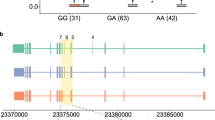
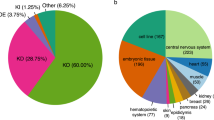
Abstract
Alternative pre–messenger RNA splicing influences development, physiology and disease, but its regulation in humans is not well understood, partially because of the limited scale at which the expression of specific splicing events has been measured. We generated the first genome-scale expression compendium of human alternative splicing events using custom whole-transcript microarrays monitoring expression of 24,426 alternative splicing events in 48 diverse human samples. Over 11,700 genes and 9,500 splicing events were differentially expressed, providing a rich resource for studying splicing regulation. An unbiased, systematic screen of 21,760 4-mer to 7-mer words for cis-regulatory motifs identified 143 RNA 'words' enriched near regulated cassette exons, including six clusters of motifs represented by UCUCU, UGCAUG, UGCU, UGUGU, UUUU and AGGG, which map to trans-acting regulators PTB, Fox, Muscleblind, CELF/CUG-BP, TIA-1 and hnRNP F/H, respectively. Each cluster showed a distinct pattern of genomic location and tissue specificity. For example, UCUCU occurs 110 to 35 nucleotides preceding cassette exons upregulated in brain and striated muscle but depleted in other tissues. UCUCU and UGCAUG seem to have similar function but independent action, occurring 5′ and 3′, respectively, of 33% of the cassette exons upregulated in skeletal muscle but co-occurring for only 2%.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Johnson, J.M. et al. Genome-wide survey of human alternative pre-mRNA splicing with exon junction microarrays. Science 302, 2141–2144 (2003).
Kwan, T. et al. Genome-wide analysis of transcript isoform variation in humans. Nat. Genet. 40, 225–231 (2008).
Chan, R.C. & Black, D.L. Conserved intron elements repress splicing of a neuron-specific c-src exon in vitro. Mol. Cell. Biol. 15, 6377–6385 (1995).
Singh, R., Valcarcel, J. & Green, M.R. Distinct binding specificities and functions of higher eukaryotic polypyrimidine tract-binding proteins. Science 268, 1173–1176 (1995).
Gooding, C., Roberts, G.C. & Smith, C.W. Role of an inhibitory pyrimidine element and polypyrimidine tract binding protein in repression of a regulated alpha-tropomyosin exon. RNA 4, 85–100 (1998).
Underwood, J.G., Boutz, P.L., Dougherty, J.D., Stoilov, P. & Black, D.L. Homologues of the Caenorhabditis elegans Fox-1 protein are neuronal splicing regulators in mammals. Mol. Cell. Biol. 25, 10005–10016 (2005).
Minovitsky, S., Gee, S.L., Schokrpur, S., Dubchak, I. & Conboy, J.G. The splicing regulatory element, UGCAUG, is phylogenetically and spatially conserved in introns that flank tissue-specific alternative exons. Nucleic Acids Res. 33, 714–724 (2005).
Martinez-Contreras, R. et al. Intronic binding sites for hnRNP A/B and hnRNP F/H proteins stimulate pre-mRNA splicing. PLoS Biol. 4, e21 (2006).
Han, K., Yeo, G., An, P., Burge, C.B. & Grabowski, P.J. A combinatorial code for splicing silencing: UAGG and GGGG motifs. PLoS Biol. 3, e158 (2005).
Ho, T.H. et al. Muscleblind proteins regulate alternative splicing. EMBO J. 23, 3103–3112 (2004).
Forch, P. et al. The apoptosis-promoting factor TIA-1 is a regulator of alternative pre-mRNA splicing. Mol. Cell 6, 1089–1098 (2000).
Faustino, N.A. & Cooper, T.A. Identification of putative new splicing targets for ETR-3 using sequences identified by systematic evolution of ligands by exponential enrichment. Mol. Cell. Biol. 25, 879–887 (2005).
Kuroyanagi, H., Ohno, G., Mitani, S. & Hagiwara, M. The Fox-1 family and SUP-12 coordinately regulate tissue-specific alternative splicing in vivo. Mol. Cell. Biol. 27, 8612–8621 (2007).
Ule, J. et al. Nova regulates brain-specific splicing to shape the synapse. Nat. Genet. 37, 844–852 (2005).
Ule, J. et al. CLIP identifies Nova-regulated RNA networks in the brain. Science 302, 1212–1215 (2003).
Pan, Q. et al. Revealing global regulatory features of mammalian alternative splicing using a quantitative microarray platform. Mol. Cell 16, 929–941 (2004).
Fagnani, M. et al. Functional coordination of alternative splicing in the mammalian central nervous system. Genome Biol. 8, R108 (2007).
Sugnet, C.W. et al. Unusual intron conservation near tissue-regulated exons found by splicing microarrays. PLOS Comput. Biol. 2, e4 (2006).
Clark, T.A. et al. Discovery of tissue-specific exons using comprehensive human exon microarrays. Genome Biol. 8, R64 (2007).
Griffith, M. et al. ALEXA: a microarray design platform for alternative expression analysis. Nat. Methods 5, 118 (2008).
Nakahata, S. & Kawamoto, S. Tissue-dependent isoforms of mammalian Fox-1 homologs are associated with tissue-specific splicing activities. Nucleic Acids Res. 33, 2078–2089 (2005).
Duncan, P.I., Stojdl, D.F., Marius, R.M. & Bell, J.C. In vivo regulation of alternative pre-mRNA splicing by the Clk1 protein kinase. Mol. Cell. Biol. 17, 5996–6001 (1997).
Rupp, U. et al. Safety and pharmacokinetics of bivatuzumab mertansine in patients with CD44v6-positive metastatic breast cancer: final results of a phase I study. Anticancer Drugs 18, 477–485 (2007).
Riechelmann, H. et al. Phase I trial with the CD44v6-targeting immunoconjugate bivatuzumab mertansine in head and neck squamous cell carcinoma. Oral Oncol. 44, 823–829 (2008).
Brudno, M. et al. Computational analysis of candidate intron regulatory elements for tissue-specific alternative pre-mRNA splicing. Nucleic Acids Res. 29, 2338–2348 (2001).
Yeo, G.W., Van Nostrand, E., Holste, D., Poggio, T. & Burge, C.B. Identification and analysis of alternative splicing events conserved in human and mouse. Proc. Natl. Acad. Sci. USA 102, 2850–2855 (2005).
Yeo, G.W., Nostrand, E.L. & Liang, T.Y. Discovery and analysis of evolutionarily conserved intronic splicing regulatory elements. PLoS Genet. 3, e85 (2007).
Yeo, G.W. et al. Alternative splicing events identified in human embryonic stem cells and neural progenitors. PLOS Comput. Biol. 3, 1951–1967 (2007).
Das, D. et al. A correlation with exon expression approach to identify cis-regulatory elements for tissue-specific alternative splicing. Nucleic Acids Res. 35, 4845–4857 (2007).
Voelker, R.B. & Berglund, J.A. A comprehensive computational characterization of conserved mammalian intronic sequences reveals conserved motifs associated with constitutive and alternative splicing. Genome Res. 17, 1023–1033 (2007).
Zhang, C. et al. Defining the regulatory network of the tissue-specific splicing factors Fox-1 and Fox-2. Genes Dev. 22, 2550–2563 (2008).
Liu, H.X., Zhang, M. & Krainer, A.R. Identification of functional exonic splicing enhancer motifs recognized by individual SR proteins. Genes Dev. 12, 1998–2012 (1998).
Fairbrother, W.G., Yeh, R.F., Sharp, P.A. & Burge, C.B. Predictive identification of exonic splicing enhancers in human genes. Science 297, 1007–1013 (2002).
Zhang, W. et al. The functional landscape of mouse gene expression. J. Biol. 3, 21 (2004).
Goren, A. et al. Comparative analysis identifies exonic splicing regulatory sequences–The complex definition of enhancers and silencers. Mol. Cell 22, 769–781 (2006).
Fairbrother, W.G. et al. RESCUE-ESE identifies candidate exonic splicing enhancers in vertebrate exons. Nucleic Acids Res. 32, W187–W190 (2004).
Perez, I., Lin, C.H., McAfee, J.G. & Patton, J.G. Mutation of PTB binding sites causes misregulation of alternative 3′ splice site selection in vivo. RNA 3, 764–778 (1997).
Jensen, K.B., Musunuru, K., Lewis, H.A., Burley, S.K. & Darnell, R.B. The tetranucleotide UCAY directs the specific recognition of RNA by the Nova K-homology 3 domain. Proc. Natl. Acad. Sci. USA 97, 5740–5745 (2000).
Ule, J. et al. An RNA map predicting Nova-dependent splicing regulation. Nature 444, 580–586 (2006).
Wheeler, D.L. et al. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 36, D13–D21 (2008).
Florea, L., Hartzell, G., Zhang, Z., Rubin, G.M. & Miller, W. A computer program for aligning a cDNA sequence with a genomic DNA sequence. Genome Res. 8, 967–974 (1998).
Kan, Z., Castle, J., Johnson, J.M. & Tsinoremas, N.F. Detection of novel splice forms in human and mouse using cross-species approach. Pac. Symp. Biocomput. 9, 42–53 (2004).
Castle, J. et al. Optimization of oligonucleotide arrays and RNA amplification protocols for analysis of transcript structure and alternative splicing. Genome Biol. 4, R66 (2003).
Hughes, T.R. et al. Expression profiling using microarrays fabricated by an ink-jet oligonucleotide synthesizer. Nat. Biotechnol. 19, 342–347 (2001).
Weng, L. et al. Rosetta error model for gene expression analysis. Bioinformatics 22, 1111–1121 (2006).
International Human Genome Sequencing Consortium. Finishing the euchromatic sequence of the human genome. Nature 431, 931–945 (2004).
Fehlbaum, P., Guihal, C., Bracco, L. & Cochet, O. A microarray configuration to quantify expression levels and relative abundance of splice variants. Nucleic Acids Res. 33, e47 (2005).
Taylor, J.R. An Introduction to Error Analysis 270 (University Science Books, Mill Valley, California, 1982).
Fairbrother, W.G., Holste, D., Burge, C.B. & Sharp, P.A. Single nucleotide polymorphism-based validation of exonic splicing enhancers. PLoS Biol 2, E268 (2004).
Acknowledgements
We thank C. Armour, C. Raymond, D. Haynor, V. Emilsson, P. Garrett-Engele, F. Roth, P. Loerch, R. Chen, C. Rohl, M. Tompa, E. Schadt and L. Lim for input, Rosetta's Gene Expression Laboratory microarray hybridization facility for microarray data, and S. Carlson for project management. Funding for T.A.C. provided by R01GM076493.
Author information
Authors and Affiliations
Contributions
J.M.J. conceived the experiment; J.C.C. and J.M.J. executed the experiment and wrote the paper; J.C.C. designed the microarrays and analyzed the data; J.C.C. and C.Z. identified motifs; J.K.S. and A.V.K. processed and managed data; and A.K. and T.A.C. provided validation experiments.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Table 1, Supplementary Figures 1–6 and Supplementary Note (PDF 1449 kb)
Rights and permissions
About this article
Cite this article
Castle, J., Zhang, C., Shah, J. et al. Expression of 24,426 human alternative splicing events and predicted cis regulation in 48 tissues and cell lines. Nat Genet 40, 1416–1425 (2008). https://doi.org/10.1038/ng.264
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.264
This article is cited by
-
Comprehensive analysis of pre-mRNA alternative splicing regulated by m6A methylation in pig oxidative and glycolytic skeletal muscles
BMC Genomics (2022)
-
Genome-wide analysis reveals the association between alternative splicing and DNA methylation across human solid tumors
BMC Medical Genomics (2020)
-
Identification of Prognostic Signatures of Alternative Splicing in Glioma
Journal of Molecular Neuroscience (2020)
-
miR-1/206 downregulates splicing factor Srsf9 to promote C2C12 differentiation
Skeletal Muscle (2019)
-
The complexity of titin splicing pattern in human adult skeletal muscles
Skeletal Muscle (2018)