Abstract
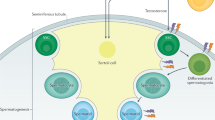
In male cancer patients, surgery, radiotherapy and chemotherapy can be followed by transient or permanent infertility by affecting ejaculatory or erectile function, or by impairing spermatogenesis. Cancer specialists should, therefore, discuss the impact of different therapies on fertility with their patients prior to treatment, and consider fertility-preserving measures before and during treatment, such as nerve-sparing operative techniques, adequate testicular shielding during radiotherapy and the avoidance of unnecessary gonadotoxic chemotherapy. Pretreatment sperm-cell cryopreservation should be offered, even in cases where the individual's risk of post-treatment infertility might seem minimal or if it might require testicular sperm-cell extraction. Samples that are severely oligospermic should also be cryopreserved. Post-treatment ejaculatory or erectile dysfunction can be reversed pharmacologically, but the success rate varies with the extent of neurologic sequelae of the treatments used. At present there is no established method to stimulate post-treatment impaired spermatogenesis, although currently available assisted reproductive techniques overcome some of the existing problems of infertility in cancer survivors, and ongoing research will hopefully increase these possibilities. A multidisciplinary approach that depends on close cooperation between relevant medical specialists is central to achieving such advances.
Key Points
-
Extensive pelvic or retroperitoneal surgery is often followed by erectile and/or ejaculatory dysfunction with subsequent fertility problems
-
Chemotherapy and infradiaphragmatic radiotherapy carry the risk of transiently or permanently impaired spermatogenesis
-
Semen cryopreservation should be offered to all men with cancer at risk of post-treatment infertility who are considering paternity
-
In males above the age of 12–13 years, mature sperm cells can be retrieved from the testes for cryopreservation by invasive procedures if they cannot be obtained by masturbation
-
Experimental approaches such as germ-cell transplantation and sperm-cell maturation in ectopic grafts sourced from testicular-tissue biopsies might enable very young patients to go on to become fathers
-
Future research with germ stem cells or embryonic stem cells will increase the number of options available for saving or restoring fertility in male cancer patients
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Møller B et al. (2002) Prediction of cancer incidence in the Nordic countries up to the year 2020. Eur J Cancer Prev 11 (Suppl 1): S1–96
Steliarova-Foucher E et al. (2004) Geographical patterns and time trends of cancer incidence and survival among children and adolescents in Europe since the 1970s (the ACCIS project): an epidemiological study. Lancet 364: 2097–2105
Landis SH et al. (1999) Cancer Statistics, 1999. Ca Cancer J Clin 49: 8–31
Sant M et al. (2003) EUROCARE-3: survival of cancer patients diagnosed 1990–94—results and commentary. Ann Oncol 14 (Suppl 5): V61–V118
Fosså SD et al. (2005) Parenthood in survivors after adulthood cancer and perinatal health in their offspring: a preliminary report. J Natl Cancer Inst Monogr 34: 77–82
Islam N and Trainer PJ (1998) The hormonal assessment of the infertile male. Br J Urol 82: 69–75
Dohle GR et al. (2003) Androgens and male fertility. World J Urol 21: 341–345
WHO (1992) WHO Laboratory manual for the examination of human semen and sperm-cervical mucus interactions, edn 3. Cambridge: Cambridge University Press
Andersson AM et al. (2004) Serum inhibin B and follicle-stimulating hormone levels as tools in the evaluation of infertile men: significance of adequate reference values from proven fertile men. J Clin Endocrinol Metab 89: 2873–2879
Klein EA (2000) Open technique for nerve-sparing retroperitoneal lymphadenectomy. Urology 55: 132–135
Mirone V et al. (2003) Erectile dysfunction after surgical treatment. Int J Androl 26: 137–140
Skakkebæk NE et al. (2001) Testicular dysgenesis syndrome: an increasingly common development disorder with encironmental aspects. Hum Reprod 16: 972–978
Lim HN et al. (2001) Genetic analysis of the INSL3 gene in patients with maldescent of the testis. Eur Endocrinol 144: 129–137
Brehm R and Steger K (2005) Regulation of Sertoli cell and germ cell differentation. Adv Anat Embryol Cell Biol 181: 1–93
Petersen PM et al. (1999) Semen quality and reproductive hormones before and after orchiectomy in men with testicular cancer. J Urol 161: 822–826
Skakkebaek NE (1978) Carcinoma in situ of the testis: frequency and relationship to invasive germ cell tumours in infertile men. Histopathology 2: 157–170
Raman JD et al. (2005) Increased incidence of testicular cancer in men presenting with infertility and abnormal semen analysis. J Urol 174: 1819–1822
Berthelsen JG and Skakkebaek NE (1983) Gonadal function in men with testis cancer. Fertil Steril 39: 68–75
Nakazumi H et al. (1996) Estrogen metabolism and impaired spermatogenesis in germ cell tumors of the testis. J Clin Endocrinol Metab 81: 1289–1295
Rueffer U et al. (2001) Male gonadal dysfunction in patients with Hodgkin´s disease prior to treatment. Ann Oncol 12: 1307–1311
Viviani S et al. (1991) Testicular dysfunction in Hodgkin`s disease before and after treatment. Eur J Cancer 27: 1389–1392
Höbarth K et al. (1994) Incidence of antisperm antibodies in patients with carcinoma of the testis and in subfertile men with normogonadotropic oligoasthenoteratozoospermia. Urol Int 52: 162–165
Fosså SD et al. (1984) Unilateral retroperitoneal lymph node dissection in patients with nonseminomatous testicular tumor in clinical stage I. Eur Urol 10: 17–23
Montorsi F et al. (2004) Current and future strategies for preventing and managing erectile dysfunction following radical prostatectomy. Eur Urol 45: 123–133
Hollenbeck BK et al. (2003) Determinants of long-term sexual health outcome after radical prostatectomy measured by a validated instrument. J Urol 169: 1453–1457
Havenga K et al. (2000) Avoiding long-term disturbance to bladder and sexual function in pelvic surgery, particularly with rectal cancer. Sem Surg Oncol 18; 235–243
Aus G et al. (2005) EAU Guidelines on prostate cancer. Eur Urol 48: 546–551
Hobday TJ (2005) An overview of approaches to adjuvant therapy for colorectal cancer in the United States. Clin Colorectal Cancer 5 (Suppl 1): F11–F18
Shipley WU et al. (2005) Selective bladder preservation by trimodality therapy for patients with muscularis propria-invasive bladder cancer and who are cystectomy candidates—the Massachusetts general Hospital and Radiation Therapy Oncology Group experiences. Semin Radiat Oncol 15: 36–41
Opjordsmoen S et al. (1994) Sexuality in patients treated for penile cancer: patients' experience and doctors' judgement. Br J Urol 73: 554–560
Albers P et al. (2005) Guidelines on testicular cancer. Eur Urol 48: 885–894
Petersen PM et al. (2002) Effect of graded testicular doses of radiotherapy in patients treated for carcinoma-in-situ in the testis. J Clin Oncol 20: 1537–1543
Budgell GJ et al. (2001) Prediction of scattered dose to the testes in abdominopelvic radiotherapy. Clin Oncol 13: 120–125
Hermann RM et al. (2005) Testicular dose and hormonal changes after radiotherapy of rectal cancer. Rad Onclol 75: 83–88
Mydlo JH and Lebed B (2004) Does brachytherapy of the prostate affect sperm quality and/or fertility in younger men? Scand J Urol Nephrol 38: 221–224
Jacobsen KD et al. (1997) External beam abdominal radiotherapy in patients with seminoma stage I: field type, testicular dose, and spermatogenesis. Int J Radiat Oncol Biol Phys 38: 95–102
Aass N et al. (1991) Prediction of long-term gonadal toxicity after standard treatment for testicular cancer. Eur J Cancer 27: 1087–1091
Brydøy M et al. (2005) Paternity following treatment for testicular cancer. J Natl Cancer Inst 97: 1580–1588
Huyghe E et al. (2004) Fertility after testicular cancer treatments. Cancer 100: 732–737
Huddart RA et al. (2005) Fertility, gonadal and sexual function in survivors of testicular cancer. Br J Cancer 93: 200–207
Kinsella TJ et al. (1989) Long-term follow-up of testicular function following radiation therapy for early-stage Hodgkin`s disease. J Clin Oncol 7: 718–724
Basavaraju SR and Easterly CE (2002) Pathophysiological effects of radiation on atherosclerosis development and progression, and the incidence of cardiovascular complications. Med Phys 29: 2391–2403
Potosky AL et al. (2004) Five-year outcomes after prostatectomy or radiotherapy for prostate cancer: the prostate cancer outcomes study. J Nat Cancer Inst 96: 1358–1367
Falah M et al. (2005) Neuromuscular complications of cancer diagnosis and treatment. J Support Oncol 3: 271–282
Socié G et al. (2003) Nonmalignant late effects after allogeneic stem cell transplantation. Blood 101: 3373–3385
Hyer S et al. (2002) Testicular dose and fertility in men following I131 therapy for thyroid cancer. Clin Endocrinol 56: 755–758
Viviani S et al. (1985) Gonadal toxicity after combination chemotherapy for Hodgkin´s disease. Comparative results of MOPP vs ABVD. Eur J Cancer Oncol 21: 601–605
Lampe H et al. (1997) Fertility after chemotherapy for testicular germ cell cancers. J Clin Oncol 15: 239–245
Donohue JP et al. (1990) Nervesparing retroperitoneal lymphadenectomy with preservation of ejaculation. J Urol 144: 287–292
Shetty G and Meistrich ML (2005) Hormonal approaches to preservation and restoration of male fertility after cancer treatment. J Natl Cancer Inst Monogr 34: 36–39
Schrader M et al. (2003) “Onco-tese”: testicular sperm extraction in azoospermic cancer patients before chemotherapy—new guidelines? Urology 61: 421–425
Magelssen H et al. (2005) Twenty years experience with semen cryopreservation in testicular cancer patients: who needs it? Eur Urol 48: 779–785
Ragni G et al. (2003) Sperm banking and rate of assisted reproduction treatment: insights from a 15-year cryopreservation program for male cancer patients. Cancer 97: 1624–1629
Sherins RJ et al. (1978) Gynecomastia and gonadal dysfunction in adolescent boys treated with combination chemotherapy for Hodgkin´s disease. N Engl J Med 229: 12–16
Ben Arush MW et al. (2000) Male gonadal function in survivors of childhood Hodgkin and non-Hodgkin lymohoma. Pediatr Hematol Oncol 17: 239–245
Aslam I et al. (2000) Fertility preservation of boys undergoing anti-cancer therapy: a review of the existing situation and prospects for the future. Hum Reprod 15: 2154–2159
Brougham MFH et al. (2003) Male fertility following childhood cancer: current concepts and future therapies. Asian J Androl 5: 325–337
Rosenlund B et al. (1998) In-vitro fertilization and intracytoplasmic sperm injection in the treatment of infertility after testicular cancer. Hum Reprod 13: 414–418
Schwarzer JU et al. (2003) Sperm retrieval procedures and intracytoplasmatic spermatozoa injection with epididymal and testicular sperms. Urol Int 70: 119–123
Lombardo F et al. (2005) Androgens and fertility. J Endocrinol Invest 28 (Suppl 3): S51–S55
Kamischke A and Nieschlag E (2002) Update on medical treatment of ejaculatory disorders. Int J Androl 25: 333–344
Damani MN et al. (2002) Postchemotherapy ejaculatory azoospermia: fatherhood with sperm from testis tissue with intracytoplasmic sperm injection. J Clin Oncol 20: 930–936
Orwig KE and Schlatt S (2005) Cryopreservation and transplantation of spermatogonia and testicular tissue for preservation of male fertility. J Natl Cancer Inst Monogr 34: 51–56
Radford JA et al. (1999) Fertility after treatment for cancer. BMJ 319: 935–936
Geijsen N et al. (2004) Derivation of embryonic germ cells and male gametes from embryonic stem cells. Nature 427: 148–154
Meistrich ML and Shetty G (2003) Suppression of testosterone stimulates recovery of spermatogenesis after cancer treatment. Int J Androl 26: 141–146
Byrne J et al. (1998) Genetic disease in offspring of long-term survivors of childhood and adolescent cancer. Am J Hum Genet 62: 45–52
Giwercman A (2003) Gonadotoxic cancer treatment in males—a reason for andrological counseling? Rad Oncol 68: 213–215
Mohr BA et al. (2005) Normal, bound and nonbound testosterone levels in normally ageing men: results from the Massachusetts male ageing study. Clin Endocrinol 62: 64–73
Andersson AM et al. (1998) Longitudinal reproductive hormone profiles in infants: peak of inhibin B levels in infant boys exceeds levels in adult men. J Clin Endocrinol Metab 83: 675–681
Giwercman A and Petersen PM (2000) Cancer and male infertility. Baillieres Best Pract Res Clin Endocrinol Metab 14: 453–471
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Magelssen, H., Brydøy, M. & Fosså, S. The effects of cancer and cancer treatments on male reproductive function. Nat Rev Urol 3, 312–322 (2006). https://doi.org/10.1038/ncpuro0508
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpuro0508
This article is cited by
-
Inflammatory Modulation of miR-155 Inhibits Doxorubicin-Induced Testicular Dysfunction via SIRT1/FOXO1 Pathway: Insight into the Role of Acacetin and Bacillus cereus Protease
Applied Biochemistry and Biotechnology (2022)
-
Japan Society of Clinical Oncology Clinical Practice Guidelines 2017 for fertility preservation in childhood, adolescent, and young adult cancer patients: part 1
International Journal of Clinical Oncology (2022)
-
Protective role of N-acetylcysteine (NAC) on human sperm exposed to etoposide
Basic and Clinical Andrology (2019)
-
Semen quality of 4480 young cancer and systemic disease patients: baseline data and clinical considerations
Basic and Clinical Andrology (2016)
-
Fertility Preservation in the Male with Cancer
Current Urology Reports (2013)