Abstract
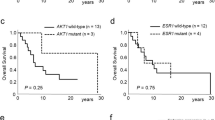
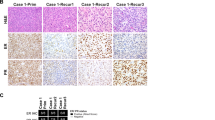
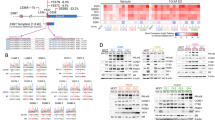
Breast cancer is the most prevalent cancer in women, and over two-thirds of cases express estrogen receptor-α (ER-α, encoded by ESR1). Through a prospective clinical sequencing program for advanced cancers, we enrolled 11 patients with ER-positive metastatic breast cancer. Whole-exome and transcriptome analysis showed that six cases harbored mutations of ESR1 affecting its ligand-binding domain (LBD), all of whom had been treated with anti-estrogens and estrogen deprivation therapies. A survey of The Cancer Genome Atlas (TCGA) identified four endometrial cancers with similar mutations of ESR1. The five new LBD-localized ESR1 mutations identified here (encoding p.Leu536Gln, p.Tyr537Ser, p.Tyr537Cys, p.Tyr537Asn and p.Asp538Gly) were shown to result in constitutive activity and continued responsiveness to anti-estrogen therapies in vitro. Taken together, these studies suggest that activating mutations in ESR1 are a key mechanism in acquired endocrine resistance in breast cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chin, L., Andersen, J.N. & Futreal, P.A. Cancer genomics: from discovery science to personalized medicine. Nat. Med. 17, 297–303 (2011).
Meyerson, M., Gabriel, S. & Getz, G. Advances in understanding cancer genomes through second-generation sequencing. Nat. Rev. Genet. 11, 685–696 (2010).
Roychowdhury, S. et al. Personalized oncology through integrative high-throughput sequencing: a pilot study. Sci. Transl. Med. 3, 111ra121 (2011).
Welch, J.S. et al. Use of whole-genome sequencing to diagnose a cryptic fusion oncogene. J. Am. Med. Assoc. 305, 1577–1584 (2011).
Gorre, M.E. et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 293, 876–880 (2001).
Korpal, M. et al. An F876L mutation in androgen receptor confers genetic and phenotypic resistance to MDV3100 (enzalutamide). Cancer Discov. 3, 1030–1043 (2013).
Joseph, J.D. et al. A clinically relevant androgen receptor mutation confers resistance to 2nd generation anti-androgens enzalutamide and ARN-509. Cancer Discov. 3, 1020–1029 (2013).
Ariazi, E.A., Ariazi, J.L., Cordera, F. & Jordan, V.C. Estrogen receptors as therapeutic targets in breast cancer. Curr. Top. Med. Chem. 6, 181–202 (2006).
Riggins, R.B., Schrecengost, R.S., Guerrero, M.S. & Bouton, A.H. Pathways to tamoxifen resistance. Cancer Lett. 256, 1–24 (2007).
Lønning, P.E. & Eikesdal, H.P. Aromatase inhibition 2013: clinical state of the art and questions that remain to be solved. Endocr. Relat. Cancer 20, R183–R201 (2013).
Ingle, J.N. et al. Fulvestrant in women with advanced breast cancer after progression on prior aromatase inhibitor therapy: North Central Cancer Treatment Group Trial N0032. J. Clin. Oncol. 24, 1052–1056 (2006).
Osborne, C.K. & Schiff, R. Mechanisms of endocrine resistance in breast cancer. Annu. Rev. Med. 62, 233–247 (2011).
Wu, Y.M. et al. Identification of targetable FGFR gene fusions in diverse cancers. Cancer Discov. 3, 636–647 (2013).
Nik-Zainal, S. et al. Mutational processes molding the genomes of 21 breast cancers. Cell 149, 979–993 (2012).
Feil, R., Wagner, J., Metzger, D. & Chambon, P. Regulation of Cre recombinase activity by mutated estrogen receptor ligand-binding domains. Biochem. Biophys. Res. Commun. 237, 752–757 (1997).
TCGA. Comprehensive molecular portraits of human breast tumours. Nature 490, 61–70 (2012).
Ellis, M.J. et al. Whole-genome analysis informs breast cancer response to aromatase inhibition. Nature 486, 353–360 (2012).
Huang, H.J., Norris, J.D. & McDonnell, D.P. Identification of a negative regulatory surface within estrogen receptor α provides evidence in support of a role for corepressors in regulating cellular responses to agonists and antagonists. Mol. Endocrinol. 16, 1778–1792 (2002).
Shiau, A.K. et al. The structural basis of estrogen receptor/coactivator recognition and the antagonism of this interaction by tamoxifen. Cell 95, 927–937 (1998).
Skafar, D.F. Formation of a powerful capping motif corresponding to start of “helix 12” in agonist-bound estrogen receptor-α contributes to increased constitutive activity of the protein. Cell Biochem. Biophys. 33, 53–62 (2000).
Carlson, K.E., Choi, I., Gee, A., Katzenellenbogen, B.S. & Katzenellenbogen, J.A. Altered ligand binding properties and enhanced stability of a constitutively active estrogen receptor: evidence that an open pocket conformation is required for ligand interaction. Biochemistry 36, 14897–14905 (1997).
Weis, K.E., Ekena, K., Thomas, J.A., Lazennec, G. & Katzenellenbogen, B.S. Constitutively active human estrogen receptors containing amino acid substitutions for tyrosine 537 in the receptor protein. Mol. Endocrinol. 10, 1388–1398 (1996).
Pearce, S.T., Liu, H. & Jordan, V.C. Modulation of estrogen receptor α function and stability by tamoxifen and a critical amino acid (Asp-538) in helix 12. J. Biol. Chem. 278, 7630–7638 (2003).
Zhao, C. et al. Mutation of Leu-536 in human estrogen receptor-α alters the coupling between ligand binding, transcription activation, and receptor conformation. J. Biol. Chem. 278, 27278–27286 (2003).
Ellis, M.J. et al. Lower-dose vs high-dose oral estradiol therapy of hormone receptor–positive, aromatase inhibitor–resistant advanced breast cancer: a phase 2 randomized study. J. Am. Med. Assoc. 302, 774–780 (2009).
Swaby, R.F. & Jordan, V.C. Low-dose estrogen therapy to reverse acquired antihormonal resistance in the treatment of breast cancer. Clin. Breast Cancer 8, 124–133 (2008).
Sokolosky, M.L. et al. Involvement of Akt-1 and mTOR in sensitivity of breast cancer to targeted therapy. Oncotarget 2, 538–550 (2011).
Kan, Z. et al. Diverse somatic mutation patterns and pathway alterations in human cancers. Nature 466, 869–873 (2010).
Shrestha, Y. et al. PAK1 is a breast cancer oncogene that coordinately activates MAPK and MET signaling. Oncogene 31, 3397–3408 (2012).
Barone, I., Brusco, L. & Fuqua, S.A. Estrogen receptor mutations and changes in downstream gene expression and signaling. Clin. Cancer Res. 16, 2702–2708 (2010).
Kandoth, C. et al. Integrated genomic characterization of endometrial carcinoma. Nature 497, 67–73 (2013).
Fisher, B. et al. Endometrial cancer in tamoxifen-treated breast cancer patients: findings from the National Surgical Adjuvant Breast and Bowel Project (NSABP) B-14. J. Natl. Cancer Inst. 86, 527–537 (1994).
Dawson, S.J. et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N. Engl. J. Med. 368, 1199–1209 (2013).
Diehl, F. et al. Circulating mutant DNA to assess tumor dynamics. Nat. Med. 14, 985–990 (2008).
Kim, D. & Salzberg, S.L. TopHat-Fusion: an algorithm for discovery of novel fusion transcripts. Genome Biol. 12, R72 (2011).
Trapnell, C. et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 7, 562–578 (2012).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Koboldt, D.C. et al. VarScan 2: somatic mutation and copy number alteration discovery in cancer by exome sequencing. Genome Res. 22, 568–576 (2012).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010).
Lonigro, R.J. et al. Detection of somatic copy number alterations in cancer using targeted exome capture sequencing. Neoplasia 13, 1019–1025 (2011).
Acknowledgements
The authors thank D. Miller, T. Barrette and D. Gibbs for hardware and database management, K. Giles for assistance with manuscript preparation, physicians M. Wicha, L. Pierce, D. Smith, K. Levin and F. Feng for referring patients, and C. Betts and J. Athanikar for assistance with tumor boards. We also thank the larger MI-ONCOSEQ team, including clinical research coordinator E. Williams, pathologist R. Mehra, genetic counselors J. Everett, S. Gustafson and V. Raymond, and radiologists E. Higgins, E. Caoili and R. Dunnick. This project is supported in part by the Prostate Cancer Foundation with funding for our sequencing infrastructure, the National Cancer Institute Early Detection Research Network (U01 CA111275), National Human Genome Research Institute Clinical Sequencing Exploratory Research (CSER) Consortium (1UM1HG006508), US Department of Defense contract W81XWH-12-1-0080 and a Department of Defense Era of Hope Scholar Award. A.M.C. is also supported by the Alfred A. Taubman Institute, the American Cancer Society, the Howard Hughes Medical Institute and a Doris Duke Charitable Foundation Clinical Scientist Award.
Author information
Authors and Affiliations
Contributions
D.R.R., Y.-M.W. and A.M.C. conceived the experiments. D.R.R., Y.-M.W., X.C., R.W., F.S. and Y.N. performed exome and transcriptome sequencing. P.V., R.J.L., S.K.-S. and D.R.R. carried out bioinformatics analysis of high-throughput sequencing data for somatic mutation, copy number and tumor content determination and performed gene expression and gene fusion analysis. D.R.R., Y.-M.W. and F.S. generated ESR1 constructs and carried out in vitro experiments. L.H. coordinated patients for clinical research. J.S. and A.G. collected and processed clinical tissue samples for next-generation sequencing. L.P.K. and S.A.T. provided pathology review. J.M.R. provided experimental analysis. C.H.V.P., D.F.H., R.C. and A.F.S. enrolled patients and provided clinical data and consultation at tumor boards. D.R.R., X.C., Y.-M.W., P.V., R.J.L., S.K.-S., S.Y.K., J.S.R., S.R., M.T., K.J.P. and A.M.C. developed the integrated clinical sequencing protocol. D.R.R., Y.-M.W. and A.M.C. prepared the manuscript, which was reviewed by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Tables 1–5 (PDF 12863 kb)
Rights and permissions
About this article
Cite this article
Robinson, D., Wu, YM., Vats, P. et al. Activating ESR1 mutations in hormone-resistant metastatic breast cancer. Nat Genet 45, 1446–1451 (2013). https://doi.org/10.1038/ng.2823
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2823
This article is cited by
-
The value of oral selective estrogen receptor degraders in patients with HR-positive, HER2-negative advanced breast cancer after progression on ≥ 1 line of endocrine therapy: systematic review and meta-analysis
BMC Cancer (2024)
-
Cyclin-dependent kinase 7 (CDK7) inhibitors as a novel therapeutic strategy for different molecular types of breast cancer
British Journal of Cancer (2024)
-
Matched analysis of circulating selenium with the breast cancer selenotranscriptome: a multicentre prospective study
Journal of Translational Medicine (2023)
-
Proteogenomics decodes the evolution of human ipsilateral breast cancer
Communications Biology (2023)
-
Targeting sex steroid biosynthesis for breast and prostate cancer therapy
Nature Reviews Cancer (2023)