Abstract
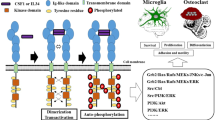
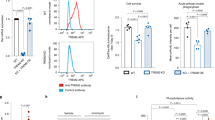
Hereditary diffuse leukoencephalopathy with spheroids (HDLS) is an autosomal-dominant central nervous system white-matter disease with variable clinical presentations, including personality and behavioral changes, dementia, depression, parkinsonism, seizures and other phenotypes1,2. We combined genome-wide linkage analysis with exome sequencing and identified 14 different mutations affecting the tyrosine kinase domain of the colony stimulating factor 1 receptor (encoded by CSF1R) in 14 families with HDLS. In one kindred, we confirmed the de novo occurrence of the mutation. Follow-up sequencing identified an additional CSF1R mutation in an individual diagnosed with corticobasal syndrome. In vitro, CSF-1 stimulation resulted in rapid autophosphorylation of selected tyrosine residues in the kinase domain of wild-type but not mutant CSF1R, suggesting that HDLS may result from partial loss of CSF1R function. As CSF1R is a crucial mediator of microglial proliferation and differentiation in the brain, our findings suggest an important role for microglial dysfunction in HDLS pathogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Axelsson, R., Roytta, M., Sourander, P., Akesson, H.O. & Andersen, O. Hereditary diffuse leucoencephalopathy with spheroids. Acta Psychiatr. Scand. Suppl. 314, 1–65 (1984).
Wider, C. et al. Leukoencephalopathy with spheroids (HDLS) and pigmentary leukodystrophy (POLD): a single entity? Neurology 72, 1953–1959 (2009).
Schiffmann, R. & van der Knaap, M.S. Invited article: an MRI-based approach to the diagnosis of white matter disorders. Neurology 72, 750–759 (2009).
Baba, Y. et al. Hereditary diffuse leukoencephalopathy with spheroids: clinical, pathologic and genetic studies of a new kindred. Acta Neuropathol. 111, 300–311 (2006).
Freeman, S.H. et al. Adult onset leukodystrophy with neuroaxonal spheroids: clinical, neuroimaging and neuropathologic observations. Brain Pathol. 19, 39–47 (2009).
Hancock, N., Poon, M., Taylor, B. & McLean, C. Hereditary diffuse leucoencephalopathy with spheroids. J. Neurol. Neurosurg. Psychiatry 74, 1345–1347 (2003).
Itoh, K. et al. Autosomal dominant leukodystrophy with axonal spheroids and pigmented glia: clinical and neuropathological characteristics. Acta Neuropathol. 111, 39–45 (2006).
Marotti, J.D., Tobias, S., Fratkin, J.D., Powers, J.M. & Rhodes, C.H. Adult onset leukodystrophy with neuroaxonal spheroids and pigmented glia: report of a family, historical perspective, and review of the literature. Acta Neuropathol. 107, 481–488 (2004).
Mateen, F.J. et al. Sporadic leucodystrophy with neuroaxonal spheroids: persistence of DWI changes and neurocognitive profiles: a case study. J. Neurol. Neurosurg. Psychiatry 81, 619–622 (2010).
Swerdlow, R.H. et al. Autosomal dominant subcortical gliosis presenting as frontotemporal dementia. Neurology 72, 260–267 (2009).
van der Knaap, M.S., Naidu, S., Kleinschmidt-Demasters, B.K., Kamphorst, W. & Weinstein, H.C. Autosomal dominant diffuse leukoencephalopathy with neuroaxonal spheroids. Neurology 54, 463–468 (2000).
Van Gerpen, J.A. et al. Insights into the dynamics of hereditary diffuse leukoencephalopathy with axonal spheroids. Neurology 71, 925–929 (2008).
Blume-Jensen, P. & Hunter, T. Oncogenic kinase signalling. Nature 411, 355–365 (2001).
Stanley, E.R. et al. Biology and action of colony-stimulating factor-1. Mol. Reprod. Dev. 46, 4–10 (1997).
Pixley, F.J. & Stanley, E.R. CSF-1 regulation of the wandering macrophage: complexity in action. Trends Cell Biol. 14, 628–638 (2004).
Hamilton, J.A. CSF-1 signal transduction. J. Leukoc. Biol. 62, 145–155 (1997).
Sengupta, A. et al. Identification and subcellular localization of proteins that are rapidly phosphorylated in tyrosine in response to colony-stimulating factor 1. Proc. Natl. Acad. Sci. USA 85, 8062–8066 (1988).
Yeung, Y.G. & Stanley, E.R. Proteomic approaches to the analysis of early events in colony-stimulating factor-1 signal transduction. Mol. Cell. Proteomics 2, 1143–1155 (2003).
Akiyama, H. et al. Expression of the receptor for macrophage colony stimulating factor by brain microglia and its upregulation in brains of patients with Alzheimer's disease and amyotrophic lateral sclerosis. Brain Res. 639, 171–174 (1994).
Raivich, G. et al. Regulation of MCSF receptors on microglia in the normal and injured mouse central nervous system: a quantitative immunofluorescence study using confocal laser microscopy. J. Comp. Neurol. 395, 342–358 (1998).
Wang, Y., Berezovska, O. & Fedoroff, S. Expression of colony stimulating factor-1 receptor (CSF-1R) by CNS neurons in mice. J. Neurosci. Res. 57, 616–632 (1999).
Ridge, S.A., Worwood, M., Oscier, D., Jacobs, A. & Padua, R.A. FMS mutations in myelodysplastic, leukemic, and normal subjects. Proc. Natl. Acad. Sci. USA 87, 1377–1380 (1990).
Soares, M.J. et al. CSF1R copy number changes, point mutations, and RNA and protein overexpression in renal cell carcinomas. Mod. Pathol. 22, 744–752 (2009).
Browne, L., Sweeney, B.J. & Farrell, M.A. Late-onset neuroaxonal leucoencephalopathy with spheroids and vascular amyloid. Eur. Neurol. 50, 85–90 (2003).
Keegan, B.M. et al. Sporadic adult-onset leukoencephalopathy with neuroaxonal spheroids mimicking cerebral MS. Neurology 70, 1128–1133 (2008).
Levin, N. et al. Leukoencephalopathy with neuroaxonal spheroids presenting as frontotemporal dementia. Isr. Med. Assoc. J. 10, 386–387 (2008).
Mascalchi, M. et al. CT and MR imaging of neuroaxonal leukodystrophy presenting as early-onset frontal dementia. AJNR Am. J. Neuroradiol. 27, 1037–1039 (2006).
Moro-de-Casillas, M.L., Cohen, M.L. & Riley, D.E. Leucoencephalopathy with neuroaxonal spheroids (LENAS) presenting as the cerebellar subtype of multiple system atrophy. J. Neurol. Neurosurg. Psychiatry 75, 1070–1072 (2004).
Yamashita, M. & Yamamoto, T. Neuroaxonal leukoencephalopathy with axonal spheroids. Eur. Neurol. 48, 20–25 (2002).
Akiguchi, I., Tomimoto, H., Suenaga, T., Wakita, H. & Budka, H. Alterations in glia and axons in the brains of Binswanger's disease patients. Stroke 28, 1423–1429 (1997).
Lassmann, H. Axonal and neuronal pathology in multiple sclerosis: what have we learnt from animal models. Exp. Neurol. 225, 2–8 (2010).
Smith, T.W., DeGirolami, U., Henin, D., Bolgert, F. & Hauw, J.J. Human immunodeficiency virus (HIV) leukoencephalopathy and the microcirculation. J. Neuropathol. Exp. Neurol. 49, 357–370 (1990).
Klünemann, H.H. et al. The genetic causes of basal ganglia calcification, dementia, and bone cysts: DAP12 and TREM2. Neurology 64, 1502–1507 (2005).
Paloneva, J. et al. CNS manifestations of Nasu-Hakola disease: a frontal dementia with bone cysts. Neurology 56, 1552–1558 (2001).
Tanaka, J. Nasu-Hakola disease: a review of its leukoencephalopathic and membranolipodystrophic features. Neuropathology 20 (suppl.), S25–S29 (2000).
Paloneva, J. et al. Loss-of-function mutations in TYROBP (DAP12) result in a presenile dementia with bone cysts. Nat. Genet. 25, 357–361 (2000).
Paloneva, J. et al. Mutations in two genes encoding different subunits of a receptor signaling complex result in an identical disease phenotype. Am. J. Hum. Genet. 71, 656–662 (2002).
Otero, K. et al. Macrophage colony-stimulating factor induces the proliferation and survival of macrophages via a pathway involving DAP12 and beta-catenin. Nat. Immunol. 10, 734–743 (2009).
Chouery, E. et al. Mutations in TREM2 lead to pure early-onset dementia without bone cysts. Hum. Mutat. 29, E194–E204 (2008).
Bianchin, M.M., Martin, K.C., de Souza, A.C., de Oliveira, M.A. & Rieder, C.R. Nasu-Hakola disease and primary microglial dysfunction. Nat. Rev. Neurol. 6, doi:10.1038/nrneurol.2010.17-c1 (2010).
Gnirke, A. et al. Solution hybrid selection with ultra-long oligonucleotides for massively parallel targeted sequencing. Nat. Biotechnol. 27, 182–189 (2009).
Bentley, D.R. et al. Accurate whole human genome sequencing using reversible terminator chemistry. Nature 456, 53–59 (2008).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Acknowledgements
We thank all family members who participated in this study, D. Gamble for help in coordinating sample and data collection from subjects with ischemic stroke, and R. Ossi for review of brain MRI scans from subjects with ischemic stroke. This work is funded by a Mayo Benefactor and the Mayo Foundation. Mayo Clinic Jacksonville is a Morris K. Udall Parkinson's Disease Research Center of Excellence supported by the US National Institute of Neurological Disorders and Stroke (P50 NS072187). Z.K.W. is partially funded by the US National Institutes of Health (NIH; R01 NS057567 and 1RC2NS070276) and by Mayo Clinic Florida CR programs (MCF 90052018 and MCF 90052030). Z.K.W. is further supported by the family of Carl Edward Bolch Jr. and Susan Bass Bolch, and by Dystonia Medical Research Foundation. R.R. is funded by NIH grants R01 NS065782, R01 AG026251 and P50 AG016574 and by the Peebler PSP Research Foundation. O.A.R. is supported by the American Heart Association, the James & Esther King Biomedical Research Program, the Florida Department of Health and the Myron and Jane Hanley Award in Stroke research. C.W. is supported by the Leenaards Foundation and the Swiss Parkinson Foundation. The Mayo Clinic Florida Cerebrovascular Diseases Registry (IRB no. 08-003878; J.F.M., Principal Investigator) is supported by the Mayo Foundation for Medical Education and Research. C.S. is supported by grants from Sven and Dagmar Saléns, Stiftelse, Sweden; the Swedish and Gothenburg Societies for the Neurologically Disabled; The Swedish Society of Medicine Gothenburg; the Gothenburg Foundation for Neurological Research; and the Björnsson Foundation, Gothenburg, Sweden. This work is further funded by NIH PHS P30 AG 10133 (Indiana Alzheimer Disease Center, to B.G.) and NIH U24 AG 21886-01S1 (National Cell Repository for Alzheimer's disease, to B.G.).
Author information
Authors and Affiliations
Contributions
R.R. and Z.K.W. directed the study. R.R., M.B. and A.M.N. wrote the manuscript. M.B. directed the genetic studies and performed the linkage analysis. A.M.N. directed the functional studies. S.L. performed the exome sequencing and variant calling analysis. M.B., N.J.R., A.S.-O., O.A.R. and A.W. performed the gene sequencing and genotyping studies. A.M.N., N.F., A.W., N.K. and P.D. performed the mutagenesis, cell biology and protein biochemistry studies. Z.K.W. directed the international consortium, assisted by J.L., C.W., C.S., M.D.-H. and J.A. E.A.S., J.A., J.M., S.R., H.A.K., N.J.C., B.G., S.S., A.C.T., J.G., J.A.V.G., B.F.B., D.S.K., R.C.P., D.F.B., J.F.M., R.U., N.G.-R., R.H.S., B.B.M., D.W.D. and Z.K.W. performed clinical evaluations. B.B.M. performed radiological studies. D.W.D. performed neuropathological studies. All authors contributed to manuscript revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6, Supplementary Tables 1–6 and Supplementary Note (PDF 1211 kb)
Rights and permissions
About this article
Cite this article
Rademakers, R., Baker, M., Nicholson, A. et al. Mutations in the colony stimulating factor 1 receptor (CSF1R) gene cause hereditary diffuse leukoencephalopathy with spheroids. Nat Genet 44, 200–205 (2012). https://doi.org/10.1038/ng.1027
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.1027
This article is cited by
-
Progranulin haploinsufficiency mediates cytoplasmic TDP-43 aggregation with lysosomal abnormalities in human microglia
Journal of Neuroinflammation (2024)
-
The aging mouse CNS is protected by an autophagy-dependent microglia population promoted by IL-34
Nature Communications (2024)
-
Brain abnormalities, neurodegeneration, and dysosteosclerosis (BANDDOS): new cases, systematic literature review, and associations with CSF1R-ALSP
Orphanet Journal of Rare Diseases (2023)
-
An AARS1 variant identified to cause adult-onset leukoencephalopathy with neuroaxonal spheroids and pigmented glia
Translational Neurodegeneration (2023)
-
Physiology and diseases of tissue-resident macrophages
Nature (2023)