Abstract
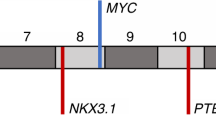
Prostate cancer is the second most common cancer in men worldwide and causes over 250,000 deaths each year1. Overtreatment of indolent disease also results in significant morbidity2. Common genetic alterations in prostate cancer include losses of NKX3.1 (8p21)3,4 and PTEN (10q23)5,6, gains of AR (the androgen receptor gene)7,8 and fusion of ETS family transcription factor genes with androgen-responsive promoters9,10,11. Recurrent somatic base-pair substitutions are believed to be less contributory in prostate tumorigenesis12,13 but have not been systematically analyzed in large cohorts. Here, we sequenced the exomes of 112 prostate tumor and normal tissue pairs. New recurrent mutations were identified in multiple genes, including MED12 and FOXA1. SPOP was the most frequently mutated gene, with mutations involving the SPOP substrate-binding cleft in 6–15% of tumors across multiple independent cohorts. Prostate cancers with mutant SPOP lacked ETS family gene rearrangements and showed a distinct pattern of genomic alterations. Thus, SPOP mutations may define a new molecular subtype of prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jemal, A. et al. Global cancer statistics. CA Cancer J. Clin. 61, 69–90 (2011).
Daskivich, T.J. et al. Overtreatment of men with low-risk prostate cancer and significant comorbidity. Cancer 117, 2058–2066 (2011).
He, W.W. et al. A novel human prostate-specific, androgen-regulated homeobox gene (NKX3.1) that maps to 8p21, a region frequently deleted in prostate cancer. Genomics 43, 69–77 (1997).
Bhatia-Gaur, R. et al. Roles for Nkx3.1 in prostate development and cancer. Genes Dev. 13, 966–977 (1999).
Li, J. et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275, 1943–1947 (1997).
Cairns, P. et al. Frequent inactivation of PTEN/MMAC1 in primary prostate cancer. Cancer Res. 57, 4997–5000 (1997).
Linja, M.J. & Visakorpi, T. Alterations of androgen receptor in prostate cancer. J. Steroid Biochem. Mol. Biol. 92, 255–264 (2004).
Visakorpi, T. et al. In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nat. Genet. 9, 401–406 (1995).
Perner, S. et al. TMPRSS2:ERG fusion–associated deletions provide insight into the heterogeneity of prostate cancer. Cancer Res. 66, 8337–8341 (2006).
Tomlins, S.A. et al. Distinct classes of chromosomal rearrangements create oncogenic ETS gene fusions in prostate cancer. Nature 448, 595–599 (2007).
Tomlins, S.A. et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310, 644–648 (2005).
Taylor, B.S. et al. Integrative genomic profiling of human prostate cancer. Cancer Cell 18, 11–22 (2010).
Kumar, A. et al. Exome sequencing identifies a spectrum of mutation frequencies in advanced and lethal prostate cancers. Proc. Natl. Acad. Sci. U.S.A. 108, 17087–17092 (2011).
Berger, M.F. et al. The genomic complexity of primary human prostate cancer. Nature 470, 214–220 (2011).
Nagai, Y. et al. Identification of a novel nuclear speckle-type protein, SPOP. FEBS Lett. 418, 23–26 (1997).
Zhuang, M. et al. Structures of SPOP-substrate complexes: insights into molecular architectures of BTB-Cul3 ubiquitin ligases. Mol. Cell 36, 39–50 (2009).
Kan, Z. et al. Diverse somatic mutation patterns and pathway alterations in human cancers. Nature 466, 869–873 (2010).
Majumder, P.K. et al. A prostatic intraepithelial neoplasia–dependent p27Kip1 checkpoint induces senescence and inhibits cell proliferation and cancer progression. Cancer Cell 14, 146–155 (2008).
Kibel, A.S. et al. CDKN1A and CDKN1B polymorphisms and risk of advanced prostate carcinoma. Cancer Res. 63, 2033–2036 (2003).
Gao, N. et al. Forkhead box A1 regulates prostate ductal morphogenesis and promotes epithelial cell maturation. Development 132, 3431–3443 (2005).
Zhang, C. et al. Definition of a FoxA1 cistrome that is crucial for G1 to S-phase cell-cycle transit in castration-resistant prostate cancer. Cancer Res. 71, 6738–6748 (2011).
Gao, N. et al. The role of hepatocyte nuclear factor–3α (Forkhead Box A1) and androgen receptor in transcriptional regulation of prostatic genes. Mol. Endocrinol. 17, 1484–1507 (2003).
Williamson, E.A. et al. BRCA1 and FOXA1 proteins coregulate the expression of the cell cycle–dependent kinase inhibitor p27Kip1. Oncogene 25, 1391–1399 (2006).
Clark, K.L., Halay, E.D., Lai, E. & Burley, S.K. Co-crystal structure of the HNF-3/fork head DNA-recognition motif resembles histone H5. Nature 364, 412–420 (1993).
Zhou, R. et al. SOX9 interacts with a component of the human thyroid hormone receptor–associated protein complex. Nucleic Acids Res. 30, 3245–3252 (2002).
Wang, Q., Sharma, D., Ren, Y. & Fondell, J.D. A coregulatory role for the TRAP-mediator complex in androgen receptor–mediated gene expression. J. Biol. Chem. 277, 42852–42858 (2002).
Donner, A.J., Szostek, S., Hoover, J.M. & Espinosa, J.M. CDK8 is a stimulus-specific positive coregulator of p53 target genes. Mol. Cell 27, 121–133 (2007).
Mäkinen, N. et al. MED12, the mediator complex subunit 12 gene, is mutated at high frequency in uterine leiomyomas. Science 334, 252–255 (2011).
Liu, J. et al. Analysis of Drosophila segmentation network identifies a JNK pathway factor overexpressed in kidney cancer. Science 323, 1218–1222 (2009).
Wang, C., Pan, Y. & Wang, B. Suppressor of fused and Spop regulate the stability, processing and function of Gli2 and Gli3 full-length activators but not their repressors. Development 137, 2001–2009 (2010).
Li, C. et al. Tumor-suppressor role for the SPOP ubiquitin ligase in signal-dependent proteolysis of the oncogenic co-activator SRC-3/AIB1. Oncogene 30, 4350–4364 (2011).
Mosquera, J.M. et al. Prevalence of TMPRSS2-ERG fusion prostate cancer among men undergoing prostate biopsy in the United States. Clin. Cancer Res. 15, 4706–4711 (2009).
Demichelis, F. et al. Distinct genomic aberrations associated with ERG rearranged prostate cancer. Genes Chromosom. Cancer 48, 366–380 (2009).
Lapointe, J. et al. Genomic profiling reveals alternative genetic pathways of prostate tumorigenesis. Cancer Res. 67, 8504–8510 (2007).
Yang, J.Y. & Hung, M.C. A new fork for clinical application: targeting forkhead transcription factors in cancer. Clin. Cancer Res. 15, 752–757 (2009).
Mandelbaum, J. et al. BLIMP1 is a tumor suppressor gene frequently disrupted in activated B cell–like diffuse large B cell lymphoma. Cancer Cell 18, 568–579 (2010).
Fisher, S. et al. A scalable, fully automated process for construction of sequence-ready human exome targeted capture libraries. Genome Biol. 12, R1 (2011).
Stransky, N. et al. The mutational landscape of head and neck squamous cell carcinoma. Science 333, 1157–1160 (2011).
Chapman, M.A. et al. Initial genome sequencing and analysis of multiple myeloma. Nature 471, 467–472 (2011).
Reich, M. et al. GenePattern 2.0. Nat. Genet. 38, 500–501 (2006).
Cibulskis, K. et al. ContEst: estimating cross-contamination of human samples in next-generation sequencing data. Bioinformatics 27, 2601–2602 (2011).
The Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature 474, 609–615 (2011).
Robinson, J.T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Fujita, P.A. et al. The UCSC Genome Browser database: update 2011. Nucleic Acids Res. 39, D876–D882 (2011).
Kozomara, A. & Griffiths-Jones, S. miRBase: integrating microRNA annotation and deep-sequencing data. Nucleic Acids Res. 39, D152–D157 (2011).
Sherry, S.T. et al. dbSNP: the NCBI database of genetic variation. Nucleic Acids Res. 29, 308–311 (2001).
Griffith, O.L. et al. ORegAnno: an open-access community-driven resource for regulatory annotation. Nucleic Acids Res. 36, D107–D113 (2008).
UniProt Consortium. Ongoing and future developments at the Universal Protein Resource. Nucleic Acids Res. 39, D214–D219 (2011).
Forbes, S.A. et al. COSMIC: mining complete cancer genomes in the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 39, D945–D950 (2011).
Carter, S.L. et al. Absolute quantification of somatic DNA alterations in human cancer. Nat. Biotechnol. published online, doi:10.1038/nbt.2203 (29 April 2012).
Pflueger, D. et al. Discovery of non-ETS gene fusions in human prostate cancer using next-generation RNA sequencing. Genome Res. 21, 56–67 (2011).
Espina, V. et al. Laser-capture microdissection. Nat. Protoc. 1, 586–603 (2006).
Svensson, M.A. et al. Testing mutual exclusivity of ETS rearranged prostate cancer. Lab. Invest. 91, 404–412 (2011).
Park, K. et al. Antibody-based detection of ERG rearrangement–positive prostate cancer. Neoplasia 12, 590–598 (2010).
Acknowledgements
We are grateful for the assistance of members of the Broad Institute Biological Samples Platform, Genetic Analysis Platform and Genome Sequencing Platform. We thank S. Banerjee for computational assistance, R. Kim and R. Leung for their critical contributions to the Weill Cornell Prostate Cancer Tumor Bank, P. Schraml, S. Dettwiler and M. Storz for assistance with the University Hospital Zurich cohort and biobank and the members of the University of Washington Rapid Autopsy Program. We thank the University of Michigan Prostate Cancer Special Program of Research Excellence (SPORE) (K. Pienta) for sample contribution to this study. We are also grateful to the individuals with prostate cancer and the families who contributed to these studies. This work was supported by the US National Human Genome Research Institute (NHGRI) Large Scale Sequencing Program (U54 HG003067 to the Broad Institute, E.S.L.), the Kohlberg Foundation (L.A.G.), the Starr Cancer Consortium (M.A.R., F.D. and L.A.G), the Prostate Cancer Foundation (M.A.R.), US Department of Defense Synergy Awards (PC101020 to F.D., L.A.G. and M.A.R. and PC093372 to T.W. and P.S.N.), the Dana-Farber/Harvard Cancer Center Prostate Cancer SPORE (US National Institutes of Health (NIH) P50 CA090381), a New Investigator Award (PC094516 to F.D.), the US National Cancer Institute, Early Detection Research Network (U01CA111275 and NCI EDRN to F.D. and M.A.R.), the US National Cancer Institute (R01 CA125612 to F.D. and M.A.R.), the Pacific Northwest Prostate Cancer SPORE (P50CA097186 to C.M. and P.S.N.), the Swiss Science Foundation (PASMP3_134379/1 to J.-P.T.) and a US NIH Director's New Innovator Award (DP2OD002750 to L.A.G.). S.C.B. is supported by a Medical Scientist Training Program (MSTP) grant from the US NIH. C.E.B. is supported by a Prostate Cancer Foundation Young Investigator Award.
Author information
Authors and Affiliations
Contributions
S.C.B., M.S.L., P.S., W.M.H., E.V.A., N.S., K.C., A.S., S.L.C., G.S., D.V. and A.H.R. performed computational analyses. M.B., J.-P.T., T.A.W., S.-S.C., K.S., G.B., T.Y.M., K.P. and T.V. designed and performed experiments. E.N., D.A., R.C.O., C.G., W.W., M.C.R., K.A. and S.B.G. processed samples and supervised exome sequencing. J.M.M., K.P., N.K., A.K.T., N.R., P.J.W., H.M., C.M. and P.S.N. coordinated sample acquisition, processing, pathologic review and analysis. C.E.B., S.C.B., F.D., P.W.K., T.R.G., M.M., E.S.L., G.G., M.A.R. and L.A.G. designed the study. C.E.B., S.C.B., F.D., G.G., M.A.R. and L.A.G. analyzed the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Note, Supplementary Figures 1–18 and Supplementary Tables 1, 4–8 and 10–12 (PDF 8357 kb)
Supplementary Tables
Supplementary Tables 2, 3 and 9 (XLSX 6486 kb)
Rights and permissions
About this article
Cite this article
Barbieri, C., Baca, S., Lawrence, M. et al. Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nat Genet 44, 685–689 (2012). https://doi.org/10.1038/ng.2279
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2279
This article is cited by
-
UBE2J1 is the E2 ubiquitin-conjugating enzyme regulating androgen receptor degradation and antiandrogen resistance
Oncogene (2024)
-
Hepatocyte-specific HDAC3 ablation promotes hepatocellular carcinoma in females by suppressing Foxa1/2
BMC Cancer (2023)
-
Genomic alterations related to HPV infection status in a cohort of Chinese prostate cancer patients
European Journal of Medical Research (2023)
-
Tumor immune contexture predicts recurrence after prostatectomy and efficacy of androgen deprivation and immunotherapy in prostate cancer
Journal of Translational Medicine (2023)
-
Glucocorticoid receptor-induced non-muscle caldesmon regulates metastasis in castration-resistant prostate cancer
Oncogenesis (2023)