Abstract
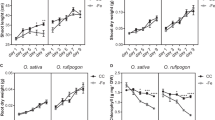
Asian cultivated rice (Oryza sativa L.) consists of two main subspecies, indica and japonica. Indica has higher nitrate-absorption activity than japonica, but the molecular mechanisms underlying that activity remain elusive. Here we show that variation in a nitrate-transporter gene, NRT1.1B (OsNPF6.5), may contribute to this divergence in nitrate use. Phylogenetic analysis revealed that NRT1.1B diverges between indica and japonica. NRT1.1B-indica variation was associated with enhanced nitrate uptake and root-to-shoot transport and upregulated expression of nitrate-responsive genes. The selection signature of NRT1.1B-indica suggests that nitrate-use divergence occurred during rice domestication. Notably, field tests with near-isogenic and transgenic lines confirmed that the japonica variety carrying the NRT1.1B-indica allele had significantly improved grain yield and nitrogen-use efficiency (NUE) compared to the variety without that allele. Our results show that variation in NRT1.1B largely explains nitrate-use divergence between indica and japonica and that NRT1.1B-indica can potentially improve the NUE of japonica.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Oka, H.I. Origin of Cultivated Rice (Japan Scientific Societies Press, 1983).
Koutroubas, S.D. & Ntanos, D.A. Genotypic differences for grain yield and nitrogen utilization in Indica and Japonica rice under Mediterranean conditions. Field Crops Res. 83, 251–260 (2003).
Kronzucker, H.J., Glass, A.D.M., Siddiqi, M.Y. & Kirk, G.J.D. Comparative kinetic analysis of ammonium and nitrate acquisition by tropical lowland rice: implications for rice cultivation and yield potential. New Phytol. 145, 471–476 (2000).
Kirk, G.J. & Kronzucker, H.J. The potential for nitrification and nitrate uptake in the rhizosphere of wetland plants: a modelling study. Ann. Bot. 96, 639–646 (2005).
Ta, T.C. & Ohira, K. Effects of various environmental and medium conditions on the response of indica and japonica rice plants to ammonium and nitrate nitrogen. Soil Sci. Plant Nutr. 27, 347–355 (1981).
Tsay, Y.F., Schroeder, J.I., Feldmann, K.A. & Crawford, N.M. The herbicide sensitivity gene Chl1 of Arabidopsis encodes a nitrate-inducible nitrate transporter. Cell 72, 705–713 (1993).
Scholten, H.J. & Feenstra, W.J. Expression of the mutant character of chlorate-resistant mutants of Arabidopsis thaliana in cell-culture. J. Plant Physiol. 123, 45–54 (1986).
Oostindiër-Braaksma, F.J. & Feenstra, W.J. Isolation and characterization of chlorate-resistant mutants of Arabidopsis thaliana. Mutat. Res. 19, 175–185 (1973).
Wang, X.M., Scholl, R.L. & Feldmann, K.A. Characterization of a chlorate-hypersensitive, high nitrate reductase Arabidopsis thaliana mutant. Theor. Appl. Genet. 72, 328–336 (1986).
Teng, S. et al. QTLs and candidate genes for chlorate resistance in rice (Oryza sativa L.). Euphytica 152, 141–148 (2006).
Plett, D. et al. Dichotomy in the NRT gene families of dicots and grass species. PLoS One 5, e15289 (2010).
Monden, I., Olsowski, A., Krause, G. & Keller, K. The large cytoplasmic loop of the glucose transporter GLUT1 is an essential structural element for function. Biol. Chem. 382, 1551–1558 (2001).
Mandal, A. et al. A key structural domain of the Candida albicans Mdr1 protein. Biochem. J. 445, 313–322 (2012).
Zhao, Y. et al. Crystal structure of the E. coli peptide transporter YbgH. Structure 22, 1152–1160 (2014).
Liu, K.H., Huang, C.Y. & Tsay, Y.F. CHL1 is a dual-affinity nitrate transporter of Arabidopsis involved in multiple phases of nitrate uptake. Plant Cell 11, 865–874 (1999).
Ho, C.H., Lin, S.H., Hu, H.C. & Tsay, Y.F. CHL1 functions as a nitrate sensor in plants. Cell 138, 1184–1194 (2009).
Ho, C.H. & Tsay, Y.F. Nitrate, ammonium, and potassium sensing and signaling. Curr. Opin. Plant Biol. 13, 604–610 (2010).
Gojon, A., Krouk, G., Perrine-Walker, F. & Laugier, E. Nitrate transceptor(s) in plants. J. Exp. Bot. 62, 2299–2308 (2011).
Biswas, S. & Akey, J.M. Genomic insights into positive selection. Trends Genet. 22, 437–446 (2006).
Caicedo, A.L. et al. Genome-wide patterns of nucleotide polymorphism in domesticated rice. PLoS Genet. 3, 1745–1756 (2007).
Gao, L.Z. & Innan, H. Nonindependent domestication of the two rice subspecies, Oryza sativa ssp. indica and ssp. japonica, demonstrated by multilocus microsatellites. Genetics 179, 965–976 (2008).
Huang, X. et al. A map of rice genome variation reveals the origin of cultivated rice. Nature 490, 497–501 (2012).
Moll, R.H., Kamprath, E.J. & Jackson, W.A. Analysis and interpretation of factors which contribute to efficiency of nitrogen-utilization. Agron. J. 74, 562–564 (1982).
Moll, R.H., Kamprath, E.J. & Jackson, W.A. Development of nitrogen efficient prolific hybrids of maize. Crop Sci. 27, 181–186 (1987).
Bart, R., Chern, M., Park, C.J., Bartley, L. & Ronald, P.C. A novel system for gene silencing using siRNAs in rice leaf and stem-derived protoplasts. Plant Methods 2, 13 (2006).
Almagro, A., Lin, S.H. & Tsay, Y.F. Characterization of the Arabidopsis nitrate transporter NRT1.6 reveals a role of nitrate in early embryo development. Plant Cell 20, 3289–3299 (2008).
Li, C. et al. A rice plastidial nucleotide sugar epimerase is involved in galactolipid biosynthesis and improves photosynthetic efficiency. PLoS Genet. 7, e1002196 (2011).
Felsenstein, J. PHYLIP—phylogeny inference package (version 3.2). Cladistics 5, 164–166 (1989).
Zhang, H., Gao, S., Lercher, M.J., Hu, S. & Chen, W.H. EvolView, an online tool for visualizing, annotating and managing phylogenetic trees. Nucleic Acids Res. 40, W569–W572 (2012).
Kawahara, Y. et al. Improvement of the Oryza sativa Nipponbare reference genome using next generation sequence and optical map data. Rice (NY) 6, 4 (2013).
Edgar, R.C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Tamura, K., Stecher, G., Peterson, D., Filipski, A. & Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol. Biol. Evol. 30, 2725–2729 (2013).
Jensen, J.D., Thornton, K.R., Bustamante, C.D. & Aquadro, C.E. On the utility of linkage disequilibrium as a statistic for identifying targets of positive selection in nonequilibrium populations. Genetics 176, 2371–2379 (2007).
Alachiotis, N., Stamatakis, A. & Pavlidis, P. OmegaPlus: a scalable tool for rapid detection of selective sweeps in whole-genome datasets. Bioinformatics 28, 2274–2275 (2012).
Acknowledgements
We thank H. Xue (Shanghai Institute of Plant Physiology and Ecology, Chinese Academy of Sciences, Shanghai, China) for providing the nrt1.1b mutant and M. Schläppi (Marquette University, Milwaukee, Wisconsin, USA) and X. Wang (China Agricultural University, Beijing, China) for language editing. This work was supported by grants from the Ministry of Science and Technology of China (2014AA10A602-5, 2015CB755702) and the Chinese Academy of Sciences (XDA08010400).
Author information
Authors and Affiliations
Contributions
B.H. performed most of the experiments. J.T. and W.W. constructed the NIL. W.W., Z.Z., Z.P., Q.D. and Y.L. performed the field test. S.O. and H.W. performed the population genetic analysis. H.L. assayed 15N accumulation in rice cultivars. R.C. carried out the in situ hybridization. X.C. and L. Li performed the Xenopus oocyte injection. Y.W., C.L., L. Liu, K.D. and C.X. performed the chlorate-sensitivity assay. C.C., B.H. and L.Z. designed the experiments. B.H., C.C. and S.O. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14, Supplementary Tables 1–3 and Supplementary Note (PDF 2815 kb)
Rights and permissions
About this article
Cite this article
Hu, B., Wang, W., Ou, S. et al. Variation in NRT1.1B contributes to nitrate-use divergence between rice subspecies. Nat Genet 47, 834–838 (2015). https://doi.org/10.1038/ng.3337
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3337
This article is cited by
-
Natural variation in BnaA9.NF-YA7 contributes to drought tolerance in Brassica napus L
Nature Communications (2024)
-
A microscopic scenario on recovery mechanisms under waterlogging and submergence stress in rice
Planta (2024)
-
Overexpression of rice OsNRT1.1A/OsNPF6.3 enhanced the nitrogen use efficiency of wheat under low nitrogen conditions
Planta (2024)
-
OsCBL1 modulates rice nitrogen use efficiency via negative regulation of OsNRT2.2 by OsCCA1
BMC Plant Biology (2023)
-
Plastid-localized amino acid metabolism coordinates rice ammonium tolerance and nitrogen use efficiency
Nature Plants (2023)