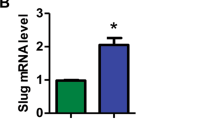
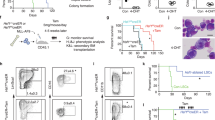
Abstract
Targeting of cancer stem cells is believed to be essential for curative therapy of cancers, but supporting evidence is limited. Few selective target genes in cancer stem cells have been identified. Here we identify the arachidonate 5-lipoxygenase (5-LO) gene (Alox5) as a critical regulator for leukemia stem cells (LSCs) in BCR-ABL–induced chronic myeloid leukemia (CML). In the absence of Alox5, BCR-ABL failed to induce CML in mice. This Alox5 deficiency caused impairment of the function of LSCs but not normal hematopoietic stem cells (HSCs) through affecting differentiation, cell division and survival of long-term LSCs (LT-LSCs), consequently causing a depletion of LSCs and a failure of CML development. Treatment of CML mice with a 5-LO inhibitor also impaired the function of LSCs similarly by affecting LT-LSCs, and prolonged survival. These results demonstrate that a specific target gene can be found in cancer stem cells and its inhibition can completely inhibit the function of these stem cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Al-Hajj, M., Wicha, M.S., Benito-Hernandez, A., Morrison, S.J. & Clarke, M.F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl. Acad. Sci. USA 100, 3983–3988 (2003).
Jordan, C.T., Guzman, M.L. & Noble, M. Cancer stem cells. N. Engl. J. Med. 355, 1253–1261 (2006).
Pardal, R., Clarke, M.F. & Morrison, S.J. Applying the principles of stem-cell biology to cancer. Nat. Rev. Cancer 3, 895–902 (2003).
Reya, T., Morrison, S.J., Clarke, M.F. & Weissman, I.L. Stem cells, cancer, and cancer stem cells. Nature 414, 105–111 (2001).
Rossi, D.J., Jamieson, C.H. & Weissman, I.L. Stems cells and the pathways to aging and cancer. Cell 132, 681–696 (2008).
Singh, S.K. et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 63, 5821–5828 (2003).
Wang, J.C. & Dick, J.E. Cancer stem cells: lessons from leukemia. Trends Cell Biol. 15, 494–501 (2005).
Molofsky, A.V., Pardal, R. & Morrison, S.J. Diverse mechanisms regulate stem cell self-renewal. Curr. Opin. Cell Biol. 16, 700–707 (2004).
Reya, T. et al. A role for Wnt signalling in self-renewal of haematopoietic stem cells. Nature 423, 409–414 (2003).
Taipale, J. & Beachy, P.A. The Hedgehog and Wnt signalling pathways in cancer. Nature 411, 349–354 (2001).
Zhao, C. et al. Loss of beta-catenin impairs the renewal of normal and CML stem cells in vivo. Cancer Cell 12, 528–541 (2007).
Lessard, J. & Sauvageau, G. Bmi-1 determines the proliferative capacity of normal and leukaemic stem cells. Nature 423, 255–260 (2003).
Park, I.K. et al. Bmi-1 is required for maintenance of adult self-renewing haematopoietic stem cells. Nature 423, 302–305 (2003).
Molofsky, A.V., He, S., Bydon, M., Morrison, S.J. & Pardal, R. Bmi-1 promotes neural stem cell self-renewal and neural development but not mouse growth and survival by repressing the p16Ink4a and p19Arf senescence pathways. Genes Dev. 19, 1432–1437 (2005).
Lowe, S.W. & Sherr, C.J. Tumor suppression by Ink4a-Arf: progress and puzzles. Curr. Opin. Genet. Dev. 13, 77–83 (2003).
Yilmaz, O.H. et al. Pten dependence distinguishes haematopoietic stem cells from leukaemia-initiating cells. Nature 441, 475–482 (2006).
Bonnet, D. & Dick, J.E. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat. Med. 3, 730–737 (1997).
Lapidot, T. et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 367, 645–648 (1994).
Bowie, M.B. et al. Identification of a new intrinsically timed developmental checkpoint that reprograms key hematopoietic stem cell properties. Proc. Natl. Acad. Sci. USA 104, 5878–5882 (2007).
Dierks, C. et al. Expansion of Bcr-Abl-positive leukemic stem cells is dependent on Hedgehog pathway activation. Cancer Cell 14, 238–249 (2008).
Huntly, B.J. et al. MOZ-TIF2, but not BCR-ABL, confers properties of leukemic stem cells to committed murine hematopoietic progenitors. Cancer Cell 6, 587–596 (2004).
Neering, S.J. et al. Leukemia stem cells in a genetically defined murine model of blast-crisis CML. Blood 110, 2578–2585 (2007).
Hu, Y. et al. Targeting multiple kinase pathways in leukemic prognitors and stem cells is essential for improved treatment of Ph+ leukemia in mice. Proc. Natl. Acad. Sci. USA 103, 16870–16875 (2006).
Graham, S.M. et al. Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. Blood 99, 319–325 (2002).
Marley, S.B., Deininger, M.W., Davidson, R.J., Goldman, J.M. & Gordon, M.Y. The tyrosine kinase inhibitor STI571, like interferon-alpha, preferentially reduces the capacity for amplification of granulocyte-macrophage progenitors from patients with chronic myeloid leukemia. Exp. Hematol. 28, 551–557 (2000).
Catalano, A., Rodilossi, S., Caprari, P., Coppola, V. & Procopio, A. 5-Lipoxygenase regulates senescence-like growth arrest by promoting ROS-dependent p53 activation. EMBO J. 24, 170–179 (2005).
Chen, X.S., Sheller, J.R., Johnson, E.N. & Funk, C.D. Role of leukotrienes revealed by targeted disruption of the 5-lipoxygenase gene. Nature 372, 179–182 (1994).
Radmark, O., Werz, O., Steinhilber, D. & Samuelsson, B. 5-Lipoxygenase: regulation of expression and enzyme activity. Trends Biochem. Sci. 32, 332–341 (2007).
Soberman, R.J. & Christmas, P. The organization and consequences of eicosanoid signaling. J. Clin. Invest. 111, 1107–1113 (2003).
Taylor, P.M. et al. Breast cancer cell-derived EMMPRIN stimulates fibroblast MMP2 release through a phospholipase A(2) and 5-lipoxygenase catalyzed pathway. Oncogene 21, 5765–5772 (2002).
Wymann, M.P. & Schneiter, R. Lipid signalling in disease. Nat. Rev. Mol. Cell Biol. 9, 162–176 (2008).
Yokomizo, T., Izumi, T. & Shimizu, T. Leukotriene B4: metabolism and signal transduction. Arch. Biochem. Biophys. 385, 231–241 (2001).
Zhao, L. et al. The 5-lipoxygenase pathway promotes pathogenesis of hyperlipidemia-dependent aortic aneurysm. Nat. Med. 10, 966–973 (2004).
Li, S., Ilaria, R.L. Jr, Million, R.P., Daley, G.Q. & Van Etten, R.A. The P190, P210, and p230 forms of the BCR/ABL oncogene induce a similar chronic myeloid leukemia-like syndrome in mice but have different lymphoid leukemogenic activity. J. Exp. Med. 189, 1399–1412 (1999).
Knapp, H.R. Reduced allergen-induced nasal congestion and leukotriene synthesis with an orally active 5-lipoxygenase inhibitor. N. Engl. J. Med. 323, 1745–1748 (1990).
Ferreira, R., Ohneda, K., Yamamoto, M. & Philipsen, S. GATA1 function, a paradigm for transcription factors in hematopoiesis. Mol. Cell. Biol. 25, 1215–1227 (2005).
Tsang, A.P. et al. FOG, a multitype zinc finger protein, acts as a cofactor for transcription factor GATA-1 in erythroid and megakaryocytic differentiation. Cell 90, 109–119 (1997).
Stenke, L., Lauren, L., Reizenstein, P. & Lindgren, J.A. Leukotriene production by fresh human bone marrow cells: evidence of altered lipoxygenase activity in chronic myelocytic leukemia. Exp. Hematol. 15, 203–207 (1987).
Tornhamre, S. et al. Inverse relationship between myeloid maturation and leukotriene C4 synthase expression in normal and leukemic myelopoiesis-consistent overexpression of the enzyme in myeloid cells from patients with chronic myeloid leukemia. Exp. Hematol. 31, 122–130 (2003).
Peng, C. et al. Inhibition of heat shock protein 90 prolongs survival of mice with BCR-ABL-T315I-induced leukemia and suppresses leukemic stem cells. Blood 110, 678–685 (2007).
Graham, S.M., Vass, J.K., Holyoake, T.L. & Graham, G.J. Transcriptional analysis of quiescent and proliferating CD34+ human hemopoietic cells from normal and chronic myeloid leukemia sources. Stem Cells 25, 3111–3120 (2007).
Radich, J.P. et al. Gene expression changes associated with progression and response in chronic myeloid leukemia. Proc. Natl. Acad. Sci. USA 103, 2794–2799 (2006).
Anderson, K.M. et al. Selective inhibitors of 5-lipoxygenase reduce CML blast cell proliferation and induce limited differentiation and apoptosis. Leuk. Res. 19, 789–801 (1995).
Hu, Y. et al. Requirement of Src kinases Lyn, Hck and Fgr for BCR-ABL1-induced B-lymphoblastic leukemia but not chronic myeloid leukemia. Nat. Genet. 36, 453–461 (2004).
Li, S., Couvillon, A.D., Brasher, B.B. & Van Etten, R.A. Tyrosine phosphorylation of Grb2 by Bcr/Abl and epidermal growth factor receptor: a novel regulatory mechanism for tyrosine kinase signaling. EMBO J. 20, 6793–6804 (2001).
Acknowledgements
This work was supported by the grants from the Leukemia & Lymphoma Society and the National Institutes of Health (R01-CA122142, R01-CA114199) to S.L. S.L. is a Scholar of the Leukemia & Lymphoma Society.
Author information
Authors and Affiliations
Contributions
Y.C. designed and performed experiments, and analyzed the data; Y.H. performed experiments and analyzed the data; H.Z. performed experiments; C.P. helped with the experiments; S.L. designed and performed experiments, analyzed the data and wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Table 1 (PDF 555 kb)
Rights and permissions
About this article
Cite this article
Chen, Y., Hu, Y., Zhang, H. et al. Loss of the Alox5 gene impairs leukemia stem cells and prevents chronic myeloid leukemia. Nat Genet 41, 783–792 (2009). https://doi.org/10.1038/ng.389
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.389
This article is cited by
-
Knock-out of 5-lipoxygenase in overexpressing tumor cells—consequences on gene expression and cellular function
Cancer Gene Therapy (2023)
-
COX-2/PGE2 upregulation contributes to the chromosome 17p-deleted lymphoma
Oncogenesis (2023)
-
Mitochondrial metabolism as a potential therapeutic target in myeloid leukaemia
Leukemia (2022)
-
Leukotrienes promote stem cell self-renewal and chemoresistance in acute myeloid leukemia
Leukemia (2022)
-
A study of 5-lipoxygenase inhibitors invoking DFT-based descriptor nucleophilicity index
Monatshefte für Chemie - Chemical Monthly (2022)