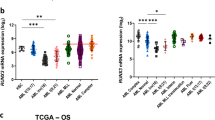
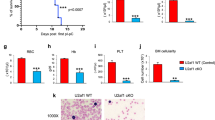
Abstract
Both PU.1 (also called SFPI1), an Ets-family transcription factor, and AML1 (also called RUNX1), a DNA-binding subunit of the CBF transcription factor family, are crucial for the generation of all hematopoietic lineages, and both act as tumor suppressors in leukemia. An upstream regulatory element (URE) of PU.1 has both enhancer and repressor activity and tightly regulates PU.1 expression. Here we show that AML1 binds to functionally important sites within the PU.1 upstream regulatory element and regulates PU.1 expression at both embryonic and adult stages of development. Analysis of mice carrying conditional AML1 knockout alleles and knock-in mice carrying mutations in all three AML1 sites of the URE proximal region demonstrated that AML1 regulates PU.1 both positively and negatively in a lineage dependent manner. Dysregulation of PU.1 expression contributed to each of the phenotypes observed in these mice, and restoration of proper PU.1 expression rescued or partially rescued each phenotype. Thus, our data demonstrate that PU.1 is a major downstream target gene of AML1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
23 January 2008
In the version of this article initially published, the affiliation for Stephen D Nimer was incorrect. Dr. Nimer is affiliated with the Division of Hematologic Oncology, Memorial Sloan-Kettering Cancer Center, New York, and not with Kyoto Prefectural University of Medicine. The error has been corrected in the PDF version of the article.
References
Canon, J. & Banerjee, U. In vivo analysis of a developmental circuit for direct transcriptional activation and repression in the same cell by a Runx protein. Genes Dev. 17, 838–843 (2003).
Flores, G.V. et al. Combinatorial signaling in the specification of unique cell fates. Cell 103, 75–85 (2000).
Flores, G.V., Daga, A., Kalhor, H.R. & Banerjee, U. Lozenge is expressed in pluripotent precursor cells and patterns multiple cell types in the Drosophila eye through the control of cell-specific transcription factors. Development 125, 3681–3687 (1998).
Tenen, D.G. Disruption of differentiation in human cancer: AML shows the way. Nat. Rev. Cancer 3, 89–101 (2003).
Koschmieder, S., Rosenbauer, F., Steidl, U., Owens, B.M. & Tenen, D.G. Role of transcription factors C/EBPalpha and PU.1 in normal hematopoiesis and leukemia. Int. J. Hematol. 81, 368–377 (2005).
Zhu, J. & Emerson, S.G. Hematopoietic cytokines, transcription factors and lineage commitment. Oncogene 21, 3295–3313 (2002).
Osato, M. Point mutations in the RUNX1/AML1 gene: another actor in RUNX leukemia. Oncogene 23, 4284–4296 (2004).
Song, W.J. et al. Haploinsufficiency of CBFA2 causes familial thrombocytopenia with propensity to develop acute myelogenous leukaemia. Nat. Genet. 23, 166–175 (1999).
Osato, M. et al. Biallelic and heterozygous point mutations in the runt domain of the AML1/PEBP2alphaB gene associated with myeloblastic leukemias. Blood 93, 1817–1824 (1999).
Harada, H. et al. High incidence of somatic mutations in the AML1/RUNX1 gene in myelodysplastic syndrome and low blast percentage myeloid leukemia with myelodysplasia. Blood 103, 2316–2324 (2004).
Okuda, T., van Deursen, J., Hiebert, S.W., Grosveld, G. & Downing, J.R. AML1, the target of multiple chromosomal translocations in human leukemia, is essential for normal fetal liver hematopoiesis. Cell 84, 321–330 (1996).
Wang, Q. et al. Disruption of the Cbfa2 gene causes necrosis and hemorrhaging in the central nervous system and blocks definitive hematopoiesis. Proc. Natl. Acad. Sci. USA 93, 3444–3449 (1996).
Lorsbach, R.B. et al. Role of RUNX1 in adult hematopoiesis: analysis of RUNX1-IRES-GFP knock-in mice reveals differential lineage expression. Blood 103, 2522–2529 (2004).
North, T.E., Stacy, T., Matheny, C.J., Speck, N.A. & de Bruijn, M.F. Runx1 is expressed in adult mouse hematopoietic stem cells and differentiating myeloid and lymphoid cells, but not in maturing erythroid cells. Stem Cells 22, 158–168 (2004).
Ichikawa, M. et al. AML-1 is required for megakaryocytic maturation and lymphocytic differentiation, but not for maintenance of hematopoietic stem cells in adult hematopoiesis. Nat. Med. 10, 299–304 (2004).
Growney, J.D. et al. Loss of Runx1 perturbs adult hematopoiesis and is associated with a myeloproliferative phenotype. Blood 106, 494–504 (2005).
Putz, G., Rosner, A., Nuesslein, I., Schmitz, N. & Buchholz, F. AML1 deletion in adult mice causes splenomegaly and lymphomas. Oncogene 25, 929–939 (2006).
Peterson, L.F. & Zhang, D.E. The 8;21 translocation in leukemogenesis. Oncogene 23, 4255–4262 (2004).
de Bruijn, M.F. & Speck, N.A. Core-binding factors in hematopoiesis and immune function. Oncogene 23, 4238–4248 (2004).
Bystrykh, L. et al. Uncovering regulatory pathways that affect hematopoietic stem cell function using 'genetical genomics'. Nat. Genet. 37, 225–232 (2005).
Canon, J. & Banerjee, U. Runt and Lozenge function in Drosophila development. Semin. Cell Dev. Biol. 11, 327–336 (2000).
Rosenbauer, F. et al. Lymphoid cell growth and transformation are suppressed by a key regulatory element of the gene encoding PU.1. Nat. Genet. 38, 27–37 (2006).
Rosenbauer, F. et al. Acute myeloid leukemia induced by graded reduction of a lineage-specific transcription factor, PU.1. Nat. Genet. 36, 624–630 (2004).
Nutt, S.L., Metcalf, D., D'Amico, A., Polli, M. & Wu, L. Dynamic regulation of PU.1 expression in multipotent hematopoietic progenitors. J. Exp. Med. 201, 221–231 (2005).
Suraweera, N. et al. Mutations of the PU.1 Ets domain are specifically associated with murine radiation-induced, but not human therapy-related, acute myeloid leukaemia. Oncogene 24, 3678–3683 (2005).
Walter, M.J. et al. Reduced PU.1 expression causes myeloid progenitor expansion and increased leukemia penetrance in mice expressing PML-RARalpha. Proc. Natl. Acad. Sci. USA 102, 12513–12518 (2005).
Du, Y., Spence, S.E., Jenkins, N.A. & Copeland, N.G. Cooperating cancer-gene identification through oncogenic-retrovirus-induced insertional mutagenesis. Blood 106, 2498–2505 (2005).
Cook, W.D. et al. PU.1 is a suppressor of myeloid leukemia, inactivated in mice by gene deletion and mutation of its DNA binding domain. Blood 104, 3437–3444 (2004).
Chen, H. et al. PU.1 (Spi-1) autoregulates its expression in myeloid cells. Oncogene 11, 1549–1560 (1995).
Li, Y. et al. Regulation of the PU.1 gene by distal elements. Blood 98, 2958–2965 (2001).
Okuno, Y. et al. Potential autoregulation of transcription factor PU.1 by an upstream regulatory element. Mol. Cell. Biol. 25, 2832–2845 (2005).
Okada, H. et al. AML1(−/−) embryos do not express certain hematopoiesis-related gene transcripts including those of the PU.1 gene. Oncogene 17, 2287–2293 (1998).
Higuchi, M. et al. Expression of a conditional AML1-ETO oncogene bypasses embryonic lethality and establishes a murine model of human t(8;21) acute myeloid leukemia. Cancer Cell 1, 63–74 (2002).
Chen, Z., HU, M. & Shivdasani, R.A. Expression analysis of primary mouse megakaryocyte differentiation and its application in identifying stage-specific molecular markers and a novel transcriptional target of NF-E2. Blood 109, 1451–1459 (2007).
DeKoter, R.P. & Singh, H. Regulation of B lymphocyte and macrophage development by graded expression of PU.1. Science 288, 1439–1441 (2000).
Zhang, D.E. et al. CCAAT enhancer-binding protein (C/EBP) and AML1 (CBF alpha2) synergistically activate the macrophage colony-stimulating factor receptor promoter. Mol. Cell. Biol. 16, 1231–1240 (1996).
Akashi, K., Traver, D., Miyamoto, T. & Weissman, I.L. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature 404, 193–197 (2000).
Miyamoto, T. et al. Myeloid or lymphoid promiscuity as a critical step in hematopoietic lineage commitment. Dev. Cell 3, 137–147 (2002).
Moreau-Gachelin, F., Tavitian, A. & Tambourin, P. Spi-1 is a putative oncogene in virally induced murine erythroleukaemias. Nature 331, 277–280 (1988).
Moreau-Gachelin, F. et al. Spi-1/PU.1 transgenic mice develop multistep erythroleukemias. Mol. Cell. Biol. 16, 2453–2463 (1996).
Anderson, M.K., Weiss, A.H., Hernandez-Hoyos, G., Dionne, C.J. & Rothenberg, E.V. Constitutive expression of PU.1 in fetal hematopoietic progenitors blocks T cell development at the pro-T cell stage. Immunity 16, 285–296 (2002).
Vangala, R.K. et al. The myeloid master regulator transcription factor PU.1 is inactivated by AML1-ETO in t(8;21) myeloid leukemia. Blood 101, 270–277 (2003).
Liu, Y. et al. The tetramer structure of the Nervy homology two domain, NHR2, is critical for AML1/ETO's activity. Cancer Cell 9, 249–260 (2006).
Huang, G. et al. Molecular basis for a dominant inactivation of RUNX1/AML1 by the leukemogenic inversion 16 chimera. Blood 103, 3200–3207 (2004).
Li, Z. et al. Developmental stage-selective effect of somatically mutated leukemogenic transcription factor GATA1. Nat. Genet. 37, 613–619 (2005).
Boyer, L.A. et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122, 947–956 (2005).
Iwasaki, H. et al. Distinctive and indispensable roles of PU.1 in maintenance of hematopoietic stem cells and their differentiation. Blood 106, 1590–1600 (2005).
Kuhn, R., Schwenk, F., Aguet, M. & Rajewsky, K. Inducible gene targeting in mice. Science 269, 1427–1429 (1995).
Villeval, J.L. et al. High thrombopoietin production by hematopoietic cells induces a fatal myeloproliferative syndrome in mice. Blood 90, 4369–4383 (1997).
Acknowledgements
We thank H. Singh for the MIGR1-PU.1 and MIGR1-PU.1-ER retroviral constructs, and M. Osato and members of the Tenen laboratory for critical discussions and suggestions. This work was supported by US National Institutes of Health grants CA41456 and CA66996 (to D.G.T.) and DK52208 (to S.D.N. and D.G.T.), Leukemia Lymphoma Society SCOR grants (to S.D.N. and D.G.T.) and Deutsche Forschungsgemeinschaft grant KO2155/1-1 (to S.K.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1; Supplementary Tables 1–2 (PDF 212 kb)
Rights and permissions
About this article
Cite this article
Huang, G., Zhang, P., Hirai, H. et al. PU.1 is a major downstream target of AML1 (RUNX1) in adult mouse hematopoiesis. Nat Genet 40, 51–60 (2008). https://doi.org/10.1038/ng.2007.7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2007.7
This article is cited by
-
A novel AML1-ETO/FTO positive feedback loop promotes leukemogenesis and Ara-C resistance via stabilizing IGFBP2 in t(8;21) acute myeloid leukemia
Experimental Hematology & Oncology (2024)
-
Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
Expression analysis of Carassius auratus-leukocyte-immune-type receptors (CaLITRs) during goldfish kidney macrophage development and in activated kidney leukocyte cultures
Immunogenetics (2023)
-
How transcription factors drive choice of the T cell fate
Nature Reviews Immunology (2021)
-
Targeting transcription factors in acute myeloid leukemia
International Journal of Hematology (2019)