Abstract
In healthy cartilage, effective weight-bearing requires a high concentration of intact aggrecan. Degradation and loss of aggrecan are features of osteoarthritis (OA). It is unclear whether ADAMTS-4, ADAMTS-5, or both of these aggrecanases from the ADAMTS (a disintegrin and metalloproteinase with thrombospondin motifs) enzyme family, are responsible for aggrecanolysis in human OA, and at what stage of disease these enzymes are active. Several potential disease-modifying agents for OA include glucosamine and chondroitin sulfate, diacerhein, and pentosan polysulfate; although their mechanisms of action in vivo are unknown, data from in vitro studies and animal models suggest that their efficacy might be partly due to inhibition of proinflammatory pathways that lead to downregulation of ADAMTS enzymes. Some histone deacetylase inhibitors that are successfully used to treat cancer can block ADAMTS-5 expression; however, these inhibitors will only be considered as potential therapies for OA if their toxicity is markedly reduced. ADAMTS inhibitors currently in development are expected to show excellent specificity now that crystal structures for several ADAMTS enzymes are available to guide drug design. ADAMTS-4 and ADAMTS-5 are appropriate targets for OA therapies, but ultimately, inhibitors of these enzymes will form only part of a larger arsenal of therapies.
Key Points
-
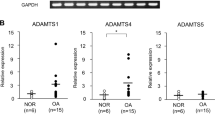
Most aggrecan fragments in the synovial fluid and cartilage of patients with osteoarthritis (OA) are the products of aggrecanase cleavage
-
The possibility that both a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS)-4 and ADAMTS-5 have key roles in human OA cannot be excluded, as changes in ADAMTS expression and activity at different stages of disease remain poorly characterized
-
Studies using ADAMTS inhibitors, or inducible ADAMTS knockout strategies, are needed to determine whether inhibiting ADAMTS activity in established disease can prevent progression of cartilage erosion
-
In humans, ADAMTS-5 is constitutively expressed in chondrocytes and synoviocytes, whereas ADAMTS-4 expression is induced by proinflammatory cytokines; this differential gene regulation has implications, and complications, for therapeutic strategies aimed at inhibiting both aggrecanases
-
Glucosamine and chondroitin sulfate, pentosan polysulfate and diacerhein might indirectly inhibit ADAMTS activity, although a direct cause-and-effect relationship between effects seen in vitro or in animal models, and efficacy in clinical studies, has not been established
-
Many therapies showing promise for the management of OA inhibit components of proinflammatory pathways that lead to altered ADAMTS activity
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Glasson SS et al. (2005) Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 434: 644–648
Stanton H et al. (2005) ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 434: 648–652
Fosang AJ et al. (2008) ADAMTS-5: the story so far. Eur Cell Mater 15: 11–26
Tortorella MD and Malfait AM (2008) Will the real aggrecanase(s) step up: evaluating the criteria that define aggrecanase activity in osteoarthritis. Curr Pharm Biotechnol 9: 16–23
Bondeson J et al. (2006) The role of synovial macrophages and macrophage-produced cytokines in driving aggrecanases, matrix metalloproteinases, and other destructive and inflammatory responses in osteoarthritis. Arthritis Res Ther 8: R187
Struglics A et al. (2006) Human osteoarthritis synovial fluid and joint cartilage contain both aggrecanase- and matrix metalloproteinase-generated aggrecan fragments. Osteoarthritis Cartilage 14: 101–113
Fosang AJ et al. (1996) Aggrecan is degraded by matrix metalloproteinases in human arthritis. Evidence that matrix metalloproteinase and aggrecanase activities can be independent. J Clin Invest 98: 2292–2299
Lark MW et al. (1997) Aggrecan degradation in human cartilage. Evidence for both metalloproteinase and aggrecanase activity in normal, osteoarthritic, and rheumatoid joints. J Clin Invest 100: 93–106
van Meurs JB et al. (1999) Kinetics of aggrecanase- and metalloproteinase-induced neoepitopes in various stages of cartilage destruction in murine arthritis. Arthritis Rheum 42: 1128–1139
Maehara H et al. (2007) G1-G2 aggrecan product that can be generated by M-calpain on truncation at Ala709-Ala710 is present abundantly in human articular cartilage. J Biochem (Tokyo) 141: 469–477
Little CB et al. (2007) Blocking aggrecanase cleavage in the aggrecan interglobular domain abrogates cartilage erosion and promotes cartilage repair. J Clin Invest 117: 1627–1636
East CJ et al. (2007) ADAMTS-5 deficiency does not block aggrecanolysis at preferred cleavage sites in the chondroitin sulphate-rich region of aggrecan. J Biol Chem 282: 8632–8640
Glasson SS et al. (2004) Characterization of and osteoarthritis susceptibility in ADAMTS-4-knockout mice. Arthritis Rheum 50: 2547–2558
Behera AK et al. (2006) Role of aggrecanase 1 in Lyme arthritis. Arthritis Rheum 54: 3319–3329
Rogerson FM et al. (2008) Identification of a novel aggrecan-degrading activity in cartilage: studies of mice deficient in both ADAMTS-4 and ADAMTS-5. Arthritis Rheum 58: 1664–1673
Appleton CT et al. (2007) Global analyses of gene expression in early experimental osteoarthritis. Arthritis Rheum 56: 1854–1868
Pelletier JP et al. (2005) The protective effect of licofelone on experimental osteoarthritis is correlated with the downregulation of gene expression and protein synthesis of several major cartilage catabolic factors: MMP-13, cathepsin K and aggrecanases. Arthritis Res Ther 7: R1091–R1102
Little CB et al. (2006) Regional changes in expression and activity of ADAMTS and MMP enzymes in cartilage in an ovine model of early osteoarthritis. Trans Orthop Res Soc 31: 1461
Bau B et al. (2002) Relative messenger RNA expression profiling of collagenases and aggrecanases in human articular chondrocytes in vivo and in vitro. Arthritis Rheum 46: 2648–2657
Plaas A et al. (2007) Aggrecanolysis in human osteoarthritis: confocal localization and biochemical characterization of ADAMTS5-hyaluronan complexes in articular cartilages. Osteoarthritis Cartilage 15: 719–734
Malfait A-M et al. (2002) Inhibition of ADAM-TS4 and ADAM-TS5 prevents aggrecan degradation in osteoarthritic cartilage. J Biol Chem 277: 22201–22208
Naito S et al. (2007) Expression of ADAMTS4 (aggrecanase-1) in human osteoarthritic cartilage. Pathol Int 57: 703–711
Roach HI et al. (2005) Association between the abnormal expression of matrix-degrading enzymes by human osteoarthritic chondrocytes and demethylation of specific CpG sites in the promoter regions. Arthritis Rheum 52: 3110–3124
Bastow ER et al. (2008) Hyaluronan synthesis and degradation in cartilage and bone. Cell Mol Life Sci 65: 395–413
Song RH et al. (2007) Aggrecan degradation in human articular cartilage explants is mediated by both ADAMTS-4 and ADAMTS-5. Arthritis Rheum 56: 575–585
Abbaszade I et al. (1999) Cloning and characterization of ADAMTS11, an aggrecanase from the ADAMTS family. J Biol Chem 274: 23443–23450
Tortorella MD et al. (1999) Purification and cloning of aggrecanase-1: a member of the ADAMTS family of proteins. Science 284: 1664–1666
Cooper C et al. (2000) Risk factors for the incidence and progression of radiographic knee osteoarthritis. Arthritis Rheum 43: 995–1000
Massardo L et al. (1989) Osteoarthritis of the knee joint: an eight year prospective study. Ann Rheum Dis 48: 893–897
Spector TD et al. (1992) Radiological progression of osteoarthritis: an 11 year follow up study of the knee. Ann Rheum Dis 51: 1107–1110
Majumdar MK et al. (2007) Double-knockout of ADAMTS-4 and ADAMTS-5 in mice results in physiologically normal animals and prevents the progression of osteoarthritis. Arthritis Rheum 56: 3670–3674
Ilic MZ et al. (2007) Distinguishing aggrecan loss from aggrecan proteolysis in ADAMTS-4 and ADAMTS-5 single and double deficient mice. J Biol Chem 282: 37420–37428
Vlad SC et al. (2007) Glucosamine for pain in osteoarthritis: why do trial results differ? Arthritis Rheum 56: 2267–2277
Reginster JY (2007) The efficacy of glucosamine sulfate in osteoarthritis: financial and nonfinancial conflict of interest. Arthritis Rheum 56: 2105–2110
Clegg DO et al. (2006) Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med 354: 795–808
Herrero-Beaumont G et al. (2007) Glucosamine sulfate in the treatment of knee osteoarthritis symptoms: a randomized, double-blind, placebo-controlled study using acetaminophen as a side comparator. Arthritis Rheum 56: 555–567
Zhang W et al. (2007) OARSI recommendations for the management of hip and knee osteoarthritis, part I: critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis Cartilage 15: 981–1000
Sandy JD et al. (1998) Chondrocyte-mediated catabolism of aggrecan: aggrecanase-dependent cleavage induced by interleukin-1 or retinoic acid can be inhibited by glucosamine. Biochem J 335: 59–66
Chan PS et al. (2006) Short-term gene expression changes in cartilage explants stimulated with interleukin beta plus glucosamine and chondroitin sulfate. J Rheumatol 33: 1329–1340
Piepoli T et al. (2005) Glucosamine sulfate inhibits IL-1-stimulated gene expression at concentrations found in humans after oral intake. Arthritis Rheum 52 (Suppl 9): S502
Chan PS et al. (2007) Effects of glucosamine and chondroitin sulfate on bovine cartilage explants under long-term culture conditions. Am J Vet Res 68: 709–715
Persiani S et al. (2007) Synovial and plasma glucosamine concentrations in osteoarthritic patients following oral crystalline glucosamine sulphate at therapeutic dose. Osteoarthritis Cartilage 15: 764–772
Ghosh P (1999) The pathobiology of osteoarthritis and the rationale for the use of pentosan polysulfate for its treatment. Semin Arthritis Rheum 28: 211–267
Takizawa M et al. (2000) Production of tissue inhibitor of metalloproteinases 3 is selectively enhanced by calcium pentosan polysulfate in human rheumatoid synovial fibroblasts. Arthritis Rheum 43: 812–820
Munteanu SE et al. (2000) Calcium pentosan polysulfate inhibits the catabolism of aggrecan in articular cartilage explant cultures. Arthritis Rheum 43: 2211–2218
Altman RD (1999) Calcium pentosan in a canine model of osteoarthritis. Osteoarthritis Cartilage 7 (Suppl A): 72
Ghosh P et al. (2005) Evaluation of the symptom-modifying effects of pentosan polysulfate in patients with knee osteoarthritis. Results of a randomized placebo-controlled double-blind study. Curr Ther Res 66: 552–571
Rintelen B et al. (2006) A meta-analysis of controlled clinical studies with diacerein in the treatment of osteoarthritis. Arch Intern Med 166: 1899–1906
Fidelix TS et al. (2006) Diacerein for osteoarthritis. Cochrane Database of Systematic Reviews 2006, Issue 1. Art. No.: CD005117. 10.1002/14651858.CD005117.pub2
Smith GNJ et al. (1999) Diacerhein treatment reduces the severity of osteoarthritis in the canine cruciate-deficiency model of osteoarthritis. Arthritis Rheum 42: 545–554
Tamura T et al. (2001) Rhein, an active metabolite of diacerein, down-regulates the production of pro-matrix metalloproteinases-1, -3, -9 and -13 and up-regulates the production of tissue inhibitor of metalloproteinase-1 in cultured rabbit articular chondrocytes. Osteoarthritis Cartilage 9: 257–263
Tamura T and Ohmori K (2001) Rhein, an active metabolite of diacerein, suppresses the interleukin-1alpha-induced proteoglycan degradation in cultured rabbit articular chondrocytes. Jpn J Pharmacol 85: 101–104
Legendre F et al. (2007) Rhein, a diacerhein-derived metabolite, modulates the expression of matrix degrading enzymes and the cell proliferation of articular chondrocytes by inhibiting ERK and JNK-AP-1 dependent pathways. Clin Exp Rheumatol 25: 546–555
Mendes AF et al. (2002) Diacerhein and rhein prevent interleukin-1beta-induced nuclear factor-kappaB activation by inhibiting the degradation of inhibitor kappaB-alpha. Pharmacol Toxicol 91: 22–28
Little CB et al. (2002) Cyclosporin A inhibition of aggrecanase-mediated proteoglycan catabolism in articular cartilage. Arthritis Rheum 46: 124–129
Yoo SA et al. (2007) Calcineurin modulates the catabolic and anabolic activity of chondrocytes and participates in the progression of experimental osteoarthritis. Arthritis Rheum 56: 2299–2311
Henry ML et al. (1995) A clinical trial of cyclosporine G in cadaveric renal transplantation. Pediatr Nephrol 9 (Suppl): S49–S51
Roach HI and Aigner T (2007) DNA methylation in osteoarthritic chondrocytes: a new molecular target. Osteoarthritis Cartilage 15: 128–137
Young DA et al. (2005) Histone deacetylase inhibitors modulate metalloproteinase gene expression in chondrocytes and block cartilage resorption. Arthritis Res Ther 7: R503–R512
Lin HS et al. (2007) Anti-rheumatic activities of histone deacetylase (HDAC) inhibitors in vivo in collagen-induced arthritis in rodents. Br J Pharmacol 150: 862–872
Gendron C et al. (2007) Proteolytic activities of human ADAMTS-5: comparative studies with ADAMTS-4. J Biol Chem 282: 18294–18306
Kashiwagi M et al. (2004) Altered proteolytic activities of ADAMTS-4 expressed by C-terminal processing. J Biol Chem 279: 10109–10119
Zeng W et al. (2006) Glycosaminoglycan-binding properties and aggrecanase activities of truncated ADAMTSs: comparative analyses with ADAMTS-5, -9, -16 and -18. Biochim Biophys Acta 1760: 517–524
Acknowledgements
The authors are grateful to Professor Stefan Lohmander for his clinical insights and improvements to the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
AJ Fosang declared grant/research support from AstraZeneca and Pfizer, Inc. CB Little declared grant/research support from Fidia Farmaceutucci, Mesoblast Inc and Pfizer, Inc.
Rights and permissions
About this article
Cite this article
Fosang, A., Little, C. Drug Insight: aggrecanases as therapeutic targets for osteoarthritis. Nat Rev Rheumatol 4, 420–427 (2008). https://doi.org/10.1038/ncprheum0841
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0841
This article is cited by
-
Expression and regulation of recently discovered hyaluronidases, HYBID and TMEM2, in chondrocytes from knee osteoarthritic cartilage
Scientific Reports (2022)
-
Synergistic upregulation of ADAMTS4 (aggrecanase-1) by cytokines and its suppression in knee osteoarthritic synovial fibroblasts
Laboratory Investigation (2022)
-
The IL-1β/AP-1/miR-30a/ADAMTS-5 axis regulates cartilage matrix degradation in human osteoarthritis
Journal of Molecular Medicine (2016)
-
The ADAMTS (A Disintegrin and Metalloproteinase with Thrombospondin motifs) family
Genome Biology (2015)
-
Novel Insights into Osteoarthritis Joint Pathology from Studies in Mice
Current Rheumatology Reports (2015)