Abstract
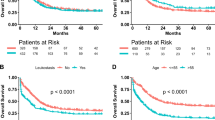
Leukocytapheresis is a controversial nonpharmacologic treatment for IBD, in which white blood cells—the effector cells of the inflammatory process—are mechanically removed from the circulation. Current controversy centers on the uncontrolled nature of the leukocytapheresis trials performed and their use of different outcome measures in patient groups that have very variable disease activity and severity. Nonetheless, the efficacy data obtained are generally quite consistent: an excellent response (remission >80%) has been achieved in corticosteroid-naïve patients with ulcerative colitis and an average remission rate of more than 50% has been achieved in patients who have steroid-dependent or refractory ulcerative colitis. Interestingly, the largest randomized, double-blind, sham-controlled study of granulocyte–monocyte apheresis in patients with moderate to severe ulcerative colitis failed to demonstrate efficacy for the induction of clinical remission or response. Regardless, leukocytapheresis seems to be remarkably safe. The precise positioning of leukocytapheresis in the treatment of ulcerative colitis is uncertain at present and will vary according to geography and patient preference for a safe, nonpharmacologic treatment. Further efficacy studies are required to assess what the optimal number and frequency of treatments is, in addition to the need for head-to-head comparisons with established drugs.
Key Points
-
Enthusiasm for the use of leukocytapheresis in the treatment of patients with IBD (mainly ulcerative colitis) is fueled by the lack of serious adverse effects and tempered by the lack of conventional placebo-controlled data or head-to-head comparisons with potential competitors
-
Leukocytapheresis induces clinical remission in more than 80% of corticosteroid-naïve patients who have ulcerative colitis
-
Average remission rates are in excess of 50% in patients with moderate or severe ulcerative colitis and in patients with corticosteroid-dependent or corticosteroid-resistant ulcerative colitis
-
Data in Crohn's disease are sparse and further studies are required
-
Leukocyte apheresis clearly has potential use in patients with ulcerative colitis, but to decide its precise positioning in treatment algorithms will require targeted studies
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bjarnason I et al. (1993) Non-steroidal anti-inflammatory drugs and Crohn's disease. In Inflammatory Bowel Diseases: Pathophysiology as Basis of Treatment, 208–222 (Eds Scholmeric J) Falk Symposium No 67. Lancaster: Kluwer Academic Publishers
Tibble J et al. (2000) Surrogate markers of intestinal inflammation are predictive for relapse in patients with inflammatory bowel disease. Gastroenterology 119: 15–22
Kozuch PL and Hanauer SB (2008) Treatment of inflammatory bowel disease: a review of medical therapy. World J Gastroenterol 14: 354–377
Yamamoto-Furusho JK (2007) Innovative therapeutics for inflammatory bowel disease. World J Gastroenterol 13: 1893–1896
Brown SL et al. (2002) Tumor necrosis factor antagonist therapy and lymphoma development: twenty-six cases reported to the Food and Drug Administration. Arthritis Rheum 46: 3151–3158
Gomez-Reino JJ et al. (2003) Treatment of rheumatoid arthritis with tumor necrosis factor inhibitors may predispose to significant increase in tuberculosis risk: a multicenter active-surveillance report. Arthritis Rheum 48: 2122–2127
Sandborn WJ et al. (2005) Natalizumab induction and maintenance therapy for Crohn's disease. N Engl J Med 353: 1912–1925
Seegers D et al. (2002) Review article: a critical approach to new forms of treatment of Crohn's disease and ulcerative colitis. Aliment Pharmacol Ther 16 (Suppl 4): 53–58
Morse EE et al. (1966) Repeated leukapheresis of patients with chronic myelocytic leukemia. Transfusion 6: 175–182
Buckner D et al. (1969) Leukapheresis by continous flow centrifugation (CFC) in patients with chronic myelocytic leukemia (CML). Blood 33: 353–369
Curtis JE et al. (1972) Leukoapheresis therapy of chronic lymphocytic leukemia. Blood 39: 163–175
Ohara M et al. (1997) Granulocytapheresis in the treatment of patients with rheumatoid arthritis. Artif Organs 2: 989–994
Shimoyama T et al. (2001) Safety and efficacy of granulocyte and monocyte adsorption apheresis in patients with active ulcerative colitis: a multicenter study. J Clin Apher 16: 1–9
Lazarus HM (1991) Selective in vivo removal of rheumatoid factor by an extracorporeal treatment device in rheumatoid arthritis patients. Transfusion 31: 122–128
Bicks RO et al. (1985) The treatment of severe chronically active Crohn's disease by T8 (suppressor cell) lymphapheresis. Gastroenterology 88: A1325
Bicks RO et al. (1987) Total parenteral nutrition (TPN) plus T-lymphocyte apheresis (TLA) in the treatment of severe chronic active Crohn's disease. Gastroenterology 94: A34
Sato T KN et al. (1991) Improved on-line thoracic duct drainage for lymphocytapheresis. Int J Artif Organs 14: 800–804
Hidaka T and Suzuki K (1997) Efficacy of filtration leukocytapheresis on rheumatoid arthritis with vasculitis. Ther Apher 1: 212–214
Onuma S et al. (2006) Investigation of the clinical effect of large volume leukocytapheresis on methotrexate-resistant rheumatoid arthritis. Ther Apher Dial 10: 404–411
Kashiwagi N et al. (1998) A role for granulocyte and monocyte apheresis in the treatment of rheumatoid arthritis. Ther Apher 2: 134–141
Muratov V et al. (2006) Downregulation of interferon-γ parallels clinical response to selective leukocyte apheresis in patients with inflammatory bowel disease: a 12-month follow-up study. Int J Colorectal Dis 21: 493–504
Saniabadi AR et al. (2007) Therapeutic leukocytapheresis for inflammatory bowel disease. Transfus Apher Sci 37: 191–200
Hanai H et al. (2008) Adsorptive depletion of elevated proinflammatory CD14+CD16+DR++ monocytes in patients with inflammatory bowel disease. Am J Gastroenterol 103: 1213–1216
Saniabadi AR et al. (2005) Adacolumn for selective leukocytapheresis as a non-pharmacological treatment for patients with disorders of the immune system: an adjunct or an alternative to drug therapy. J Clin Apher 3: 171–184
Saniabadi AR et al. (2003) Adacolumn, an adsorptive carrier based granulocyte and monocyte apheresis device for the treatment of inflammatory and refractory diseases associated with leukocytes. Ther Apher Dial 1: 48–59
Hanai H et al. (2002) Therapeutic efficacy of granulocyte and monocyte adsorption apheresis in severe active ulcerative colitis. Dig Dis Sci 47: 2349–2353
Yokoyama Y et al. (2007) Demonstration of low-regulatory CD25High+CD4+ and high-pro-inflammatory CD28−CD4+ T-cell subsets in patients with ulcerative colitis: modified by selective granulocyte and monocyte adsorption apheresis. Dig Dis Sci 52: 2725–2731
Aoki H et al. (2007) Adacolumn selective leukocyte adsorption apheresis in patients with active ulcerative colitis: clinical efficacy, effects on plasma IL-8 and the expression of Toll-like receptor 2 on granulocytes. Dig Dis Sci 52: 1427–1433
Hanai H et al. (2006) Effects of Adacolumn selective leukocytapheresis on plasma cytokines during active disease in patients with ulcerative colitis. World J Gastroenterol 12: 3393–3399
Hanai H et al. (2004) Correlation of serum soluble TNF-alpha receptors I and II levels with disease activity in patients with ulcerative colitis. Am J Gastroenterol 99: 1532–1538
Yamamoto T et al. (2006) Impact of selective leukocytapheresis on mucosal inflammation and ulcerative colitis: cytokine profiles and endoscopic findings. Inflamm Bowel Dis 12: 719–726
Lerebours E et al. (1994) Treatment of Crohn's disease by lymphocyte apheresis: a randomized controlled trial. Groupe d'Etudes Thérapeutiques des Affections Inflammatoires Digestives. Gastroenterology 107: 357–361
Ayabe T et al. (1997) A pilot study of centrifugal leukocyte apheresis for corticosteroid-resistant active ulcerative colitis. Intern Med 36: 322–326
Ayabe T et al. (1998) Centrifugal leukocyte apheresis for ulcerative colitis. Ther Apher 2: 125–128
Kohgo Y et al. (2002) Leukocyte apheresis using a centrifugal cell separator in refractory ulcerative colitis; A multicenter open label trial. Ther Apher 6: 255–260
Sawada K et al. (2003) Multicenter randomized controlled trial for the treatment of ulcerative colitis with a leukocytapheresis column. Curr Pharm 9: 307–321
Kanke K et al. (2004) Clinical evaluation of granulocyte/monocyte apheresis therapy for active ulcerative colitis. Dig Liver Dis 36: 811–817
Kanai T et al. (2006) The logics of leukocytapheresis as a natural biological therapy for inflammatory bowel disease. Expert Opin Biol Ther 6: 453–466
Hanai H et al. (2003) Leukocyte adsorptive apheresis for the treatment of active ulcerative colitis: a prospective, uncontrolled, pilot study. Clin Gastroenterol Hepatol 1: 28–35
Hanai H (2006) Positions of selective leukocytapheresis in the medical therapy of ulcerative colitis. World J Gastroenterol 12: 7568–7577
Naganuma M et al. (2004) Granulocytapheresis is useful as an alternative therapy in patients with steroid-refractory or -dependent ulcerative colitis. Inflamm Bowel Dis 10: 251–257
Tomomasa T et al. (2003) Granulocyte adsorptive apheresis for pediatric patients with ulcerative colitis. Dig Dis Sci 48: 750–754
Yamamoto T et al. (2004) Granulocyte and monocyte adsorptive apheresis in the treatment of active distal ulcerative colitis: a prospective, pilot study. Aliment Pharmacol Ther 20: 783–792
Honma T et al. (2005) Leukocytapheresis is effective in inducing but not in maintaining remission in ulcerative colitis. J Clin Gastroenterol 39: 886–890
Domenech E et al. (2004) Granulocyteaphaeresis in steroid-dependent inflammatory bowel disease: a prospective, open, pilot study. Aliment Pharmacol Ther 20: 1347–1352
Ljung T et al. (2007) Granulocyte, monocyte/macrophage apheresis for inflammatory bowel disease: the first 100 patients treated in Scandinavia. Scand J Gastroenterol 42: 221–227
D'Ovidio V et al. (2006) Mucosal features and granulocyte-monocyte-apheresis in steroid-dependent/refractory ulcerative colitis. Dig Liver Dis 38: 389–394
Hanai H et al. (2004) Adsorptive granulocyte and monocyte apheresis versus prednisolone in patients with corticosteroid-dependent moderately severe ulcerative colitis. Digestion 70: 36–44
Fukuda Y et al. (2004) Adsorptive granulocyte and monocyte apheresis for refractory Crohn's disease: an open multicenter prospective study. J Gastroenterol 39: 1158–1164
Giampaolo B et al. (2006) Treatment of active steroid-refractory inflammatory bowel diseases with granulocytapheresis: our experience with a prospective study. World J Gastroenterol 12: 2201–2204
Suzuki Y et al. (2006) A retrospective search for predictors of clinical response to selective granulocyte and monocyte apheresis in patients with ulcerative colitis. Dig Dis Sci 51: 2031–2038
Suzuki Y et al. (2004) Selective granulocyte and monocyte adsorptive apheresis as a first-line treatment for steroid naive patients with active ulcerative colitis: a prospective uncontrolled study. Dig Dis Sci 49: 565–571
Sakuraba A et al. (2005) A multicenter randomized controlled trial between weekly and semiweekly treatment with granulocyte and monocyte adsorption apheresis for active ulcerative colitis. Gut 54 (Suppl VII): A57
Adacolumn [http://www.adacolumn.com/]
Tanaka T et al. (2007) Adsorptive granulocytapheresis reduces mucosal level of neutrophils in patients with active ulcerative colitis and is most effective in steroid naïve patients. Gastroenterology 132 (Suppl S1): A347
Cohen RD (2005) Treating ulcerative colitis without medications—“Look Mom, No Drugs! Gastroenterology 128: 235–236
Yamamoto T et al. (2007) Factors affecting clinical and endoscopic efficacies of selective leucocytapheresis for ulcerative colitis. Dig Liver Dis 39: 626–633
Sawada K (2002) Therapeutic cytapheresis for ulcerative colitis. Medical Tribune Inc 133–149
Sawada K et al. (2005) Leukocytapheresis in ulcerative colitis: results of a multicenter double-blind prospective case-control study with sham apheresis as placebo treatment. Am J Gastroenterol 100: 1362–1369
Sands BE et al. (2008) A randomized, double-blind, sham-controlled study of granulocyte/monocyte apheresis for active ulcerative colitis. Gastroenterology [10.1053/j.gastro.2008.04.023]
Maiden L et al. (2008) Selective white cell apheresis reduces relapse rates in patients with IBD at significant risk of clinical relapse. Inflamm Bowel Dis [10.1002/ibd.20505]
Kruis W et al. (2005) Open label trial of granulocyte apheresis suggests therapeutic efficacy in chronically active steroid refractory ulcerative colitis. World J Gastroenterol 11: 7001–7005
Domenech E et al. (2004) Granulocyteaphaeresis in steroid-dependent inflammatory bowel disease: a prospective, open, pilot study. Aliment Pharmacol Ther 20: 1347–1352
Suzuki Y (2002) Efficacy of granulocyte adsorption apheresis with G-1 column in the treatment of patients with active ulcerative colitis. Medical Tribune Inc 371–381
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
During the past 2 years, Ingvar Bjarnason has been on the speakers' bureau for, and received grant/research support from, Otsuka. Fridrik T Sigurbjörnsson declared no competing interests.
Rights and permissions
About this article
Cite this article
Sigurbjörnsson, F., Bjarnason, I. Leukocytapheresis for the treatment of IBD. Nat Rev Gastroenterol Hepatol 5, 509–516 (2008). https://doi.org/10.1038/ncpgasthep1209
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncpgasthep1209
This article is cited by
-
A high-throughput microfluidic device based on controlled incremental filtration to enable centrifugation-free, low extracorporeal volume leukapheresis
Scientific Reports (2022)
-
Evidence-based clinical practice guidelines for inflammatory bowel disease 2020
Journal of Gastroenterology (2021)
-
Evidence-based clinical practice guidelines for inflammatory bowel disease
Journal of Gastroenterology (2018)
-
Successful treatment of ulcerative colitis complicated by Sweet’s syndrome by corticosteroid therapy and leukocytapheresis
Clinical Journal of Gastroenterology (2011)