Abstract
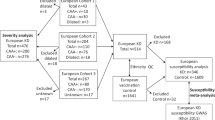
To find new candidate loci predisposing individuals to Kawasaki disease, an acute vasculitis that affects children, we conducted a genome-wide association study in 622 individuals with Kawasaki disease (cases) and 1,107 controls in a Han Chinese population residing in Taiwan, with replication in an independent Han Chinese sample of 261 cases and 550 controls. We report two new loci, one at BLK (encoding B-lymphoid tyrosine kinase) and one at CD40, that are associated with Kawasaki disease at genome-wide significance (P < 5 × 10−8). Our findings may lead to a better understanding of the role of immune activation and inflammation in Kawasaki disease pathogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kato, H., Koike, S., Yamamoto, M., Ito, Y. & Yano, E. Coronary aneurysms in infants and young children with acute febrile mucocutaneous lymph node syndrome. J. Pediatr. 86, 892–898 (1975).
Kato, H. et al. Long-term consequences of Kawasaki disease. A 10- to 21-year follow-up study of 594 patients. Circulation 94, 1379–1385 (1996).
Yanagawa, H. et al. Results of the nationwide epidemiologic survey of Kawasaki disease in 1995 and 1996 in Japan. Pediatrics 102, E65 (1998).
Yanagawa, H. et al. Incidence survey of Kawasaki disease in 1997 and 1998 in Japan. Pediatrics 107, E33 (2001).
Park, Y.W. et al. Epidemiological features of Kawasaki disease in Korea, 2006–2008. Pediatr. Int. 53, 36–39 (2011).
Chang, L.Y. et al. Epidemiologic features of Kawasaki disease in Taiwan, 1996–2002. Pediatrics 114, e678–e682 (2004).
Huang, W.C. et al. Epidemiologic features of Kawasaki disease in Taiwan, 2003–2006. Pediatrics 123, e401–e405 (2009).
Matsubara, T., Furukawa, S. & Yabuta, K. Serum levels of tumor necrosis factor, interleukin 2 receptor, and interferon-γ in Kawasaki disease involved coronary-artery lesions. Clin. Immunol. Immunopathol. 56, 29–36 (1990).
Lin, C.Y., Lin, C.C., Hwang, B. & Chiang, B. Serial changes of serum interleukin-6, interleukin-8, and tumor necrosis factor α among patients with Kawasaki disease. J. Pediatr. 121, 924–926 (1992).
Onouchi, Y. Molecular genetics of Kawasaki disease. Pediatr. Res. 65, 46R–54R (2009).
Onouchi, Y. et al. A genomewide linkage analysis of Kawasaki disease: evidence for linkage to chromosome 12. J. Hum. Genet. 52, 179–190 (2007).
Onouchi, Y. et al. ITPKC functional polymorphism associated with Kawasaki disease susceptibility and formation of coronary artery aneurysms. Nat. Genet. 40, 35–42 (2008).
Burgner, D. et al. A genome-wide association study identifies novel and functionally related susceptibility loci for Kawasaki disease. PLoS Genet. 5, e1000319 (2009).
Kim, J.J. et al. A genome-wide association analysis reveals 1p31 and 2p13.3 as susceptibility loci for Kawasaki disease. Hum. Genet. 129, 487–495 (2011).
Tsai, F.J. et al. Identification of novel susceptibility loci for Kawasaki disease in a Han Chinese population by a genome-wide association study. PLoS ONE 6, e16853 (2011).
Khor, C.C. et al. Genome-wide association study identifies FCGR2A as a susceptibility locus for Kawasaki disease. Nat. Genet. 43, 1241–1246 (2011).
Dymecki, S.M., Zwollo, P., Zeller, K., Kuhajda, F.P. & Desiderio, S.V. Structure and developmental regulation of the B-lymphoid tyrosine kinase gene blk. J. Biol. Chem. 267, 4815–4823 (1992).
Wasserman, R., Li, Y.S. & Hardy, R.R. Differential expression of the blk and ret tyrosine kinases during B lineage development is dependent on Ig rearrangement. J. Immunol. 155, 644–651 (1995).
Nemazee, D. & Weigert, M. Revising B cell receptors. J. Exp. Med. 191, 1813–1817 (2000).
Hom, G. et al. Association of systemic lupus erythematosus with C8orf13-BLK and ITGAM-ITGAX. N. Engl. J. Med. 358, 900–909 (2008).
Gregersen, P.K. et al. REL, encoding a member of the NF-κB family of transcription factors, is a newly defined risk locus for rheumatoid arthritis. Nat. Genet. 41, 820–823 (2009).
Peters, A.L., Stunz, L.L. & Bishop, G.A. CD40 and autoimmunity: the dark side of a great activator. Semin. Immunol. 21, 293–300 (2009).
Akiyama, T. et al. The tumor necrosis factor family receptors RANK and CD40 cooperatively establish the thymic medullary microenvironment and self-tolerance. Immunity 29, 423–437 (2008).
Iezzi, G. et al. CD40-CD40L cross-talk integrates strong antigenic signals and microbial stimuli to induce development of IL-17–producing CD4+ T cells. Proc. Natl. Acad. Sci. USA 106, 876–881 (2009).
Jacobson, E.M. et al. A CD40 Kozak sequence polymorphism and susceptibility to antibody-mediated autoimmune conditions: the role of CD40 tissue-specific expression. Genes Immun. 8, 205–214 (2007).
Wagner, D.H. Jr. et al. Expression of CD40 identifies a unique pathogenic T cell population in type 1 diabetes. Proc. Natl. Acad. Sci. USA 99, 3782–3787 (2002).
Gerritse, K. et al. CD40–CD40 ligand interactions in experimental allergic encephalomyelitis and multiple sclerosis. Proc. Natl. Acad. Sci. USA 93, 2499–2504 (1996).
Ohta, Y. & Hamada, Y. In situ expression of CD40 and CD40 ligand in psoriasis. Dermatology 209, 21–28 (2004).
Danese, S. et al. TNF-α blockade down-regulates the CD40/CD40L pathway in the mucosal microcirculation: a novel anti-inflammatory mechanism of infliximab in Crohn's disease. J. Immunol. 176, 2617–2624 (2006).
Brennan, F.M. & McInnes, I.B. Evidence that cytokines play a role in rheumatoid arthritis. J. Clin. Invest. 118, 3537–3545 (2008).
Pyrovolaki, K. et al. Increased expression of CD40 on bone marrow CD34+ hematopoietic progenitor cells in patients with systemic lupus erythematosus: contribution to Fas-mediated apoptosis. Arthritis Rheum. 60, 543–552 (2009).
Tomer, Y., Concepcion, E. & Greenberg, D.A. A C/T single-nucleotide polymorphism in the region of the CD40 gene is associated with Graves' disease. Thyroid 12, 1129–1135 (2002).
Ban, Y., Tozaki, T., Taniyama, M. & Tomita, M. Association of a C/T single-nucleotide polymorphism in the 5′ untranslated region of the CD40 gene with Graves' disease in Japanese. Thyroid 16, 443–446 (2006).
Kurylowicz, A. et al. Association of CD40 gene polymorphism (C-1T) with susceptibility and phenotype of Graves' disease. Thyroid 15, 1119–1124 (2005).
Raychaudhuri, S. et al. Common variants at CD40 and other loci confer risk of rheumatoid arthritis. Nat. Genet. 40, 1216–1223 (2008).
Blanco-Kelly, F. et al. CD40: novel association with Crohn's disease and replication in multiple sclerosis susceptibility. PLoS ONE 5, e11520 (2010).
Law, C.L. & Grewal, I.S. Therapeutic interventions targeting CD40L (CD154) and CD40: the opportunities and challenges. Adv. Exp. Med. Biol. 647, 8–36 (2009).
Hassan, G.S., Merhi, Y. & Mourad, W.M. CD154 and its receptors in inflammatory vascular pathologies. Trends Immunol. 30, 165–172 (2009).
Wang, C.L. et al. Expression of CD40 ligand on CD4+ T-cells and platelets correlated to the coronary artery lesion and disease progress in Kawasaki disease. Pediatrics 111, E140–E147 (2003).
Newburger, J.W. et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Pediatrics 114, 1708–1733 (2004).
Kim, S. & Dedeoglu, F. Update on pediatric vasculitis. Curr. Opin. Pediatr. 17, 695–702 (2005).
Pan, W.H. et al. Han Chinese cell and genome bank in Taiwan: purpose, design and ethical considerations. Hum. Hered. 61, 27–30 (2006).
Price, A.L. et al. Principal components analysis corrects for stratification in genome-wide association studies. Nat. Genet. 38, 904–909 (2006).
Higgins, J.P. & Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 21, 1539–1558 (2002).
Acknowledgements
We thank all affected individuals and their families who devoted their time and effort to participate in this study. We gratefully acknowledge the members of the Translational Resource Center (TRC) (NSC100-2325-B-001-023) for Genomic Medicine and the National Center for Genome Medicine (NCGM) (NSC100-2319-B-001-001) at Academia Sinica for their support in subject recruitment, genotyping and statistical analysis. We especially thank H. Lue for his inspirational discussion. This study was supported by the Academia Sinica Genomic Medicine Multicenter Study, Taiwan (40-05-GMM). The funders had no role in study design, data collection or analysis, the decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Y.-T.C., F.-J.T. and J.-Y.W. are the principal investigators who conceived and obtained funding for this project. Y.-C.L., C.-H.C. and J.-Y.W. organized and supervised the GWAS and replication genotyping pipeline and devised the overall analysis plan. Y.-C.L. wrote the first draft of the manuscript with input from C.-H.C. and J.-Y.W. Y.-C.L., L.-C.C. and C.-H.C. analyzed the data. C.-D.L., J.-S.C., L.-Y.C., L.-M.H., M.-R.C., H.-C.K., H.C., F.-Y.H., M.-L.L., Y.-C.H., B.H., N.-C.C., K.-P.H., P.-C.L., Y.-M.L., Y.-J.C. and the Taiwan Pediatric ID Alliance coordinated and contributed subject and database phenotype collections.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Note, Supplementary Figures 1–5 and Supplementary Tables 1–9 (PDF 1847 kb)
Rights and permissions
About this article
Cite this article
Lee, YC., Kuo, HC., Chang, JS. et al. Two new susceptibility loci for Kawasaki disease identified through genome-wide association analysis. Nat Genet 44, 522–525 (2012). https://doi.org/10.1038/ng.2227
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2227
This article is cited by
-
Whole-exome sequencing analysis identifies novel variants associated with Kawasaki disease susceptibility
Pediatric Rheumatology (2023)
-
Viral load of Torquetenovirus correlates with Sano’s score and levels of total bilirubin and aspartate aminotransferase in Kawasaki disease
Scientific Reports (2023)
-
Preterm birth and Kawasaki disease: a nationwide Japanese population-based study
Pediatric Research (2022)
-
IL-1 and autoinflammatory disease: biology, pathogenesis and therapeutic targeting
Nature Reviews Rheumatology (2022)
-
Increasing risk of gestational diabetes and preeclampsia associated with long-term exposure effects of air pollution
Air Quality, Atmosphere & Health (2022)