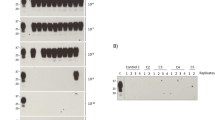
Abstract
Prion infections lead to invariably fatal diseases of the CNS, including Creutzfeldt–Jakob disease (CJD) in humans, bovine spongiform encephalopathy (BSE), and scrapie in sheep. There have been hundreds of instances in which prions have been transmitted iatrogenically among humans, usually through neurosurgical procedures or administration of pituitary tissue extracts. Prions have not generally been regarded as blood-borne infectious agents, and case–control studies have failed to identify CJD in transfusion recipients. Previous understanding was, however, questioned by reports of prion infections in three recipients of blood donated by individuals who subsequently developed variant CJD. On reflection, hematogenic prion transmission does not come as a surprise, as involvement of extracerebral compartments such as lymphoid organs and skeletal muscle is common in most prion infections, and prions have been recovered from the blood of rodents and sheep. Novel diagnostic strategies, which might include the use of surrogate markers of prion infection, along with prion removal strategies, might help to control the risk of iatrogenic prion spread through blood transfusions.
Key Points
-
Prion diseases are transmissible neurodegenerative conditions characterized by accumulation of a misfolded prion protein (PrPSc) in the brain
-
The prion diseases include Creutzfeldt–Jakob disease (CJD) in humans, bovine spongiform encephalopathy (BSE), and scrapie in sheep
-
Blood has not generally been regarded as a prion vector, but three individuals who contracted variant CJD through blood transfusion have recently been identified
-
Whole-blood fractionation experiments in animals revealed that the bulk of prion infectivity resides in leukocytes, plasma, cryoprecipitate, and Cohn fractions I–III
-
There is no effective screening method for identifying PrPSc in blood, so surrogate markers for prion diseases need to be identified in order that the quality of blood for transfusion can be controlled
-
The efficacy of filter systems for reducing prion titers in blood is currently being assessed
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Silveira JR et al. (2005) The most infectious prion protein particles. Nature 437: 257–261
Aguzzi A and Miele G (2004) Recent advances in prion biology. Curr Opin Neurol 17: 337–342
Brandner S et al. (1996) Normal host prion protein necessary for scrapie-induced neurotoxicity. Nature 379: 339–343
Mead S et al. (2003) Balancing selection at the prion protein gene consistent with prehistoric kurulike epidemics. Science 300: 640–643
Creutzfeldt HG (1920) Über eine eigenartige herdförmige Erkrankung des Zentralnervensystems. Z ges Neurol Psychiatr 57: 1–19
Aguzzi A and Polymenidou M (2004) Mammalian prion biology: one century of evolving concepts. Cell 116: 313–327
Jakob A (1921) Über eigenartige Erkrankungen des Zentralnervensystems mit bemerkenswertem anatomischem Befunde. (Spastische Pseudosklerose-Encephalomyelopathie mit disseminierten Degenerationsherden). Z ges Neurol Psychiatr 64: 147–228
Glatzel M et al. (2005) Human prion diseases: molecular and clinical aspects. Arch Neurol 62: 545–552
Ghani AC et al. (2002) The transmission dynamics of BSE and vCJD. CR Biol 325: 37–47
Hill AF et al. (1997) The same prion strain causes vCJD and BSE. Nature 389: 448–450
Bruce ME et al. (1997) Transmissions to mice indicate that 'new variant' CJD is caused by the BSE agent. Nature 389: 498–501
Aguzzi A and Weissmann C (1996) Spongiform encephalopathies: a suspicious signature. Nature 383: 666–667
Aguzzi A (1996) Between cows and monkeys. Nature 381: 734
Andrews NJ et al. (2003) Deaths from variant Creutzfeldt–Jakob disease in the UK. Lancet 361: 751–752
The National Creutzfeldt–Jakob Disease Surveillance Unit [http://www.cjd.ed.ac.uk]
Frigg R et al. (1999) Scrapie pathogenesis in subclinically infected B-cell-deficient mice. J Virol 73: 9584–9588
Hilton DA et al. (2004) Prevalence of lymphoreticular prion protein accumulation in UK tissue samples. J Pathol 203: 733–739
Peden AH et al. (2004) Preclinical vCJD after blood transfusion in a PRNP codon 129 heterozygous patient. Lancet 364: 527–529
Collins S et al. (1999) Surgical treatment and risk of sporadic Creutzfeldt–Jakob disease: a case–control study. Lancet 353: 693–697
Brown P et al. (1994) Human spongiform encephalopathy: the National Institutes of Health series of 300 cases of experimentally transmitted disease. Ann Neurol 35: 513–529
Glatzel M et al. (2003) Extraneural pathologic prion protein in sporadic Creutzfeldt–Jakob disease. N Engl J Med 349: 1812–1820
Ricketts MN et al. (1997) Is Creutzfeldt–Jakob disease transmitted in blood? Emerg Infect Dis 3: 155–163
Bruce ME et al. (2001) Detection of variant Creutzfeldt–Jakob disease infectivity in extraneural tissues. Lancet 358: 208–209
Hill AF et al. (1997) Diagnosis of new variant Creutzfeldt–Jakob disease by tonsil biopsy. Lancet 349: 99
Llewelyn CA et al. (2004) Possible transmission of variant Creutzfeldt–Jakob disease by blood transfusion. Lancet 363: 417–421
Health Protection Agency press statement: new case of variant CJD associated with blood transfusion [http://www.hpa.org.uk/hpa/news/articles/press_releases/2006/060209_cjd.htm]
Weissmann C and Aguzzi A (1997) Bovine spongiform encephalopathy and early onset variant Creutzfeldt–Jakob disease. Curr Opin Neurobiol 7: 695–700
Casalone C et al. (2004) Identification of a second bovine amyloidotic spongiform encephalopathy: molecular similarities with sporadic Creutzfeldt–Jakob disease. Proc Natl Acad Sci USA 101: 3065–3070
Heikenwalder M et al. (2005) Chronic lymphocytic inflammation specifies the organ tropism of prions. Science 307: 1107–1110
Wells GA et al. (2005) Pathogenesis of experimental bovine spongiform encephalopathy: preclinical infectivity in tonsil and observations on the distribution of lingual tonsil in slaughtered cattle. Vet Rec 156: 401–407
Buschmann A and Groschup MH (2005) Highly bovine spongiform encephalopathy-sensitive transgenic mice confirm the essential restriction of infectivity to the nervous system in clinically diseased cattle. J Infect Dis 192: 934–942
Ryder S et al. (2004) Demonstration of lateral transmission of scrapie between sheep kept under natural conditions using lymphoid tissue biopsy. Res Vet Sci 76: 211–217
Seeger H et al. (2005) Coincident scrapie infection and nephritis lead to urinary prion excretion. Science 310: 324–326
Houston F et al. (2000) Transmission of BSE by blood transfusion in sheep. Lancet 356: 999–1000
Hunter N et al. (2002) Transmission of prion diseases by blood transfusion. J Gen Virol 83: 2897–2905
Brown P et al. (1998) The distribution of infectivity in blood components and plasma derivatives in experimental models of transmissible spongiform encephalopathy. Transfusion 38: 810–816
Holada K et al. (2002) Scrapie infectivity in hamster blood is not associated with platelets. J Virol 76: 4649–4650
Gregori L et al. (2004) Partitioning of TSE infectivity during ethanol fractionation of human plasma. Biologicals 32: 1–10
DebBurman SK et al. (1997) Chaperone-supervised conversion of prion protein to its protease-resistant form. Proc Natl Acad Sci USA 94: 13938–13943
Fischer MB et al. (2000) Binding of disease-associated prion protein to plasminogen. Nature 408: 479–483
Polymenidou M et al. (2005) Coexistence of multiple PrPSc types in individuals with Creutzfeldt–Jakob disease. Lancet Neurol 4: 805–814
Bolton DC et al. (1991) Molecular location of a species-specific epitope on the hamster scrapie agent protein. J Virol 65: 3667–3675
Korth C et al. (1997) Prion (PrPSc)-specific epitope defined by a monoclonal antibody. Nature 390: 74–77
Saborio GP et al. (2001) Sensitive detection of pathological prion protein by cyclic amplification of protein misfolding. Nature 411: 810–813
Castilla J et al. (2005) Detection of prions in blood. Nat Med 11: 982–985
Miele G et al. (2001) A novel erythroid-specific marker of transmissible spongiform encephalopathies. Nat Med 7: 361–364
Maury C (2001) Is EDRF a specific marker for TSEs? Nat Med 7: 641–642
Wilson K and Ricketts MN (2004) Transfusion transmission of vCJD: a crisis avoided? Lancet 364: 477–479
Sowemimo-Coker S et al. (2005) Removal of exogenous (spiked) and endogenous prion infectivity from red cells with a new prototype of leukoreduction filter. Transfusion 45: 1839–1844
Schoch G et al. (2006) Analysis of prion strains by PrPSc profiling in sporadic Creutzfeldt–Jakob disease. PLoS Med 3: e14
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Aguzzi, A., Glatzel, M. Prion infections, blood and transfusions. Nat Rev Neurol 2, 321–329 (2006). https://doi.org/10.1038/ncpneuro0214
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpneuro0214
This article is cited by
-
A case-control study of sporadic Creutzfeldt-Jakob disease in Switzerland: analysis of potential risk factors with regard to an increased CJD incidence in the years 2001–2004
BMC Public Health (2009)
-
Wound healing and the immune response in swine treated with a hemostatic bandage composed of salmon thrombin and fibrinogen
Journal of Materials Science: Materials in Medicine (2009)
-
Prion biology: the quest for the test
Nature Methods (2007)
-
Insights into prion strains and neurotoxicity
Nature Reviews Molecular Cell Biology (2007)
-
Good and bad news about transmissible spongiform encephalopathies (prion diseases)
Current Neurology and Neuroscience Reports (2007)