Abstract
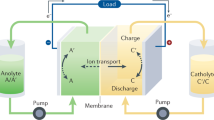
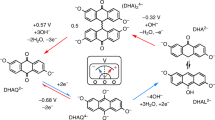
Redox-flow batteries (RFBs) can store large amounts of electrical energy from variable sources, such as solar and wind. Recently, redox-active organic molecules in aqueous RFBs have drawn substantial attention due to their rapid kinetics and low membrane crossover rates. Drawing inspiration from nature, here we report a high-performance aqueous RFB utilizing an organic redox compound, alloxazine, which is a tautomer of the isoalloxazine backbone of vitamin B2. It can be synthesized in high yield at room temperature by single-step coupling of inexpensive o-phenylenediamine derivatives and alloxan. The highly alkaline-soluble alloxazine 7/8-carboxylic acid produces a RFB exhibiting open-circuit voltage approaching 1.2 V and current efficiency and capacity retention exceeding 99.7% and 99.98% per cycle, respectively. Theoretical studies indicate that structural modification of alloxazine with electron-donating groups should allow further increases in battery voltage. As an aza-aromatic molecule that undergoes reversible redox cycling in aqueous electrolyte, alloxazine represents a class of radical-free redox-active organics for use in large-scale energy storage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Dunn, B., Kamath, H. & Tarascon, J.-M. Electrical energy storage for the grid: a battery of choices. Science 334, 928–935 (2011).
Nguyen, T. & Savinell, R. F. Flow batteries. Electrochem. Soc. Interface 19, 54–56 (2010).
Yang, Z. et al. Electrochemical energy storage for green grid. Chem. Rev. 111, 3577–3613 (2011).
Biello, D. Solar wars. Sci. Am. 311, 66–71 (2014).
Skyllas-Kazacos, M., Chakrabarti, M. H., Hajimolana, S. A., Mjalli, F. S. & Saleem, M. Progress in flow battery research and development. J. Electrochem. Soc. 158, R55–R79 (2011).
Weber, A. Z. et al. Redox flow batteries: a review. J. Appl. Electrochem. 41, 1137–1164 (2011).
Perry, M. L. & Weber, A. Z. Advanced redox-flow batteries: a perspective. J. Electrochem. Soc. 163, A5064–A5067 (2016).
Huskinson, B. et al. A metal-free organic-inorganic aqueous flow battery. Nature 505, 195–198 (2014).
Huskinson, B., Marshak, M. P., Gerhardt, M. R. & Aziz, M. J. Cycling of a quinone-bromide flow battery for large-scale electrochemical energy storage. ECS Trans. 61, 27–30 (2014).
Lin, K. et al. Alkaline quinone flow battery. Science 349, 1529–1532 (2015).
Janoschka, T. et al. An aqueous, polymer-based redox-flow battery using non-corrosive, safe, and low-cost materials. Nature 527, 78–81 (2015).
Schubert, U. S. et al. Polymer/zinc hybrid-flow battery using block copolymer micelles featuring a TEMPO corona as catholyte. Polym. Chem. 28, 2238–2243 (2016).
Liu, T., Wei, X., Nie, Z., Sprenkle, V. & Wang, W. A total organic aqueous redox flow battery employing a low cost and sustainable methyl viologen anolyte and 4-HO-TEMPO catholyte. Adv. Energy Mater. 6, 1501449 (2015).
Milstein, J. D., Liang, S., Liou, C., Badel, A. F. & Brushett, F. R. Voltammetry study of quinoxaline in aqueous electrolytes. Electrochim. Acta 180, 695–704 (2015).
Brushett, F., Jansen, A. N., Vaughey, J. T. & Milshtein, J. D. Materials for use with aqueous redox flow batteries and related methods and systems. International Patent Application WO 2015/126907.
Chen, S., Hossain, M. S. & Foss, F. W. Organocatalytic dakin oxidation by nucleophilic flavin catalysts. Org. Lett. 14, 2806–2809 (2012).
de Gonzalo, G., Smit, C., Jin, J., Minnaard, A. J. & Fraaije, M. W. Turning a riboflavin-binding protein into a self-sufficient monooxygenase by cofactor redesign. Chem. Commun. 47, 11050–11052 (2011).
Lindén, A. A., Johansson, M., Hermanns, N. & Bäckvall, J.-E. Efficient and selective sulfoxidation by hydrogen peroxide, using a recyclable flavin-[BMIm]PF6 catalytic system. J. Org. Chem. 71, 3849–3853 (2006).
Hasford, J. J. & Rizzo, C. J. Linear free energy substituent effect on flavin redox chemistry. J. Am. Chem. Soc. 120, 2251–2255 (1998).
Müller, F. in Radicals in Biochemistry (eds Boschke, F. L. et al.) 71–107 (Springer, 1983).
Hong, J. et al. Biologically inspired pteridine redox centres for rechargeable batteries. Nature Commun. 5, 5335 (2014).
Er, S., Suh, C., Marshak, M. P. & Aspuru-Guzik, A. Computational design of molecules for an all-quinone redox flow battery. Chem. Sci. 6, 885–893 (2015).
Koziol, J. & Metzler, D. E. Formation and possible structure of covalent hydrates of alloxazines. Z. Naturforsch. B 27, 1027–1029 (2014).
Surrey, A. R. & Nachod, F. C. Alkaline hydrolysis of riboflavin. J. Am. Chem. Soc. 73, 2336–2338 (1951).
Ahmad, I., Rapson, H. D. C., Heelis, P. F. & Phillips, G. O. Alkaline hydrolysis of 7,8-dimethyl-10-(formylmethyl)isoalloxazine. A kinetic study. J. Org. Chem. 45, 731–733 (1980).
Prukała, D. et al. Acid–base equilibriums of lumichrome and its 1-methyl, 3-methyl, and 1,3-dimethyl derivatives. J. Phys. Chem. A 116, 7474–7490 (2012).
Koziol, J., Tyrakowska, B. & Müller, F. The structure of covalent hydrates of alloxazines. A reinvestigation. Helv. Chim. Acta 64, 1812–1817 (1981).
O’Connor, C. Acidic and basic amide hydrolysis. Q. Rev. Chem. Soc. 24, 553–564 (1970).
Liu, Q. H. et al. High performance vanadium redox flow batteries with optimized electrode configuration and membrane selection. J. Electrochem. Soc. 159, A1246–A1252 (2012).
Li, X.-L. & Fu, Y. Theoretical study of reduction potentials of substituted flavins. J. Mol. Struct. THEOCHEM 856, 112–118 (2008).
Gómez-Bombarelli, R., González-Pérez, M., Pérez-Prior, M. T., Calle, E. & Casado, J. Computational calculation of equilibrium constants: addition to carbonyl compounds. J. Phys. Chem. A 113, 11423–11428 (2009).
Acknowledgements
This work was funded by the US DOE ARPA-E award no. DE-AR0000348, NSF no. NSF-CBET-1509041, the Massachusetts Clean Energy Technology Center, and the Harvard John A. Paulson School of Engineering and Applied Sciences. We thank C. Qian for designing the Fig. 1d scheme. We appreciate support from the Odyssey Cluster and Research Computing of Harvard University’s Faculty of Arts and Sciences.
Author information
Authors and Affiliations
Contributions
K.L., R.G.G. and M.J.A. formulated the project. K.L. and L.T. synthesized the compounds. K.L., E.S.B. and L.T. collected and analysed the NMR data. K.L., Q.C., E.S.B. and A.V. collected and analysed the electrochemical data. K.L. and A.V. measured solubility. R.G.-B. and A.A.-G. performed theoretical analysis. K.L., R.G.G. and M.J.A. wrote the paper, and all authors contributed to revising the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–13, Supplementary Table 1–2. (PDF 946 kb)
Rights and permissions
About this article
Cite this article
Lin, K., Gómez-Bombarelli, R., Beh, E. et al. A redox-flow battery with an alloxazine-based organic electrolyte. Nat Energy 1, 16102 (2016). https://doi.org/10.1038/nenergy.2016.102
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2016.102
This article is cited by
-
Understanding capacity fade in organic redox-flow batteries by combining spectroscopy with statistical inference techniques
Nature Communications (2023)
-
Identifying and preventing degradation in flavin mononucleotide-based redox flow batteries via NMR and EPR spectroscopy
Nature Communications (2023)
-
Mediated formic acid flow fuel cell (MFAFFC) based on biomimetic electrolytes
Journal of Flow Chemistry (2023)
-
RedDB, a computational database of electroactive molecules for aqueous redox flow batteries
Scientific Data (2022)
-
Development of efficient aqueous organic redox flow batteries using ion-sieving sulfonated polymer membranes
Nature Communications (2022)