Abstract
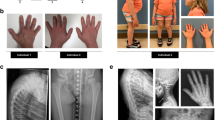
Digital clubbing, recognized by Hippocrates in the fifth century BC, is the outward hallmark of pulmonary hypertrophic osteoarthropathy, a clinical constellation that develops secondary to various acquired diseases, especially intrathoracic neoplasm1. The pathogenesis of clubbing and hypertrophic osteoarthropathy has hitherto been poorly understood, but a clinically indistinguishable primary (idiopathic) form of hypertrophic osteoarthropathy (PHO) is recognized2,3. This familial disorder can cause diagnostic confusion, as well as significant disability. By autozygosity methods, we mapped PHO to chromosome 4q33–q34 and identified mutations in HPGD, encoding 15-hydroxyprostaglandin dehydrogenase, the main enzyme of prostaglandin degradation. Homozygous individuals develop PHO secondary to chronically elevated prostaglandin E2 levels. Heterozygous relatives also show milder biochemical and clinical manifestations. These findings not only suggest therapies for PHO, but also imply that clubbing secondary to other pathologies may be prostaglandin mediated. Testing for HPGD mutations and biochemical testing for HPGD deficiency in patients with unexplained clubbing might help to obviate extensive searches for occult pathology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
26 June 2008
In the version of this article initially published, Figure 3a—d and Supplementary Figure 2a—c show 16-OH PGE2, not 15-OH PGE2. The errors have been corrected in the HTML and PDF versions of the article.
References
Coury, C. Hippocratic fingers and hypertrophic osteoarthropathy. A study of 350 cases. Br. J. Dis. Chest 54, 202–209 (1960).
Castori, M. et al. Pachydermoperiostosis: an update. Clin. Genet. 68, 477–486 (2005).
Touraine, A., Solente, G. & Gole, L. Un syndrome osteodermopathique: la pachydermie plicaturee avec pachyperiostose des extremites. Presse Med. 43, 1820–1824 (1935).
West, S. Two cases of clubbing of the fingers developing within a fortnight and four weeks, respectively, with remarks. Trans. Clin. Soc. London 30, 60–64 (1897).
Currarino, G., Tierney, R.C., Giesel, R.G. & Weihl, C. Familial idiopathic osteoarthropathy. Am. J. Roentgenol. Radium Ther. Nucl. Med. 85, 633–644 (1961).
Dabir, T. et al. Cranio-osteoarthropathy in sibs. Clin. Dysmorphol. 16, 197–201 (2007).
Forslund, T., Nyberg, A., Janne, S. & Viljanen, B. Hypertrophic osteoarthropathy and familial digital clubbing in a patient with surgical closed ductus arteriosus Botalli. Scand. J. Rheumatol. 16, 371–373 (1987).
Martinez-Lavin, M., Pineda, C., Navarro, C., Buendia, A. & Zabal, C. Primary hypertrophic osteoarthropathy: another heritable disorder associated with patent ductus arteriosus. Pediatr. Cardiol. 14, 181–182 (1993).
Joseph, B. & Chacko, V. Acro-osteolysis associated with hypertrophic pulmonary osteoarthropathy and pachydermoperiostosis. Radiology 154, 343–344 (1985).
Tai, H.-H., Ensor, C.M., Tong, M., Zhou, H. & Yan, F. Prostaglandin catabolizing enzymes. Prostaglandins Other Lipid Mediat. 68–69, 483–493 (2002).
Murphey, L.J. et al. Quantification of the major urinary metabolite of PGE2 by a liquid chromatographic/mass spectrometric assay: determination of cyclooxygenase-specific PGE2 synthesis in healthy humans and those with lung cancer. Anal. Biochem. 334, 266–275 (2004).
Chang, D.G. & Tai, H.-H. Prostaglandin 9-ketoreductase/type II 15-hydroxyprostaglandin dehydrogenase is not a prostaglandin specific enzyme. Biochem. Biophys. Res. Commun. 101, 898–904 (1981).
Tokumoto, H., Watanabe, K., Fukushima, D., Shimizu, T. & Hayaishi, O. An NADP-linked 15-hydroxyprostaglandin dehydrogenase specific for prostaglandin D2 from swine brain. J. Biol. Chem. 257, 13576–13580 (1982).
Pilbeam, C. & Raisz, L. Prostaglandins, leukotrienes and bone. in The Eicosanoids (ed. Curtis-Prior, P.) 289–298 (Wiley, Chichester, UK, 2004).
Martinez-Lavin, M. Digital clubbing and hypertrophic osteoarthropathy: a unifying hypothesis. J. Rheumatol. 14, 6–8 (1987).
Dickinson, C.J. & Martin, J.F. Megakaryocytes and platelet clumps as the cause of finger clubbing. Lancet 2, 1434–1435 (1987).
Ferreira, S.H. & Vane, J.R. Prostaglandins: their disappearance from and release into the circulation. Nature 216, 868–873 (1967).
Currie, A.E. & Gallagher, P.J. The pathology of clubbing: vascular changes in the nail bed. Br. J. Dis. Chest 82, 382–385 (1988).
Coggins, K.G. et al. Metabolism of PGE2 by prostaglandin dehydrogenase is essential for remodeling the ductus arteriosus. Nat. Med. 8, 91–92 (2002).
Ueda, K. et al. Cortical hyperostosis following long-term administration of prostaglandin E1 in infants with cyanotic congenital heart disease. J. Pediatr. 97, 834–836 (1980).
Cattral, M.S., Altraif, I., Greig, P.D., Blendis, L. & Levy, G.A. Toxic effects of intravenous and oral prostaglandin E therapy in patients with liver disease. Am. J. Med. 97, 369–373 (1994).
Backlund, M.G. et al. 15-Hydroxyprostaglandin dehydrogenase is down-regulated in colorectal cancer. J. Biol. Chem. 280, 3217–3223 (2005).
Ding, Y., Tong, M., Liu, S., Moscow, J.A. & Tai, H.H. NAD+-linked 15-hydroxyprostaglandin dehydrogenase (15-PGDH) behaves as a tumor suppressor in lung cancer. Carcinogenesis 26, 65–72 (2005).
Myung, S.J. et al. 15-Hydroxyprostaglandin dehydrogenase is an in vivo suppressor of colon tumorigenesis. Proc. Natl. Acad. Sci. USA 103, 12098–12102 (2006).
Lemen, R.J. et al. Relationships among digital clubbing, disease severity, and serum prostaglandins F2α and E concentrations in cystic fibrosis patients. Am. Rev. Respir. Dis. 117, 639–646 (1978).
Sinha, G.P. et al. Pachydermoperiostosis in childhood. Br. J. Rheumatol. 36, 1224–1227 (1997).
Latos-Bieleńska, A. et al. Pachydermoperiostosis–critical analysis with report of five unusual cases. Eur. J. Pediatr. 166, 1237–1243 (2007).
Carr, I.M., Flintoff, K.J., Taylor, G.R., Markham, A.F. & Bonthron, D.T. Interactive visual analysis of SNP data for rapid autozygosity mapping in consanguineous families. Hum. Mutat. 27, 1041–1046 (2006).
Cho, H. et al. Role of glutamine 148 of human 15-hydroxyprostaglandin dehydrogenase in catalytic oxidation of prostaglandin E2. Bioorg. Med. Chem. 14, 6486–6491 (2006).
Jung, A. et al. 15-Hydroxyprostaglandin dehydrogenase from human placenta. 1. Isolation and characterization [in German with English abstract]. Hoppe Seylers Z. Physiol. Chem. 356, 787–798 (1975).
Acknowledgements
This study was supported by the UK Medical Research Council (Clinical Research Training Fellowship to S.U.) and Cancer Research UK.
Author information
Authors and Affiliations
Contributions
S.U. and I.M.C. performed genetic mapping and mutation analysis. C.P.D. prepared and assayed recombinant HPGD and performed urine prostaglandin measurements. C.W.G.F. and S.E.V.P. performed structural analysis. S.U., M.A., G.H.I., P.S.H., A.L.-B. and C.P.B. identified subjects with PHO and performed clinical studies. A.F.M., C.P.B. and D.T.B. designed and supervised the project. D.T.B. wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3, Supplementary Tables 1–2 and Supplementary Note (PDF 6347 kb)
Rights and permissions
About this article
Cite this article
Uppal, S., Diggle, C., Carr, I. et al. Mutations in 15-hydroxyprostaglandin dehydrogenase cause primary hypertrophic osteoarthropathy. Nat Genet 40, 789–793 (2008). https://doi.org/10.1038/ng.153
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.153
This article is cited by
-
Touraine-Solente-Gole syndrome: pathogenic variant in SLCO2A1 presented with polyarthralgia and digital clubbing
Pediatric Rheumatology (2023)
-
Analysis of genetic testing in fetuses with congenital heart disease of single atria and/or single ventricle in a Chinese prenatal cohort
BMC Pediatrics (2023)
-
Paraneoplastische Syndrome in der Rheumatologie
Zeitschrift für Rheumatologie (2023)
-
Advances of autoimmune rheumatic diseases related to malignant tumors
Inflammation Research (2023)
-
Comparison of bone microstructure and strength in the distal radius and tibia between the different types of primary hypertrophic osteoarthropathy: an HR-pQCT study
Osteoporosis International (2023)