Abstract
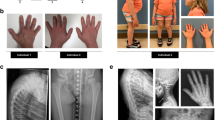
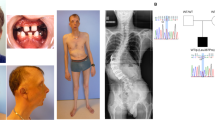
Geleophysic dysplasia is an autosomal recessive disorder characterized by short stature, brachydactyly, thick skin and cardiac valvular anomalies often responsible for an early death. Studying six geleophysic dysplasia families, we first mapped the underlying gene to chromosome 9q34.2 and identified five distinct nonsense and missense mutations in ADAMTSL2 (a disintegrin and metalloproteinase with thrombospondin repeats–like 2), which encodes a secreted glycoprotein of unknown function. Functional studies in HEK293 cells showed that ADAMTSL2 mutations lead to reduced secretion of the mutated proteins, possibly owing to the misfolding of ADAMTSL2. A yeast two-hybrid screen showed that ADAMTSL2 interacts with latent TGF-β–binding protein 1. In addition, we observed a significant increase in total and active TGF-β in the culture medium as well as nuclear localization of phosphorylated SMAD2 in fibroblasts from individuals with geleophysic dysplasia. These data suggest that ADAMTSL2 mutations may lead to a dysregulation of TGF-β signaling and may be the underlying mechanism of geleophysic dysplasia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Spranger, J.W., Gilbert, E.F., Tuffli, G.A., Rossiter, F.P. & Opitz, J.M. Geleophysic dwarfism–a “focal” mucopolysaccharidosis? Lancet 2, 97–98 (1971).
Pontz, B.F. et al. Clinical and ultrastructural findings in three patients with geleophysic dysplasia. Am. J. Med. Genet. 63, 50–54 (1996).
Shohat, M. et al. Geleophysic dysplasia: a storage disorder affecting the skin, bone, liver, heart, and trachea. J. Pediatr. 117, 227–232 (1990).
Apte, S.S. A disintegrin-like and metalloprotease (reprolysin type) with thrombospondin type 1 motifs: the ADAMTS family. Int. J. Biochem. Cell Biol. 36, 981–985 (2004).
Hirohata, S. et al. Punctin, a novel ADAMTS-like molecule, ADAMTSL-1, in extracellular matrix. J. Biol. Chem. 277, 12182–12189 (2002).
Hall, N.G., Klenotic, P., Anand-Apte, B. & Apte, S.S. ADAMTSL-3/punctin-2, a novel glycoprotein in extracellular matrix related to the ADAMTS family of metalloproteases. Matrix Biol. 22, 501–510 (2003).
Koo, B.H. et al. ADAMTS-like 2 (ADAMTSL2) is a secreted glycoprotein that is widely expressed during mouse embryogenesis and is regulated during skeletal myogenesis. Matrix Biol. 26, 431–441 (2007).
Annes, J.P., Munger, J.S. & Rifkin, D.B. Making sense of latent TGFβ activation. J. Cell Sci. 116, 217–224 (2003).
Sinha, S., Nevett, C., Shuttleworth, C.A. & Kielty, C.M. Cellular and extracellular biology of the latent transforming growth factor-β binding proteins. Matrix Biol. 17, 529–545 (1998).
Isogai, Z. et al. Latent transforming growth factor β-binding protein 1 interacts with fibrillin and is a microfibril-associated protein. J. Biol. Chem. 278, 2750–2757 (2003).
ten Dijke, P. & Arthur, H.M. Extracellular control of TGFβ signalling in vascular development and disease. Nat. Rev. Mol. Cell Biol. 8, 857–869 (2007).
Ge, G. & Greenspan, D.S. BMP1 controls TGFβ1 activation via cleavage of latent TGFβ-binding protein. J. Cell Biol. 175, 111–120 (2006).
Collod-Beroud, G. & Boileau, C. Marfan syndrome in the third millennium. Eur. J. Hum. Genet. 10, 673–681 (2002).
Loeys, B.L. et al. A syndrome of altered cardiovascular, craniofacial, neurocognitive and skeletal development caused by mutations in TGFBR1 or TGFBR2. Nat. Genet. 37, 275–281 (2005).
Kinoshita, A. et al. Domain-specific mutations in TGFB1 result in Camurati-Engelmann disease. Nat. Genet. 26, 19–20 (2000).
Neptune, E.R. et al. Dysregulation of TGF-β activation contributes to pathogenesis in Marfan syndrome. Nat. Genet. 33, 407–411 (2003).
Jones, K.B. et al. Toward an understanding of dural ectasia: a light microscopy study in a murine model of Marfan syndrome. Spine 30, 291–293 (2005).
Gordon, K.J. & Blobe, G.C. Role of transforming growth factor-β superfamily signaling pathways in human disease. Biochim. Biophys. Acta 1782, 197–228 (2008).
Wang, L.W. et al. O-fucosylation of thrombospondin type 1 repeats in ADAMTS-like-1/punctin-1 regulates secretion: implications for the ADAMTS superfamily. J. Biol. Chem. 282, 17024–17031 (2007).
Abbaszade, I. et al. Cloning and characterization of ADAMTS11, an aggrecanase from the ADAMTS family. J. Biol. Chem. 274, 23443–23450 (1999).
Acknowledgements
We thank the Kazusa DNA Research Institute for providing the KIAA0605 cDNA. We thank J. Martinovic for her help. We also thank T. Arai and M. Papouin. The work presented here was supported by French National Research Agency (ANR) award R06215KS (to V.C.-D.), the Medical Research Foundation (FRM, to C.L.G.), US National Institutes of Health (NIH) award AR53890 (to S.S.A.), NIH award GM71679 (to D.S.G.) and NIH award HD22657 (to D.K.).
Author information
Authors and Affiliations
Contributions
C.L.G. designed the experiments; performed in situ hybridization, protein blot analysis (ADAMTSL2 and SMAD2), TGF-β assays and immunocytochemistry and wrote the manuscript. F.M.-P. and N.D. performed the sequence analysis. L.W.W. performed coimmunoprecipitation studies. C.P., Y.J.C., F.B., E.F., D.K., D.B. and M.L.M. provided clinical data; C.P.-S. performed electron microscopy analysis. G.G. and D.S.G. cloned expression LTBP-1 constructs. A.M. and S.S.A. wrote the manuscript. V.C.-D. provided clinical data, designed the experiments, oversaw all aspects of the research and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 and Supplementary Table 1 (PDF 537 kb)
Rights and permissions
About this article
Cite this article
Le Goff, C., Morice-Picard, F., Dagoneau, N. et al. ADAMTSL2 mutations in geleophysic dysplasia demonstrate a role for ADAMTS-like proteins in TGF-β bioavailability regulation. Nat Genet 40, 1119–1123 (2008). https://doi.org/10.1038/ng.199
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.199
This article is cited by
-
The roles and regulatory mechanisms of TGF-β and BMP signaling in bone and cartilage development, homeostasis and disease
Cell Research (2024)
-
Emerging roles for the ADAMTS-like family of matricellular proteins in cardiovascular disease through regulation of the extracellular microenvironment
Molecular Biology Reports (2024)
-
A nonsense mutation in mouse Adamtsl2 causes uterine hypoplasia and an irregular estrous cycle
Mammalian Genome (2023)
-
Distinct whole-blood transcriptome profile of children with metabolic healthy overweight/obesity compared to metabolic unhealthy overweight/obesity
Pediatric Research (2021)
-
A case of Myhre syndrome mimicking juvenile scleroderma
Pediatric Rheumatology (2020)