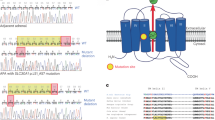
Abstract
At least 5% of individuals with hypertension have adrenal aldosterone-producing adenomas (APAs). Gain-of-function mutations in KCNJ5 and apparent loss-of-function mutations in ATP1A1 and ATP2A3 were reported to occur in APAs1,2. We find that KCNJ5 mutations are common in APAs resembling cortisol-secreting cells of the adrenal zona fasciculata but are absent in a subset of APAs resembling the aldosterone-secreting cells of the adrenal zona glomerulosa3. We performed exome sequencing of ten zona glomerulosa–like APAs and identified nine with somatic mutations in either ATP1A1, encoding the Na+/K+ ATPase α1 subunit, or CACNA1D, encoding Cav1.3. The ATP1A1 mutations all caused inward leak currents under physiological conditions, and the CACNA1D mutations induced a shift of voltage-dependent gating to more negative voltages, suppressed inactivation or increased currents. Many APAs with these mutations were <1 cm in diameter and had been overlooked on conventional adrenal imaging. Recognition of the distinct genotype and phenotype for this subset of APAs could facilitate diagnosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
Primary accessions
Gene Expression Omnibus
Referenced accessions
Ensembl
NCBI Reference Sequence
Protein Data Bank
References
Choi, M. et al. K+ channel mutations in adrenal aldosterone-producing adenomas and hereditary hypertension. Science 331, 768–772 (2011).
Beuschlein, F. et al. Somatic mutations in ATP1A1 and ATP2B3 lead to aldosterone-producing adenomas and secondary hypertension. Nat. Genet. 45, 440–444 (2013).
Azizan, E.A. et al. Microarray, qPCR and KCNJ5 sequencing of aldosterone-producing adenomas reveal differences in genotype and phenotype between zona glomerulosa– and zona fasciculata–like tumors. J. Clin. Endocrinol. Metab. 97, E819–E829 (2012).
Rossi, G.P. et al. A prospective study of the prevalence of primary aldosteronism in 1,125 hypertensive patients. J. Am. Coll. Cardiol. 48, 2293–2300 (2006).
Rossi, G.P. A comprehensive review of the clinical aspects of primary aldosteronism. Nat. Rev. Endocrinol. 7, 485–495 (2011).
Boulkroun, S. et al. Prevalence, clinical, and molecular correlates of KCNJ5 mutations in primary aldosteronism. Hypertension 59, 592–598 (2012).
Funder, J.W. The genetics of primary aldosteronism: chapter two. Hypertension 59, 537–538 (2012).
Mazzocchi, G. et al. Ghrelin enhances the growth of cultured human adrenal zona glomerulosa cells by exerting MAPK-mediated proliferogenic and antiapoptotic effects. Peptides 25, 1269–1277 (2004).
Shigematsu, K. et al. Primary aldosteronism with aldosterone-producing adenoma consisting of pure zona glomerulosa–type cells in a pregnant woman. Endocr. Pathol. 20, 66–72 (2009).
Wolkersdörfer, G.W. et al. Differential regulation of apoptosis in the normal human adrenal gland. J. Clin. Endocrinol. Metab. 81, 4129–4136 (1996).
Morth, J.P. et al. Crystal structure of the sodium-potassium pump. Nature 450, 1043–1049 (2007).
Winther, A.M. et al. The sarcolipin-bound calcium pump stabilizes calcium sites exposed to the cytoplasm. Nature 495, 265–269 (2013).
Toyoshima, C. et al. Crystal structures of the calcium pump and sarcolipin in the Mg2+-bound E1 state. Nature 495, 260–264 (2013).
Li, C., Capendeguy, O., Geering, K. & Horisberger, J.D. A third Na+-binding site in the sodium pump. Proc. Natl. Acad. Sci. USA 102, 12706–12711 (2005).
Poulsen, H. et al. Neurological disease mutations compromise a C-terminal ion pathway in the Na+/K+-ATPase. Nature 467, 99–102 (2010).
Vasilyev, A., Khater, K. & Rakowski, R.F. Effect of extracellular pH on presteady-state and steady-state current mediated by the Na+/K+ pump. J. Membr. Biol. 198, 65–76 (2004).
Efthymiadis, A., Rettinger, J. & Schwarz, W. Inward-directed current generated by the Na+,K+ pump in Na+- and K+-free medium. Cell Biol. Int. 17, 1107–1116 (1993).
Braley, L.M. & Williams, G.H. The effects of ouabain on steroid production by rat adrenal cells stimulated by angiotensin II, α1–24 adrenocorticotropin, and potassium. Endocrinology 103, 1997–2005 (1978).
Yingst, D.R., Davis, J., Krenz, S. & Schiebinger, R.J. Insights into the mechanism by which inhibition of Na,K-ATPase stimulates aldosterone production. Metabolism 48, 1167–1171 (1999).
Cushman, P. Jr. Inhibition of aldosterone secretion by ouabain in dog adrenal cortical tissue. Endocrinology 84, 808–813 (1969).
Kau, M.M., Kan, S.F., Wang, J.R. & Wang, P.S. Inhibitory effects of digoxin and ouabain on aldosterone synthesis in human adrenocortical NCI-H295 cells. J. Cell Physiol. 205, 393–401 (2005).
Hemara-Wahanui, A. et al. A CACNA1F mutation identified in an X-linked retinal disorder shifts the voltage dependence of Cav1.4 channel activation. Proc. Natl. Acad. Sci. USA 102, 7553–7558 (2005).
Splawski, I. et al. Severe arrhythmia disorder caused by cardiac L-type calcium channel mutations. Proc. Natl. Acad. Sci. USA 102, 8089–8096 (2005).
Hoda, J.C., Zaghetto, F., Koschak, A. & Striessnig, J. Congenital stationary night blindness type 2 mutations S229P, G369D, L1068P, and W1440X alter channel gating or functional expression of Cav1.4 L-type Ca2+ channels. J. Neurosci. 25, 252–259 (2005).
Brown, M.J. & Hopper, R.V. Calcium-channel blockade can mask the diagnosis of Conn's syndrome. Postgrad. Med. J. 75, 235–236 (1999).
Lee, G. et al. Homeostatic responses in the adrenal cortex to the absence of aldosterone in mice. Endocrinology 146, 2650–2656 (2005).
Nishimoto, K. et al. Adrenocortical zonation in humans under normal and pathological conditions. J. Clin. Endocrinol. Metab. 95, 2296–2305 (2010).
Burton, T.J. et al. Evaluation of the sensitivity and specificity of 11C-metomidate positron emission tomography (PET)-CT for lateralizing aldosterone secretion by Conn's adenomas. J. Clin. Endocrinol. Metab. 97, 100–109 (2012).
Koschak, A. et al. α1D (Cav1.3) subunits can form L-type Ca2+ channels activating at negative voltages. J. Biol. Chem. 276, 22100–22106 (2001).
Singh, A. et al. C-terminal modulator controls Ca2+-dependent gating of Cav1.4 L-type Ca2+ channels. Nat. Neurosci. 9, 1108–1116 (2006).
Baig, S.M. et al. Loss of Cav1.3 (CACNA1D) function in a human channelopathy with bradycardia and congenital deafness. Nat. Neurosci. 14, 77–84 (2011).
Acknowledgements
Cambridge , UK. We are grateful to M. Gurnell for discussion and care of many of the patients, to N. Jamieson for all the laparoscopic adrenalectomies and to A. Marker for pathological diagnosis. We thank Dr. Yasmin for providing peripheral DNA samples from healthy, normotensive subjects. We thank R. Kuc and the Human Research Tissue Bank of Addenbrooke's Hospital, which is supported by the NIHR Cambridge BRC, for help with storage of fresh adrenal tissue for the Cambridge cohort; we particularly acknowledge B. Haynes, D. Walters, K. Brown, M. Elazoui, C. Karpinskyj, M. Bromwich and K. Payne. The work was funded by the British Heart Foundation (PG/07/085/23349), the Wellcome Trust (085686/Z/08/A), the NIHR Cambridge Biomedical Research Centre (Cardiovascular) and an NIHR Senior Investigator award to M.J.B. The work was also supported by the Austin Doyle Award funded by Servier Australia (to E.A.B.A.). C.A.B. is supported by the Wellcome Trust PhD program in Metabolic and Cardiovascular Disease. J.Z. is supported by the Cambridge Overseas Trust and the Sun Hung Kai Properties–Kwoks' Foundation PhD program. G.S.H.Y. was supported by European Union FP7-HEALTH-2009-241592 EurOCHIP and FP7-FOOD-266408 Full4Health. Aarhus, Denmark. We thank J. Egebjerg Jensen for discussion of the electrophysiology data. H.P. was supported by grants from The Carlsberg Foundation, The Lundbeck Foundation and L'Oréal/UNESCO. University of Innsbruck, Austria. The work was supported by the Austrian Science Fund (F44020). University College London, UK. We thank W. Pratt for technical assistance. The work was supported by the Wellcome Trust (098360/Z/12/Z). Hradec Kralove, Czech Republic. We thank A. Ryska, who selected the most appropriate adrenal samples for the Czech cohort. Funding is provided by program PRVOUK P037/03. Nijmegen, The Netherlands. We thank J.W.M. Lenders for introducing the collaboration and for his leading role in the recruitment of the Dutch cohort.
Author information
Authors and Affiliations
Contributions
E.A.B.A. and M.J.B. designed and analyzed the adrenal experiments. H.P. and M.V.C. designed the electrophysiology experiments and performed cloning. H.P. performed and analyzed the electrophysiology experiments. M.V.C. made the homology model, and M.V.C., H.P. and P.N. discussed the structural analyses. A.C.D. and W.M. designed experiments on the Gly403Arg mutant of Cav1.3, undertaken by W.M. and K.C. K.C. performed protein blotting. P.T., A.L. and J.S. designed the experiments for the remaining Cav1.3 mutants. P.T. cloned the CACNA1D mutations, and A.L. performed whole-cell patch-clamp experiments. G.S.H.Y., S.G.N. and I.M. contributed to the design of RNA analyses, including for microarray analysis. E.G.B. and I.S.F. advised on the design and interpretation of exome sequencing. N.R., F.M. and J.H. designed and interpreted microfluidic sequencing. E.A.B.A. performed the H295R transfections with help from J.Z. J.Z. performed genotyping and Sanger sequencing with help from E.A.B.A., C.M., S.G. and E.G.B. Gene expression studies were performed by E.A.B.A., C.A.B., A.E.D.T., J.Z. and L.H.S. W.Z. performed immunohistochemistry, for which A.P.D. designed selective antisera to CYP11B1 and CYP11B2. For the Cambridge cohort, M.J.B., E.A.B.A., J.Z., C.M. and L.H.S. collected and prepared samples. For the Czech cohort, J.C. and M.S. collected and analyzed the clinical data, and E.A.B.A. and C.M. examined pathology, performed DNA isolation and prepared samples. For the Dutch cohort, J.D. executed the recruitment, B.K. examined pathology, B.T. performed DNA isolation, and T.D. prepared the samples. E.A.B.A. prepared the supplementary information, and I.S.F., J.S., H.P. and M.J.B. wrote the manuscript with comments from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1–4 (PDF 2053 kb)
Rights and permissions
About this article
Cite this article
Azizan, E., Poulsen, H., Tuluc, P. et al. Somatic mutations in ATP1A1 and CACNA1D underlie a common subtype of adrenal hypertension. Nat Genet 45, 1055–1060 (2013). https://doi.org/10.1038/ng.2716
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2716
This article is cited by
-
Pathogenicity of de novo CACNA1D Ca2+ channel variants predicted from sequence co-variation
European Journal of Human Genetics (2024)
-
CASZ1: a promising factor modulating aldosterone biosynthesis and mineralocorticoid receptor activity
Hypertension Research (2023)
-
Primary aldosteronism: molecular medicine meets public health
Nature Reviews Nephrology (2023)
-
Somatic SLC30A1 mutations altering zinc transporter ZnT1 cause aldosterone-producing adenomas and primary aldosteronism
Nature Genetics (2023)
-
KCNJ5 mutation is a predictor for recovery of endothelial function after adrenalectomy in patients with aldosterone-producing adenoma
Hypertension Research (2023)