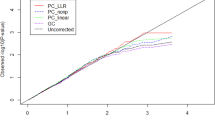
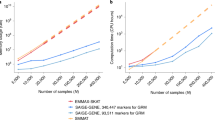
Abstract
The variance component tests used in genome-wide association studies (GWAS) including large sample sizes become computationally exhaustive when the number of genetic markers is over a few hundred thousand. We present an extremely fast variance components–based two-step method, GRAMMAR-Gamma, developed as an analytical approximation within a framework of the score test approach. Using simulated and real human GWAS data sets, we show that this method provides unbiased estimates of the SNP effect and has a power close to that of the likelihood ratio test–based method. The computational complexity of our method is close to its theoretical minimum, that is, to the complexity of the analysis that ignores genetic structure. The running time of our method linearly depends on sample size, whereas this dependency is quadratic for other existing methods. Simulations suggest that GRAMMAR-Gamma may be used for association testing in whole-genome resequencing studies of large human cohorts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Helgason, A., Yngvadóttir, B., Hrafnkelsson, B., Gulcher, J. & Stefánsson, K. An Icelandic example of the impact of population structure on association studies. Nat. Genet. 37, 90–95 (2005).
Astle, W. & Balding, D.J. Population structure and cryptic relatedness in genetic association studies. Stat. Sci. 24, 451–471 (2009).
Fisher, R.A. The correlation between relatives on the supposition of Mendelian inheritance. Trans. R. Soc. Edinb. 52, 399–433 (1918).
Henderson, C.R. Estimation of variance and covariance components. Biometrics 9, 226–252 (1953).
Boerwinkle, E., Chakraborty, R. & Sing, C.F. The use of measured genotype information in the analysis of quantitative phenotypes in man. I. Models and analytical methods. Ann. Hum. Genet. 50, 181–194 (1986).
Yu, J. et al. A unified mixed-model method for association mapping that accounts for multiple levels of relatedness. Nat. Genet. 38, 203–208 (2006).
Kang, H.M. et al. Efficient control of population structure in model organism association mapping. Genetics 178, 1709–1723 (2008).
Lippert, C. et al. FaST linear mixed models for genome-wide association studies. Nat. Methods 8, 833–835 (2011).
Chen, W.M. & Abecasis, G.R. Family-based association tests for genomewide association scans. Am. J. Hum. Genet. 81, 913–926 (2007).
Kang, H.M. et al. Variance component model to account for sample structure in genome-wide association studies. Nat. Genet. 42, 348–354 (2010).
Zhang, Z. et al. Mixed linear model approach adapted for genome-wide association studies. Nat. Genet. 42, 355–360 (2010).
Aulchenko, Y.S., de Koning, D.J. & Haley, C. Genomewide rapid association using mixed model and regression: a fast and simple method for genomewide pedigree-based quantitative trait loci association analysis. Genetics 177, 577–585 (2007).
Amin, N., van Duijn, C.M. & Aulchenko, Y.S. A genomic background based method for association analysis in related individuals. PLoS ONE 2, e1274 (2007).
Pardo, L.M. et al. The effect of genetic drift in a young genetically isolated population. Ann. Hum. Genet. 69, 288–295 (2005).
Atwell, S. et al. Genome-wide association study of 107 phenotypes in Arabidopsis thaliana inbred lines. Nature 465, 627–631 (2010).
Aulchenko, Y.S. et al. GenABEL: an R library for genome-wide association analysis. Bioinformatics 23, 1294–1296 (2007).
Devlin, B. & Roeder, K. Genomic control for association studies. Biometrics 55, 997–1004 (1999).
Bacanu, S.A., Devlin, B. & Roeder, K. Association studies for quantitative traits in structured populations. Genet. Epidemiol. 22, 78–93 (2002).
Astle, W. Population Structure and Cryptic Relatedness in Genetic Association Studies. PhD Thesis, University of London (2009).
Acknowledgements
We thank A. Kirichenko, D. Fabregat Traver and P. Bientinesi for technical support and advice and M. Axenovich, D. Balding, P. Borodin and W. Astle for discussion. This work was supported by grants from the Russian Foundation for Basic Research (RFBR) Programs of the Russian Academy of Sciences and the RFBR-Helmholtz Joint Research Groups program (research project 12-04-91322- ).
).
Author information
Authors and Affiliations
Contributions
G.R.S. developed the GRAMMAR-Gamma statistical test, ran the simulations and analyzed the simulated data. N.M.B. analyzed human and A. thaliana data and designed figures and tables. C.M.v.D. provided the human data and supervised its analyses. T.I.A. and Y.S.A. jointly designed and supervised the project and wrote the paper. All authors contributed to critical review of the manuscript during its preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–6, Supplementary Figures 1 and 2 and Supplementary Note (PDF 797 kb)
Rights and permissions
About this article
Cite this article
Svishcheva, G., Axenovich, T., Belonogova, N. et al. Rapid variance components–based method for whole-genome association analysis. Nat Genet 44, 1166–1170 (2012). https://doi.org/10.1038/ng.2410
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2410
This article is cited by
-
Large-scale GWAS of food liking reveals genetic determinants and genetic correlations with distinct neurophysiological traits
Nature Communications (2022)
-
Fine-mapping and association analysis of candidate genes for papilla number in sea cucumber, Apostichopus japonicus
Marine Life Science & Technology (2022)
-
Performance of model-based multifactor dimensionality reduction methods for epistasis detection by controlling population structure
BioData Mining (2021)
-
Association between genes regulating neural pathways for quantitative traits of speech and language disorders
npj Genomic Medicine (2021)
-
Controlling for background genetic effects using polygenic scores improves the power of genome-wide association studies
Scientific Reports (2021)