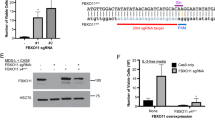
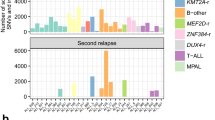
Abstract
T-cell acute lymphoblastic leukemia (T-ALL) is caused by the cooperation of multiple oncogenic lesions1,2. We used exome sequencing on 67 T-ALLs to gain insight into the mutational spectrum in these leukemias. We detected protein-altering mutations in 508 genes, with an average of 8.2 mutations in pediatric and 21.0 mutations in adult T-ALL. Using stringent filtering, we predict seven new oncogenic driver genes in T-ALL. We identify CNOT3 as a tumor suppressor mutated in 7 of 89 (7.9%) adult T-ALLs, and its knockdown causes tumors in a sensitized Drosophila melanogaster model3. In addition, we identify mutations affecting the ribosomal proteins RPL5 and RPL10 in 12 of 122 (9.8%) pediatric T-ALLs, with recurrent alterations of Arg98 in RPL10. Yeast and lymphoid cells expressing the RPL10 Arg98Ser mutant showed a ribosome biogenesis defect. Our data provide insights into the mutational landscape of pediatric versus adult T-ALL and identify the ribosome as a potential oncogenic factor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Grabher, C., von Boehmer, H. & Look, A.T. Notch 1 activation in the molecular pathogenesis of T-cell acute lymphoblastic leukaemia. Nat. Rev. Cancer 6, 347–359 (2006).
Van Vlierberghe, P. & Ferrando, A. The molecular basis of T cell acute lymphoblastic leukemia. J. Clin. Invest. 122, 3398–3406 (2012).
Ferres-Marco, D. et al. Epigenetic silencers and Notch collaborate to promote malignant tumours by Rb silencing. Nature 439, 430–436 (2006).
De Keersmaecker, K., Marynen, P. & Cools, J. Genetic insights in the pathogenesis of T-cell acute lymphoblastic leukemia. Haematologica 90, 1116–1127 (2005).
Pui, C.H., Relling, M.V. & Downing, J.R. Acute lymphoblastic leukemia. N. Engl. J. Med. 350, 1535–1548 (2004).
Homminga, I. et al. Integrated transcript and genome analyses reveal NKX2-1 and MEF2C as potential oncogenes in T cell acute lymphoblastic leukemia. Cancer Cell 19, 484–497 (2011).
Ntziachristos, P. et al. Genetic inactivation of the polycomb repressive complex 2 in T cell acute lymphoblastic leukemia. Nat. Med. 18, 298–301 (2012).
Zhang, J. et al. The genetic basis of early T-cell precursor acute lymphoblastic leukaemia. Nature 481, 157–163 (2012).
Larson, D.E. et al. SomaticSniper: identification of somatic point mutations in whole genome sequencing data. Bioinformatics 28, 311–317 (2012).
Dees, N.D. et al. MuSiC: identifying mutational significance in cancer genomes. Genome Res. 22, 1589–1598 (2012).
Narla, A. & Ebert, B.L. Ribosomopathies: human disorders of ribosome dysfunction. Blood 115, 3196–3205 (2010).
Rao, S. et al. Inactivation of ribosomal protein L22 promotes transformation by induction of the stemness factor, Lin28B. Blood 120, 3764–3773 (2012).
Hofer, A., Bussiere, C. & Johnson, A.W. Mutational analysis of the ribosomal protein Rpl10 from yeast. J. Biol. Chem. 282, 32630–32639 (2007).
Hedges, J. et al. Release of the export adapter, Nmd3p, from the 60S ribosomal subunit requires Rpl10p and the cytoplasmic GTPase Lsg1p. EMBO J. 24, 567–579 (2005).
Lo, K.Y. et al. Defining the pathway of cytoplasmic maturation of the 60S ribosomal subunit. Mol. Cell 39, 196–208 (2010).
Collart, M.A. & Panasenko, O.O. The Ccr4-not complex. Gene 492, 42–53 (2012).
Hu, G. et al. A genome-wide RNAi screen identifies a new transcriptional module required for self-renewal. Genes Dev. 23, 837–848 (2009).
Palomero, T. et al. Mutational loss of PTEN induces resistance to NOTCH1 inhibition in T-cell leukemia. Nat. Med. 13, 1203–1210 (2007).
Bossuyt, W. et al. The atonal proneural transcription factor links differentiation and tumor formation in Drosophila. PLoS Biol. 7, e40 (2009).
Quesada, V. et al. Exome sequencing identifies recurrent mutations of the splicing factor SF3B1 gene in chronic lymphocytic leukemia. Nat. Genet. 44, 47–52 (2012).
Graubert, T.A. et al. Recurrent mutations in the U2AF1 splicing factor in myelodysplastic syndromes. Nat. Genet. 44, 53–57 (2012).
Yoshida, K. et al. Frequent pathway mutations of splicing machinery in myelodysplasia. Nature 478, 64–69 (2011).
Papaemmanuil, E. et al. Somatic SF3B1 mutation in myelodysplasia with ring sideroblasts. N. Engl. J. Med. 365, 1384–1395 (2011).
Ruggero, D. & Pandolfi, P.P. Does the ribosome translate cancer? Nat. Rev. Cancer 3, 179–192 (2003).
Weng, A.P. et al. Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. Science 306, 269–271 (2004).
Fabbri, G. et al. Analysis of the chronic lymphocytic leukemia coding genome: role of NOTCH1 mutational activation. J. Exp. Med. 208, 1389–1401 (2011).
Puente, X.S. et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukaemia. Nature 475, 101–105 (2011).
Westhoff, B. et al. Alterations of the Notch pathway in lung cancer. Proc. Natl. Acad. Sci. USA 106, 22293–22298 (2009).
Agrawal, N. et al. Exome sequencing of head and neck squamous cell carcinoma reveals inactivating mutations in NOTCH1. Science 333, 1154–1157 (2011).
Stransky, N. et al. The mutational landscape of head and neck squamous cell carcinoma. Science 333, 1157–1160 (2011).
Robinson, D.R. et al. Functionally recurrent rearrangements of the MAST kinase and Notch gene families in breast cancer. Nat. Med. 17, 1646–1651 (2011).
Jiao, X. et al. Somatic mutations in the Notch, NF-κB, PIK3CA, and Hedgehog pathways in human breast cancers. Genes Chromosom. Cancer 51, 480–489 (2012).
Thompson, B.J. et al. The SCFFBW7 ubiquitin ligase complex as a tumor suppressor in T cell leukemia. J. Exp. Med. 204, 1825–1835 (2007).
Welcker, M. & Clurman, B.E. FBW7 ubiquitin ligase: a tumour suppressor at the crossroads of cell division, growth and differentiation. Nat. Rev. Cancer 8, 83–93 (2008).
Tosello, V. et al. WT1 mutations in T-ALL. Blood 114, 1038–1045 (2009).
King-Underwood, L. & Pritchard-Jones, K. Wilms' tumor (WT1) gene mutations occur mainly in acute myeloid leukemia and may confer drug resistance. Blood 91, 2961–2968 (1998).
De Keersmaecker, K. et al. The TLX1 oncogene drives aneuploidy in T cell transformation. Nat. Med. 16, 1321–1327 (2010).
Klauck, S.M. et al. Mutations in the ribosomal protein gene RPL10 suggest a novel modulating disease mechanism for autism. Mol. Psychiatry 11, 1073–1084 (2006).
Chiocchetti, A. et al. Mutation and expression analyses of the ribosomal protein gene RPL10 in an extended German sample of patients with autism spectrum disorder. Am. J. Med. Genet. A. 155A, 1472–1475 (2011).
Kalender Atak, Z. et al. High accuracy mutation detection in leukemia on a selected panel of cancer genes. PLoS ONE 7, e38463 (2012).
Durieux, A.C., Prudhon, B., Guicheney, P. & Bitoun, M. Dynamin 2 and human diseases. J. Mol. Med. 88, 339–350 (2010).
Van Vlierberghe, P. et al. PHF6 mutations in T-cell acute lymphoblastic leukemia. Nat. Genet. 42, 338–342 (2010).
Van Vlierberghe, P. et al. ETV6 mutations in early immature human T cell leukemias. J. Exp. Med. 208, 2571–2579 (2011).
Lower, K.M. et al. 1024C> T (R342X) is a recurrent PHF6 mutation also found in the original Börjeson-Forssman-Lehmann syndrome family. Eur. J. Hum. Genet. 12, 787–789 (2004).
Ono, R. et al. LCX, leukemia-associated protein with a CXXC domain, is fused to MLL in acute myeloid leukemia with trilineage dysplasia having t(10;11)(q22;q23). Cancer Res. 62, 4075–4080 (2002).
Lorsbach, R.B. et al. TET1, a member of a novel protein family, is fused to MLL in acute myeloid leukemia containing the t(10;11)(q22;q23). Leukemia 17, 637–641 (2003).
Burmeister, T. et al. The MLL recombinome of adult CD10-negative B-cell precursor acute lymphoblastic leukemia: results from the GMALL study group. Blood 113, 4011–4015 (2009).
van Haaften, G. et al. Somatic mutations of the histone H3K27 demethylase gene UTX in human cancer. Nat. Genet. 41, 521–523 (2009).
Gui, Y. et al. Frequent mutations of chromatin remodeling genes in transitional cell carcinoma of the bladder. Nat. Genet. 43, 875–878 (2011).
Lederer, D. et al. Deletion of KDM6A, a histone demethylase interacting with MLL2, in three patients with Kabuki syndrome. Am. J. Hum. Genet. 90, 119–124 (2012).
Pasqualucci, L. et al. Analysis of the coding genome of diffuse large B-cell lymphoma. Nat. Genet. 43, 830–837 (2011).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
DePristo, M.A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 43, 491–498 (2011).
Larson, D.E. et al. SomaticSniper: identification of somatic point mutations in whole genome sequencing data. Bioinformatics 28, 311–317 (2012).
Albers, C.A. et al. Dindel: accurate indel calls from short-read data. Genome Res. 21, 961–973 (2011).
Bellen, H.J. et al. The BDGP gene disruption project: single transposon insertions associated with 40% of Drosophila genes. Genetics 167, 761–781 (2004).
Dietzl, G. et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 448, 151–156 (2007).
Acknowledgements
This work was supported by grants from KU Leuven (concerted action grant to J. Cools and P.V. and PF/10/016 SymBioSys to J. Cools and S.A.), FWO-Vlaanderen (G.0546.11 to J. Cools, P.V., S.A. and A.U. and G.0704.11N to S.A.), the Foundation against Cancer (SCIE2006-34 to J. Cools and 2010-154 to S.A.), a European Research Council (ERC) starting grant (J. Cools), the Interuniversity Attraction Poles (IAP) granted by the Federal Office for Scientific, Technical and Cultural Affairs, Belgium (J. Cools and P.V.), a grant from the Ministry of Health, Cancer Plan (J. Cools, P.V. and S.A.), a grant from the French program Carte d'Identité des Tumeurs (CIT, Ligue Contre le Cancer) and from Canceropole d'Ile de France (J.S.), and a grant from the US National Institutes of Health (NIH; GM53655 to A.W.J. and S.P.). K.D.K. is a postdoctoral researcher, and P.V. is a senior clinical investigator of FWO-Vlaanderen.
Author information
Authors and Affiliations
Contributions
All authors contributed to the writing of the manuscript. K.D.K., Z.K.A., N.L., C.B., B.A.H. and A.W.J. designed and performed experiments and analyzed data. C.V. and J.Y. performed and analyzed Not3 Drosophila experiments. S.P. performed and analyzed Rpl10 yeast studies. R.L. performed and analyzed polysome profiling experiments. T.G., V.G., E.G., M.P., I.L., G.H., E.C., R.V., B.S., K.J., N.M. and I.W. performed experiments and analyzed data. H.V., B.C., J. Cloos, J.S., A.U. and P.V. collected samples and analyzed data. S.A. and J. Cools supervised the project, designed experiments and analyzed data.
Corresponding authors
Ethics declarations
Competing interests
The authors declare competing financial interests: N.L. is employed by BGI.
Supplementary information
Supplementary Text and Figures
Supplementary Note, Supplementary Figures 1–9 and Supplementary Tables 1–4, 6 and 8–11 (PDF 6404 kb)
Supplementary Table 5
Protein-altering (A) somatic SNVs and (B) somatic INDELs in 39 diagnosis-remission pairs (XLSX 249 kb)
Supplementary Table 7
Protein-altering SNVs and INDELs in 15 candidate genes in (A) diagnosis-only samples and (B) cell lines (XLSX 96 kb)
Rights and permissions
About this article
Cite this article
De Keersmaecker, K., Atak, Z., Li, N. et al. Exome sequencing identifies mutation in CNOT3 and ribosomal genes RPL5 and RPL10 in T-cell acute lymphoblastic leukemia. Nat Genet 45, 186–190 (2013). https://doi.org/10.1038/ng.2508
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2508
This article is cited by
-
Spectrum and clinical features of gene mutations in Chinese pediatric acute lymphoblastic leukemia
BMC Pediatrics (2023)
-
The ufmylation modification of ribosomal protein L10 in the development of pancreatic adenocarcinoma
Cell Death & Disease (2023)
-
Molecular profiling of a real-world breast cancer cohort with genetically inferred ancestries reveals actionable tumor biology differences between European ancestry and African ancestry patient populations
Breast Cancer Research (2023)
-
Panels of mRNAs and miRNAs for decoding molecular mechanisms of Renal Cell Carcinoma (RCC) subtypes utilizing Artificial Intelligence approaches
Scientific Reports (2022)
-
PSEN1-selective gamma-secretase inhibition in combination with kinase or XPO-1 inhibitors effectively targets T cell acute lymphoblastic leukemia
Journal of Hematology & Oncology (2021)