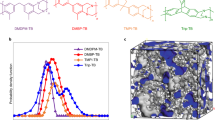
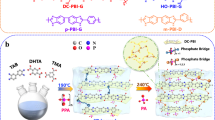
Abstract
Fuel cells are promising devices for clean power generation in a variety of economically and environmentally significant applications. Low-temperature proton exchange membrane (PEM) fuel cells utilizing Nafion require a high level of hydration, which limits the operating temperature to less than 100 ∘C. In contrast, high-temperature PEM fuel cells utilizing phosphoric acid-doped polybenzimidazole can operate effectively up to 180 ∘C; however, these devices degrade when exposed to water below 140 ∘C. Here we present a different class of PEM fuel cells based on quaternary ammonium-biphosphate ion pairs that can operate under conditions unattainable with existing fuel cell technologies. These fuel cells exhibit stable performance at 80–160 ∘C with a conductivity decay rate more than three orders of magnitude lower than that of a commercial high-temperature PEM fuel cell. By increasing the operational flexibility, this class of fuel cell can simplify the requirements for heat and water management, and potentially reduce the costs associated with the existing fully functional fuel cell systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lemmon, J. P. Reimagine fuel cells. Nature 525, 447–449 (2015).
Voelcker, J. 2016 Toyota Mirai Priced at $57,500, with $499 Monthly Lease (Green Car Reports, 2014); http://www.greencarreports.com/news/1095511_2016-toyota-mirai-priced-at-57500-with-499-monthly-lease
Park, C. H. et al. Nanocrack-regulated self-humidifying membranes. Nature 532, 480–483 (2016).
Alberti, G., Casciola, M., Massinelli, L. & Bauer, B. Polymeric proton conducting membranes for medium temperature fuel cells (110–160 ∘C). J. Membr. Sci. 185, 73–81 (2001).
Wainright, J. S., Wang, J. T., Weng, D., Savinell, R. F. & Litt, M. Acid-doped polybenzimidazoles: a new polymer electrolyte. J. Electrochem. Soc. 142, L121–L123 (1995).
Bendlamudi, G. Systematic Characterization of HT PEMFCs Containing PBI/H3PO4 Systems: Thernodynaimic Analysis and Experiemntal Investigations 104 (Logos Verlag Berlin GmbH, 2011).
Xiao, L. X. et al. High-temperature polybenzimidazole fuel cell membranes via a sol-gel process. Chem. Mater. 17, 5328–5333 (2005).
Steininger, H. et al. Intermediate temperature proton conductors for PEM fuel cells based on phosphonic acid as protogenic group: a progress report. Phys. Chem. Chem. Phys. 9, 1764–1773 (2007).
Ma, Y. L., Wainright, J. S., Litt, M. H. & Savinell, R. H. Conductivity of PBI membranes for high-temperature polymer electrolyte fuel cells. J. Electrochem. Soc. 151, A8–A16 (2004).
Kim, Y. S. et al. Structure-property-performance relationships of sulfonated poly(arylene ether sulfone)s as a polymer electrolyte for fuel cell applications. Polymer 47, 4026–4035 (2006).
Mocoteguy, P. et al. Long-term testing in dynamic mode of HT-PEMFC H3PO4/PBI Celtec-P based membrane electrode assemblies for micro-CHP applications. Fuel Cells 10, 299–311 (2010).
Schmidt, T. J. & Baurmeister, J. Properties of high-temperature PEFC Celtec (R)-P 1000 MEAs in start/stop operation mode. J. Power Sources 176, 428–434 (2008).
Hibbs, M. R., Fujimoto, C. H. & Cornelius, C. J. Synthesis and characterization of poly(phenylene)-based anion exchange membranes for alkaline fuel cells. Macromolecules 42, 8316–8321 (2009).
Kim, Y. S. & Lee, K.-S. Fuel cell membrane chracterizations. Polym. Rev. 55, 330–370 (2015).
Frisch, M. J. T. et al. Gaussian 09 (Gaussian, 2009). http://www.gaussian.com/g_prod/g09.htm
Chai, J. D. & Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys. Chem. Chem. Phys. 10, 6615–6620 (2008).
Hehre, W. J., Ditchfie, R. & Pople, J. A. Self-consistent molecular-orbital methods. XII. Further extensions of Gaussian-type basis sets for use in molecular-orbital studies of organic-molecules. J. Chem. Phys. 56, 2257–2261 (1972).
Ransil, B. J. Studies in molecular structure. IV. Potential curve for interaction of two helium atoms in single-configuration LCAO MO SCF approximation. J. Chem. Phys. 34, 2109–2118 (1961).
Boys, S. F. & Bernardi, F. Calculation of small molecular interactions by differences of separate total energies – Some procedures with reduced errors. Mol. Phys. 19, 553–566 (1970).
Fujimoto, C., Kim, D. S., Hibbs, M., Wrobleski, D. & Kim, Y. S. Backbone stability of quaternized polyaromatics for alkaline membrane fuel cells. J. Membr. Sci. 423, 438–449 (2012).
Choe, Y. K. et al. Alkaline stability of benzyl trimethyl ammonium functionalized polyaromatics: a computational and experimental study. Chem. Mater. 26, 5675–5682 (2014).
Li, Q., He, R., Jensen, J. O. & Bjerrum, N. J. PBI-based polymer membranes for high temperature fuel cells – Preparation, characterization and fuel cell demonstration. Fuel Cells 4, 147–159 (2004).
Acknowledgements
This work was supported by the US Department of Energy, Energy Efficiency and Renewable Energy, Fuel Cell Technology Office. We thank E. S. De Castro for useful discussion and for supplying PBI samples. We also thank C. Kreller for helping with the polymer thermal analysis. Y.-K.C. acknowledges financial support from the Ministry of Economy, Trade and Industry of Japan through the Japan-US Cooperation on Clean Energy Technology Program. Los Alamos National Laboratory is operated by Los Alamos National Security, LLC under Contract DE-AC52-06NA25396. Sandia National Laboratories is a multi-program laboratory operated by Sandia Corporation, a wholly owned subsidiary of Lockheed Martin Company, for the US Department of Energy’s National Nuclear Security Administration under contract DE-AC04-94AL85000.
Author information
Authors and Affiliations
Contributions
Y.S.K. developed the intellectual concept, designed all the experiments, and supervised this research. K.-S.L. performed the MEA fabrication and testing experiments of fuel cells. Y.-K.C. performed the modelling work. C.F. and K.-S.L. prepared the polymeric materials. K.-S.L., J.S.S. and Y.S.K. analysed all experimental data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–11, Supplementary Table 1 (PDF 1296 kb)
Rights and permissions
About this article
Cite this article
Lee, KS., Spendelow, J., Choe, YK. et al. An operationally flexible fuel cell based on quaternary ammonium-biphosphate ion pairs. Nat Energy 1, 16120 (2016). https://doi.org/10.1038/nenergy.2016.120
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2016.120
This article is cited by
-
Overcoming the Electrode Challenges of High-Temperature Proton Exchange Membrane Fuel Cells
Electrochemical Energy Reviews (2023)
-
Protonated phosphonic acid electrodes for high power heavy-duty vehicle fuel cells
Nature Energy (2022)
-
Fuel cells with an operational range of –20 °C to 200 °C enabled by phosphoric acid-doped intrinsically ultramicroporous membranes
Nature Energy (2022)
-
Structural architectures of polymer proton exchange membranes suitable for high-temperature fuel cell applications
Science China Materials (2022)
-
Designing the next generation of proton-exchange membrane fuel cells
Nature (2021)