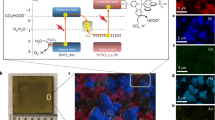
Abstract
The solar-driven electrochemical reduction of CO2 to fuels and chemicals provides a promising way for closing the anthropogenic carbon cycle. However, the lack of selective and Earth-abundant catalysts able to achieve the desired transformation reactions in an aqueous matrix presents a substantial impediment as of today. Here we introduce atomic layer deposition of SnO2 on CuO nanowires as a means for changing the wide product distribution of CuO-derived CO2 reduction electrocatalysts to yield predominantly CO. The activity of this catalyst towards oxygen evolution enables us to use it both as the cathode and anode for complete CO2 electrolysis. In the resulting device, the electrodes are separated by a bipolar membrane, allowing each half-reaction to run in its optimal electrolyte environment. Using a GaInP/GaInAs/Ge photovoltaic we achieve the solar-driven splitting of CO2 into CO and oxygen with a bifunctional, sustainable and all Earth-abundant system at an efficiency of 13.4%.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schreier, M. et al. Efficient and selective carbon dioxide reduction on low cost protected Cu2O photocathodes using a molecular catalyst. Energy Environ. Sci. 8, 855–861 (2015).
Schreier, M. et al. Covalent immobilization of a molecular catalyst on Cu2O photocathodes for CO2 reduction. J. Am. Chem. Soc. 138, 1938–1946 (2016).
Lau, G. P. S. et al. New insights into the role of imidazolium-based promoters for the electroreduction of CO2 on a silver electrode. J. Am. Chem. Soc. 138, 7820–7823 (2016).
Zhang, S. et al. Polymer-supported CuPd nanoalloy as a synergistic catalyst for electrocatalytic reduction of carbon dioxide to methane. Proc. Natl Acad. Sci. USA 112, 15809–15814 (2015).
Ren, D. et al. Selective electrochemical reduction of carbon dioxide to ethylene and ethanol on copper(I) oxide catalysts. ACS Catal. 5, 2814–2821 (2015).
Qiao, J., Liu, Y., Hong, F. & Zhang, J. A review of catalysts for the electroreduction of carbon dioxide to produce low-carbon fuels. Chem. Soc. Rev. 43, 631–675 (2014).
Kuhl, K. P., Cave, E. R., Abram, D. N. & Jaramillo, T. F. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energy Environ. Sci. 5, 7050–7059 (2012).
Kuhl, K. P. et al. Electrocatalytic conversion of carbon dioxide to methane and methanol on transition metal surfaces. J. Am. Chem. Soc. 136, 14107–14113 (2014).
Chen, Y., Li, C. W. & Kanan, M. W. Aqueous CO2 reduction at very low overpotential on oxide-derived Au nanoparticles. J. Am. Chem. Soc. 134, 19969–19972 (2012).
Feng, X., Jiang, K., Fan, S. & Kanan, M. W. Grain-boundary-dependent CO2 electroreduction activity. J. Am. Chem. Soc. 137, 4606–4609 (2015).
Min, X. & Kanan, M. W. Pd-catalyzed electrohydrogenation of carbon dioxide to formate: high mass activity at low overpotential and identification of the deactivation pathway. J. Am. Chem. Soc. 137, 4701–4708 (2015).
Lu, Q. et al. A selective and efficient electrocatalyst for carbon dioxide reduction. Nat. Commun. 5, 3242 (2014).
Hori, Y. Handbook of Fuel Cells (John Wiley, 2010).
DiMeglio, J. L. & Rosenthal, J. Selective conversion of CO2 to CO with high efficiency using an inexpensive bismuth-based electrocatalyst. J. Am. Chem. Soc. 135, 8798–8801 (2013).
Li, C. W. & Kanan, M. W. CO2 reduction at low overpotential on Cu electrodes resulting from the reduction of thick Cu2O films. J. Am. Chem. Soc. 134, 7231–7234 (2012).
Rasul, S. et al. A highly selective copper–indium bimetallic electrocatalyst for the electrochemical reduction of aqueous CO2 to CO. Angew. Chem. Int. Ed. 54, 2146–2150 (2015).
Watanabe, M., Shibata, M., Kato, A., Azuma, M. & Sakata, T. Design of alloy electrocatalysts for CO2 reduction III. The selective and reversible reduction of CO2 on Cu alloy electrodes. J. Electrochem. Soc. 138, 3382–3389 (1991).
Sarfraz, S., Garcia-Esparza, A. T., Jedidi, A., Cavallo, L. & Takanabe, K. Cu–Sn bimetallic catalyst for selective aqueous electroreduction of CO2 to CO. ACS Catal. 6, 2842–2851 (2016).
Wuttig, A. & Surendranath, Y. Impurity ion complexation enhances carbon dioxide reduction catalysis. ACS Catal. 5, 4479–4484 (2015).
Lum, Y. et al. Trace levels of copper in carbon materials show significant electrochemical CO2 reduction activity. ACS Catal. 6, 202–209 (2016).
Gattrell, M., Gupta, N. & Co, A. A review of the aqueous electrochemical reduction of CO2 to hydrocarbons at copper. J. Electroanal. Chem. 594, 1–19 (2006).
Ma, M., Djanashvili, K. & Smith, W. A. Selective electrochemical reduction of CO2 to CO on CuO-derived Cu nanowires. Phys. Chem. Chem. Phys. 17, 20861–20867 (2015).
Chen, Y. & Kanan, M. W. Tin oxide dependence of the CO2 reduction efficiency on tin electrodes and enhanced activity for Tin/Tin oxide thin-film catalysts. J. Am. Chem. Soc. 134, 1986–1989 (2012).
Zhang, S., Kang, P. & Meyer, T. J. Nanostructured tin catalysts for selective electrochemical reduction of carbon dioxide to formate. J. Am. Chem. Soc. 136, 1734–1737 (2014).
Li, C. W., Ciston, J. & Kanan, M. W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 508, 504–507 (2014).
Verdaguer-Casadevall, A. et al. Probing the active surface sites for CO reduction on oxide-derived copper electrocatalysts. J. Am. Chem. Soc. 137, 9808–9811 (2015).
Nørskov, J. K. et al. Trends in the exchange current for hydrogen evolution. J. Electrochem. Soc. 152, J23 (2005).
Eilert, A., Roberts, F. S., Friebel, D. & Nilsson, A. Formation of copper catalysts for CO2 reduction with high ethylene/methane product ratio investigated with in situ X-ray absorption spectroscopy. J. Phys. Chem. Lett. 7, 1466–1470 (2016).
Lee, S., Kim, D. & Lee, J. Electrocatalytic production of C3–C4 compounds by conversion of CO2 on a chloride-induced Bi-phasic Cu2 O–Cu catalyst. Angew. Chem. Int. Edn 54, 14701–14705 (2015).
Mistry, H. et al. Highly selective plasma-activated copper catalysts for carbon dioxide reduction to ethylene. Nat. Commun. 7, 12123 (2016).
Dutta, A., Kuzume, A., Rahaman, M., Vesztergom, S. & Broekmann, P. Monitoring the chemical state of catalysts for CO2 electroreduction: an in operando study. ACS Catal. 5, 7498–7502 (2015).
Deng, Y., Handoko, A. D., Du, Y., Xi, S. & Yeo, B. S. In situ Raman spectroscopy of copper and copper oxide surfaces during electrochemical oxygen evolution reaction: identification of CuIII oxides as catalytically active species. ACS Catal. 6, 2473–2481 (2016).
Liu, X., Cui, S., Qian, M., Sun, Z. & Du, P. In situ generated highly active copper oxide catalysts for the oxygen evolution reaction at low overpotential in alkaline solutions. Chem. Commun. 52, 5546–5549 (2016).
Liu, X. et al. Self-supported copper oxide electrocatalyst for water oxidation at low overpotential and confirmation of its robustness by Cu K-edge X-ray absorption spectroscopy. J. Phys. Chem. C 120, 831–840 (2016).
Liu, X. et al. Nanostructured copper oxide electrodeposited from copper(II) complexes as an active catalyst for electrocatalytic oxygen evolution reaction. Electrochem. Commun. 46, 1–4 (2014).
Luo, J. et al. Bipolar membrane-assisted solar water splitting in optimal pH. Adv. Energy Mater. 6, 1600100 (2016).
Vargas-Barbosa, N. M., Geise, G. M., Hickner, M. A. & Mallouk, T. E. Assessing the utility of bipolar membranes for use in photoelectrochemical water-splitting cells. ChemSusChem 7, 3017–3020 (2014).
Vermaas, D. A., Sassenburg, M. & Smith, W. A. Photo-assisted water splitting with bipolar membrane induced pH gradients for practical solar fuel devices. J. Mater Chem. A 3, 19556–19562 (2015).
Sun, K. et al. A stabilized, intrinsically safe, 10% efficient, solar-driven water-splitting cell incorporating earth-abundant electrocatalysts with steady-state PH gradients and product separation enabled by a bipolar membrane. Adv. Energy Mater. 6, 1600379 (2016).
Schreier, M. et al. Efficient photosynthesis of carbon monoxide from CO2 using perovskite photovoltaics. Nat. Commun. 6, 7326 (2015).
O’Neill, B. J. et al. Catalyst design with atomic layer deposition. ACS Catal. 5, 1804–1825 (2015).
Dry, M. E. The Fischer–Tropsch process: 1950–2000. Catal. Today 71, 227–241 (2002).
Li, Y. & Sun, Q. Recent advances in breaking scaling relations for effective electrochemical conversion of CO2 . Adv. Energy Mater. 6, 1600463 (2016).
Luo, J. et al. Cu2O nanowire photocathodes for efficient and durable solar water splitting. Nano Lett. 16, 1848–1857 (2016).
Steier, L. et al. Low-temperature atomic layer deposition of crystalline and photoactive ultrathin hematite films for solar water splitting. ACS Nano 9, 11775–11783 (2015).
Tidwell, E. K. P., Blaine, L. R. & Eugene, D. Infrared absorption and emission spectra of carbon monoxide in the region from 4 to 6 microns. J. Res. Natl Bur. Stand. 55, 183–189 (1955).
Acknowledgements
The authors acknowledge L. Pan for experimental help during revision, K. V. Thomas and S. Coudret for ICP-MS analysis, P. Mettraux for XPS analysis, M. Söderlund and H. Tholense (Beneq, Finland) for FBR-ALD depositions and D. Alexander for aberration-corrected STEM data. This work was funded by Siemens AG, and M.S. and M.G. would like to express their particular gratitude to Siemens AG for continued support. J.L. acknowledges the Marie Skłodowska-Curie Fellowship from the European Union’s Seventh Framework Programme for research, technological development and demonstration under grant agreement no. 291771 for financial support.
Author information
Authors and Affiliations
Contributions
M.S. designed the project, prepared samples, conducted experiments, analysed the data and wrote the manuscript. J.L. designed and supervised the project, and contributed to experiments, writing of the manuscript and data analysis. F.H. conducted chemisorption and TPD measurements and analysed the relevant data. L.S. assisted with ALD deposition of SnO2. J.S.L. assisted with chemisorption and TPD analysis and corrected the manuscript. M.T.M. contributed to writing the manuscript. S.A. provided the three-junction solar cells and had the idea for its use in this application. M.G. supervised the project, directed the research and established the final version of the manuscript. All authors commented on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Notes 1–5, Supplementary Figures 1–36, Supplementary Tables 1–4 and Supplementary References. (PDF 2206 kb)
Rights and permissions
About this article
Cite this article
Schreier, M., Héroguel, F., Steier, L. et al. Solar conversion of CO2 to CO using Earth-abundant electrocatalysts prepared by atomic layer modification of CuO. Nat Energy 2, 17087 (2017). https://doi.org/10.1038/nenergy.2017.87
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2017.87
This article is cited by
-
Designing Membrane Electrode Assembly for Electrochemical CO2 Reduction: a Review
Transactions of Tianjin University (2024)
-
Free-Standing Single-Atom Catalyst-Based Electrodes for CO2 Reduction
Electrochemical Energy Reviews (2024)
-
Electrochemical synthesis of propylene from carbon dioxide on copper nanocrystals
Nature Chemistry (2023)
-
Activating dynamic atomic-configuration for single-site electrocatalyst in electrochemical CO2 reduction
Nature Communications (2023)
-
Rational design of bimetallic catalysts for electrochemical CO2 reduction reaction: A review
Science China Chemistry (2023)