Abstract
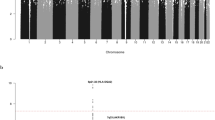
Osteosarcoma is the most common primary bone malignancy of adolescents and young adults. To better understand the genetic etiology of osteosarcoma, we performed a multistage genome-wide association study consisting of 941 individuals with osteosarcoma (cases) and 3,291 cancer-free adult controls of European ancestry. Two loci achieved genome-wide significance: a locus in the GRM4 gene at 6p21.3 (encoding glutamate receptor metabotropic 4; rs1906953; P = 8.1 × 10−9) and a locus in the gene desert at 2p25.2 (rs7591996 and rs10208273; P = 1.0 × 10−8 and 2.9 × 10−7, respectively). These two loci warrant further exploration to uncover the biological mechanisms underlying susceptibility to osteosarcoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Mirabello, L., Troisi, R. & Savage, S. Osteosarcoma incidence and survival rates from 1973 to 2004: data from the Surveillance, Epidemiology, and End Results Program. Cancer 115, 1531–1543 (2009).
Mirabello, L. et al. Height at diagnosis and birth-weight as risk factors for osteosarcoma. Cancer Causes Control 22, 899–908 (2011).
Patio-Garcia, A., Sotillo-Pieiro, E., Modesto, C. & Sierrases-Maga, L. Analysis of the human tumour necrosis factor-α (TNFα) gene promoter polymorphisms in children with bone cancer. J. Med. Genet. 37, 789–792 (2000).
Ruza, E., Sotillo, E., Sierrasesúmaga, L., Azcona, C. & Patiño-Garcia, A. Analysis of polymorphisms of the vitamin D receptor, estrogen receptor, and collagen Iα1 genes and their relationship with height in children with bone cancer. J. Pediatr. Hematol. Oncol. 25, 780–786 (2003).
Savage, S.A. et al. Germ-line genetic variation of TP53 in osteosarcoma. Pediatr. Blood Cancer 49, 28–33 (2007).
Savage, S.A. et al. Analysis of genes critical for growth regulation identifies Insulin-like Growth Factor 2 Receptor variations with possible functional significance as risk factors for osteosarcoma. Cancer Epidemiol. Biomarkers Prev. 16, 1667–1674 (2007).
Koshkina, N.V. et al. Exploratory analysis of Fas gene polymorphisms in pediatric osteosarcoma patients. J. Pediatr. Hematol. Oncol. 29, 815–821 (2007).
Toffoli, G. et al. Effect of TP53 Arg72Pro and MDM2 SNP309 polymorphisms on the risk of high-grade osteosarcoma development and survival. Clin. Cancer Res. 15, 3550–3556 (2009).
Hu, Y.S. et al. Association between TGFBR1*6A and osteosarcoma: a Chinese case-control study. BMC Cancer 10, 169 (2010).
Mirabello, L. et al. Genetic variation at chromosome 8q24 in osteosarcoma cases and controls. Carcinogenesis 31, 1400–1404 (2010).
Mirabello, L. et al. A comprehensive candidate gene approach identifies genetic variation associated with osteosarcoma. BMC Cancer 11, 209 (2011).
Savage, S.A. & Mirabello, L. Using epidemiology and genomics to understand osteosarcoma etiology. Sarcoma 2011, 548151 (2011).
Postel-Vinay, S. et al. Common variants near TARDBP and EGR2 are associated with susceptibility to Ewing sarcoma. Nat. Genet. 44, 323–327 (2012).
Papaemmanuil, E. et al. Loci on 7p12.2, 10q21.2 and 14q11.2 are associated with risk of childhood acute lymphoblastic leukemia. Nat. Genet. 41, 1006–1010 (2009).
Thomas, G. et al. Multiple loci identified in a genome-wide association study of prostate cancer. Nat. Genet. 40, 310–315 (2008).
Amundadottir, L. et al. Genome-wide association study identifies variants in the ABO locus associated with susceptibility to pancreatic cancer. Nat. Genet. 41, 986–990 (2009).
Rothman, N. et al. A multi-stage genome-wide association study of bladder cancer identifies multiple susceptibility loci. Nat. Genet. 42, 978–984 (2010).
Landi, M.T. et al. A genome-wide association study of lung cancer identifies a region of chromosome 5p15 associated with risk for adenocarcinoma. Am. J. Hum. Genet. 85, 679–691 (2009).
Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447, 661–678 (2007).
Li, J. et al. GWAS of blood cell traits identifies novel associated loci and epistatic interactions in Caucasian and African-American children. Hum. Mol. Genet. 22, 1457–1464 (2013).
Gieger, C. et al. New gene functions in megakaryopoiesis and platelet formation. Nature 480, 201–208 (2011).
Qayyum, R. et al. A meta-analysis and genome-wide association study of platelet count and mean platelet volume in African Americans. PLoS Genet. 8, e1002491 (2012).
Slager, S.L. et al. Common variation at 6p21.31 (BAK1) influences the risk of chronic lymphocytic leukemia. Blood 120, 843–846 (2012).
Rapley, E.A. et al. A genome-wide association study of testicular germ cell tumor. Nat. Genet. 41, 807–810 (2009).
1000 Genomes Project Consortium. A map of human genome variation from population-scale sequencing. Nature 467, 1061–1073 (2010).
ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57–74 (2012).
Molyneux, S.D. et al. Prkar1a is an osteosarcoma tumor suppressor that defines a molecular subclass in mice. J. Clin. Invest. 120, 3310–3325 (2010).
Griffin, K.J. et al. A transgenic mouse bearing an antisense construct of regulatory subunit type 1A of protein kinase A develops endocrine and other tumours: comparison with Carney complex and other PRKAR1A induced lesions. J. Med. Genet. 41, 923–931 (2004).
Cowan, R.W., Seidlitz, E.P. & Singh, G. Glutamate signaling in healthy and diseased bone. Front. Endocrinol. (Lausanne) 3, 89 (2012).
Skerry, T.M. The role of glutamate in the regulation of bone mass and architecture. J. Musculoskelet. Neuronal Interact. 8, 166–173 (2008).
Kalariti, N., Lembessis, P. & Koutsilieris, M. Characterization of the glutametergic system in MG-63 osteoblast-like osteosarcoma cells. Anticancer Res. 24, 3923–3929 (2004).
Chang, H.J. et al. Metabotropic glutamate receptor 4 expression in colorectal carcinoma and its prognostic significance. Clin. Cancer Res. 11, 3288–3295 (2005).
Brocke, K.S. et al. Glutamate receptors in pediatric tumors of the central nervous system. Cancer Biol. Ther. 9, 455–468 (2010).
Stepulak, A. et al. Expression of glutamate receptor subunits in human cancers. Histochem. Cell Biol. 132, 435–445 (2009).
Luksch, H. et al. Silencing of selected glutamate receptor subunits modulates cancer growth. Anticancer Res. 31, 3181–3192 (2011).
Hirschhorn, J.N. & Lettre, G. Progress in genome-wide association studies of human height. Horm. Res. 71 (suppl. 2), 5–13 (2009).
Wang, Z. et al. Improved imputation of common and uncommon SNPs with a new reference set. Nat. Genet. 44, 6–7 (2012).
Howie, B.N., Donnelly, P. & Marchini, J. A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet. 5, e1000529 (2009).
Purdue, M.P. et al. Genome-wide association study of renal cell carcinoma identifies two susceptibility loci on 2p21 and 11q13.3. Nat. Genet. 43, 60–65 (2011).
Fearnhead, P. SequenceLDhot: detecting recombination hotspots. Bioinformatics 22, 3061–3066 (2006).
Fearnhead, P. & Donnelly, P. Approximate likelihood methods for estimating local recombination rates. J. R. Stat. Soc. Series B Stat. Methodol. 64, 657–680 (2002).
Crawford, D.C. et al. Evidence for substantial fine-scale variation in recombination rates across the human genome. Nat. Genet. 36, 700–706 (2004).
Li, N. & Stephens, M. Modeling linkage disequilibrium and identifying recombination hotspots using single-nucleotide polymorphism data. Genetics 165, 2213–2233 (2003).
Luna, A. & Nicodemus, K.K. snp.plotter: an R-based SNP/haplotype association and linkage disequilibrium plotting package. Bioinformatics 23, 774–776 (2007).
Marchini, J., Howie, B., Myers, S., McVean, G. & Donnelly, P. A new multipoint method for genome-wide association studies by imputation of genotypes. Nat. Genet. 39, 906–913 (2007).
Acknowledgements
We thank G. Maganoli for tissue banking, M. Fanelli for DNA isolation and C. Ferrari for updating clinicopathological data at the Orthopaedic Rizzoli Institute. We thank A. Griffin and D. Marsilio for data collection and T. Selander and the Biospecimen Repository staff at Mount Sinai Hospital. We acknowledge the advice of F. Real at the Spanish National Cancer Research Centre (CNIO). We thank F. Tesser Gamba at the Pediatric Oncology Institute at GRAACC-UNIFESP, and we also thank the International Sarcoma Kindred Study.
This study was funded by the intramural research program of the Division of Cancer Epidemiology and Genetics, National Cancer Institute, US National Institutes of Health and the Bone Cancer Research Trust UK. Research is supported by the Chair's Grant U10 CA98543 and Human Specimen Banking Grant U24 CA114766 to the Children's Oncology Group from the National Cancer Institute, US National Institutes of Health. Additional support for research is provided by a grant from the WWW.W (QuadW) Foundation to the Children's Oncology Group. This work was supported by grants to I.L.A. and J.S.W. from the Ontario Research Fund and Canadian Foundation for Innovation. This study was also supported by biobank grants from the Regione Emilia-Romagna and by the infrastructure and personnel of the Royal National Orthopaedic Hospital Musculoskeletal Research Programme and Biobank. Support was also provided to A.M.F. by the National Institute for Health Research UCL Hospitals (UCLH) Biomedical Research Centre and the UCL Experimental Cancer Centre. Funding was also provided by PI10/01580, the Fondo de Investigación Sanitaria (FIS), the Instituto de Salud Carlos III (ISCIII) and the Caja de Ahorros de Navarra (CAN) Programme 'Tú eliges, tú decides' to A.P.-G. and L.S. and by an Asociación Española Contra el Cáncer (AECC) project to F.L.
Author information
Authors and Affiliations
Contributions
S.A.S. and S.J.C. designed the project. J.M.G.-F., R.G., C.K., A.M.F., R. Tirabosco, I.L.A., J.S.W., N.G., L.G.S., D.A.B., N. Marina, A.P.-G., L.S., F.L., M.S., C.H., P.P., N.K., I.E.I., N.S., S.R.C.d.T., A.S.P., M.F.A., D.H., D.M.T., C.D., P.S.M., S.I.B., M.P.P., N.E.C., M.T., N.R., M.T.L., D.T.S., P.K., D.J.H., N. Malats, M.K., S.W., R. Troisi, L.H., J.F.F. and R.N.H. performed sample collection and clinical characterization. K.J., C.C.C., M.Y. and Z.W. performed genotyping. Z.W. and L.M. performed statistical analysis. The manuscript was written by S.A.S., L.M., Z.W. and S.J.C. and reviewed by all coauthors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–4, Supplementary Figures 1–4 (PDF 2222 kb)
Rights and permissions
About this article
Cite this article
Savage, S., Mirabello, L., Wang, Z. et al. Genome-wide association study identifies two susceptibility loci for osteosarcoma. Nat Genet 45, 799–803 (2013). https://doi.org/10.1038/ng.2645
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2645
This article is cited by
-
Glutamate metabotropic receptor 4 (GRM4) inhibits cell proliferation, migration and invasion in breast cancer and is regulated by miR-328-3p and miR-370-3p
BMC Cancer (2019)
-
Risk-modeling of dog osteosarcoma genome scans shows individuals with Mendelian-level polygenic risk are common
BMC Genomics (2019)
-
Rhabdomyosarcoma
Nature Reviews Disease Primers (2019)
-
Whole-genome re-sequencing association study for direct genetic effects and social genetic effects of six growth traits in Large White pigs
Scientific Reports (2019)
-
Identification of key gene modules for human osteosarcoma by co-expression analysis
World Journal of Surgical Oncology (2018)