Abstract
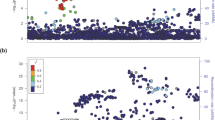
To search for sequence variants conferring risk of nonmedullary thyroid cancer, we focused our analysis on 22 SNPs with a P < 5 × 10−8 in a genome-wide association study on levels of thyroid stimulating hormone (TSH) in 27,758 Icelanders. Of those, rs965513 has previously been shown to associate with thyroid cancer. The remaining 21 SNPs were genotyped in 561 Icelandic individuals with thyroid cancer (cases) and up to 40,013 controls. Variants suggestively associated with thyroid cancer (P < 0.05) were genotyped in an additional 595 non-Icelandic cases and 2,604 controls. After combining the results, three variants were shown to associate with thyroid cancer: rs966423 on 2q35 (OR = 1.34; Pcombined = 1.3 × 10−9), rs2439302 on 8p12 (OR = 1.36; Pcombined = 2.0 × 10−9) and rs116909374 on 14q13.3 (OR = 2.09; Pcombined = 4.6 × 10−11), a region previously reported to contain an uncorrelated variant conferring risk of thyroid cancer. A strong association (P = 9.1 × 10−91) was observed between rs2439302 on 8p12 and expression of NRG1, which encodes the signaling protein neuregulin 1, in blood.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Goldgar, D.E., Easton, D.F., Cannon-Albright, L.A. & Skolnick, M.H. Systematic population-based assessment of cancer risk in first-degree relatives of cancer probands. J. Natl. Cancer Inst. 86, 1600–1608 (1994).
Czene, K., Lichtenstein, P. & Hemminki, K. Environmental and heritable causes of cancer among 9.6 million individuals in the Swedish Family-Cancer Database. Int. J. Cancer 99, 260–266 (2002).
Amundadottir, L.T. et al. Cancer as a complex phenotype: pattern of cancer distribution within and beyond the nuclear family. PLoS Med. 1, e65 (2004).
Hrafnkelsson, J., Tulinius, H., Jonasson, J.G. & Sigvaldason, H. Familial non-medullary thyroid cancer in Iceland. J. Med. Genet. 38, 189–191 (2001).
Kondo, T., Ezzat, S. & Asa, S.L. Pathogenetic mechanisms in thyroid follicular-cell neoplasia. Nat. Rev. Cancer 6, 292–306 (2006).
DeLellis, R.A. Pathology and genetics of thyroid carcinoma. J. Surg. Oncol. 94, 662–669 (2006).
Baida, A. et al. Strong association of chromosome 1p12 loci with thyroid cancer susceptibility. Cancer Epidemiol. Biomarkers Prev. 17, 1499–1504 (2008).
Jazdzewski, K. et al. Common SNP in pre-miR-146a decreases mature miR expression and predisposes to papillary thyroid carcinoma. Proc. Natl. Acad. Sci. USA 105, 7269–7274 (2008).
Jazdzewski, K. et al. Polymorphic mature microRNAs from passenger strand of pre-miR-146a contribute to thyroid cancer. Proc. Natl. Acad. Sci. USA 106, 1502–1505 (2009).
He, H. et al. A susceptibility locus for papillary thyroid carcinoma on chromosome 8q24. Cancer Res. 69, 625–631 (2009).
Gudmundsson, J. et al. Common variants on 9q22.33 and 14q13.3 predispose to thyroid cancer in European populations. Nat. Genet. 41, 460–464 (2009).
Landa, I. et al. The variant rs1867277 in FOXE1 gene confers thyroid cancer susceptibility through the recruitment of USF1/USF2 transcription factors. PLoS Genet. 5, e1000637 (2009).
Takahashi, M. et al. The FOXE1 locus is a major genetic determinant for radiation-related thyroid carcinoma in Chernobyl. Hum. Mol. Genet. 19, 2516–2523 (2010).
Kong, A. et al. Detection of sharing by descent, long-range phasing and haplotype imputation. Nat. Genet. 40, 1068–1075 (2008).
Kong, A. et al. Fine-scale recombination rate differences between sexes, populations and individuals. Nature 467, 1099–1103 (2010).
Panicker, V. et al. A locus on chromosome 1p36 is associated with thyrotropin and thyroid function as identified by genome-wide association study. Am. J. Hum. Genet. 87, 430–435 (2010).
Arnaud-Lopez, L. et al. Phosphodiesterase 8B gene variants are associated with serum TSH levels and thyroid function. Am. J. Hum. Genet. 82, 1270–1280 (2008).
Medici, M. et al. A large-scale association analysis of 68 thyroid hormone pathway genes with serum TSH and FT4 levels. Eur. J. Endocrinol. 164, 781–788 (2011).
Kutyavin, I.V. et al. A novel endonuclease IV post-PCR genotyping system. Nucleic Acids Res. 34, e128 (2006).
Parlato, R. et al. An integrated regulatory network controlling survival and migration in thyroid organogenesis. Dev. Biol. 276, 464–475 (2004).
Stefansson, H. et al. Neuregulin 1 and susceptibility to schizophrenia. Am. J. Hum. Genet. 71, 877–892 (2002).
Garcia-Barcelo, M.M. et al. Genome-wide association study identifies NRG1 as a susceptibility locus for Hirschsprung's disease. Proc. Natl. Acad. Sci. USA 106, 2694–2699 (2009).
Emilsson, V. et al. Genetics of gene expression and its effect on disease. Nature 452, 423–428 (2008).
Teumer, A. et al. Genome-wide association study identifies four genetic loci associated with thyroid volume and goiter risk. Am. J. Hum. Genet. 88, 664–673 (2011).
Gulcher, J.R., Kristjansson, K., Gudbjartsson, H. & Stefansson, K. Protection of privacy by third-party encryption in genetic research in Iceland. Eur. J. Hum. Genet. 8, 739–742 (2000).
Wetzels, J.F., Kiemeney, L.A., Swinkels, D.W., Willems, H.L. & den Heijer, M. Age- and gender-specific reference values of estimated GFR in Caucasians: the Nijmegen Biomedical Study. Kidney Int. 72, 632–637 (2007).
Kutyavin, I.V. et al. A novel endonuclease IV post-PCR genotyping system. Nucleic Acids Res. 34, e128 (2006).
He, H. et al. Allelic variation in gene expression in thyroid tissue. Thyroid 15, 660–667 (2005).
Sulem, P. et al. Identification of low-frequency variants associated with gout and serum uric acid levels. Nat. Genet. 43, 1127–1130 (2011).
Rafnar, T. et al. Mutations in BRIP1 confer high risk of ovarian cancer. Nat. Genet. 43, 1104–1107 (2011).
Stacey, S.N. et al. A germline variant in the TP53 polyadenylation signal confers cancer susceptibility. Nat. Genet. 43, 1098–1103 (2011).
Acknowledgements
We thank the affected individuals whose contribution made this work possible. This project was funded in part by US National Institutes of Health contract numbers CA16058 and CA124570.
Author information
Authors and Affiliations
Contributions
The study was designed and results were interpreted by J.G., P.S., D.F.G., A.K. and K.S. Statistical analysis was carried out by P.S., D.F.G., G.M., G.T., J.G. and A.K. Subject recruitment and biological material collection and handling was organized and carried out by J.G., J.G.J., S.N.S., H.He, W.L., R.N., M.D.R., R.T.K., M.C.H.de.V., T.S.P., M.d.H., E.A., A.P., E.P., A.G.-C., A.D.J., F.R., G.B.W., H.B., L.T., I.O., G.I.E., U.S.B., H.Holm, K.K., H.K., J.R.G., L.A.L.M.K., R.T.N.-M., T.J., H. Hjartarson, J.I.M., A.de.la.C., J.H., U.T. and T.R. Genotyping was supervised and carried out by J.G., A.J., A.S., H.He, H.J., H.Th.H., O.Th.M., W.L. and U.T. J.G., P.S., D.F.G. and K.S. drafted the manuscript. All authors contributed to the final version of the paper. Principal collaborators for the replication case-control samples were J.I.M. (Spain), A.d.l.C. (US) and R.T.N.-M. and L.A.L.M.K. (The Netherlands).
Corresponding authors
Ethics declarations
Competing interests
The authors from deCODE are employees of deCODE genetics Inc. M.D.R. has previously been on an advisory board for Veracyte, Inc. and has been on an advisory panel for AstraZeneca.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3, Supplementary Table 1–4 and Supplementary Note (PDF 456 kb)
Rights and permissions
About this article
Cite this article
Gudmundsson, J., Sulem, P., Gudbjartsson, D. et al. Discovery of common variants associated with low TSH levels and thyroid cancer risk. Nat Genet 44, 319–322 (2012). https://doi.org/10.1038/ng.1046
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.1046
This article is cited by
-
Exposure to Polybrominated Diphenyl Ethers and Thyroid Disease: a Systematic Review and Meta-analysis of Epidemiological Studies
Current Epidemiology Reports (2024)
-
Suggestive evidence of the genetic association of TMOD1 and PTCSC2 polymorphisms with thyroid carcinoma in the Chinese Han population
BMC Endocrine Disorders (2022)
-
Implementation of individualised polygenic risk score analysis: a test case of a family of four
BMC Medical Genomics (2022)
-
Yap governs a lineage-specific neuregulin1 pathway-driven adaptive resistance to RAF kinase inhibitors
Molecular Cancer (2022)
-
Characterizing the function of EPB41L4A in the predisposition to papillary thyroid carcinoma
Scientific Reports (2020)