., S.J.S., H.L.W., S.L.H., J.R.O. and D.A.H. contributed to DNA sample collection and genetic data. J.L.M., M.A.P.-V. and J.L.H. contributed to the interpretation of the results. All authors have read and contributed to the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–7 and Supplementary Figure 1 (PDF 2943 kb)
Rights and permissions
About this article
Cite this article
De Jager, P., Jia, X., Wang, J. et al. Meta-analysis of genome scans and replication identify CD6, IRF8 and TNFRSF1A as new multiple sclerosis susceptibility loci. Nat Genet 41, 776–782 (2009). https://doi.org/10.1038/ng.401
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.401
This article is cited by
-
rs10924104 in the expression enhancer motif of CD58 confers susceptibility to human autoimmune diseases
Human Genetics (2024)
-
Co-modulation of TNFR1 and TNFR2 in an animal model of multiple sclerosis
Journal of Neuroinflammation (2023)
-
Epstein–Barr virus and multiple sclerosis
Nature Reviews Microbiology (2023)
-
Immunoglobulin directly enhances differentiation of oligodendrocyte-precursor cells and remyelination
Scientific Reports (2023)
-
Pathogen-specific innate immune response patterns are distinctly affected by genetic diversity
Nature Communications (2023)