Abstract
An increase in coagulation in patients with pancreatic carcinoma has long been documented. In this Review, we present what is known about the pathophysiology of increased coagulation in cancer and how it applies to pancreatic carcinoma. The relationship between the activation of coagulation or symptomatic thromboembolic disease and the development of pancreatic carcinoma is explored. Data on the relationship between thromboembolic disease and the behavior of pancreatic cancer before, during or after a diagnosis is made are also reviewed. Finally, the rationale and evidence for the use of oral anticoagulants or heparin in patients with pancreatic carcinoma is presented. This Review is a critical appraisal of what is known, and when the evidence is acceptable, on the subject of thromboembolism, anticoagulation, and treatment with anticoagulants in patients with pancreatic carcinoma.
Key Points
-
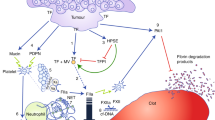
Pancreatic carcinoma is associated with activation of coagulation, and interplay between activation of coagulation, inflammation and angiogenesis seems to affect the morbidity and mortality of patients with pancreatic carcinoma
-
Thromboembolism predicts an increased risk for the development of an occult cancer, including a pancreatic carcinoma
-
Thromboembolic events are associated with a poorer prognosis in patients with pancreatic cancer than in those with other types of cancer
-
Recurrence of thromboembolism in patients with pancreatic carcinoma predicts reduced duration of survival
-
Use of either warfarin or low molecular weight heparin seems to improve survival in patients with pancreatic carcinoma
-
Two large multicenter studies on the use of low molecular weight heparin plus chemotherapy in patients with pancreatic carcinoma are under way
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Moore MJ et al. (2007) Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 25: 1960–1966
Löhr M (2006) Is it possible to survive pancreatic cancer? Nat Clin Pract Gastroenterol Hepatol 3: 236–237
Trousseau A (1865) Phlegmasia alba dolens. Clinique Medicale de l'Hotel-Dieu. Paris: Bailliere JB 3: 654–712
Bariety M (1947) Trousseau, 1801–67. Geneva: Mazenod, 234–235
Khorana AA and Fine RL (2004) Pancreatic cancer and thromboembolic disease. Lancet Oncol 5: 655–663
Sproul E (1938) Carcinoma and venous thrombosis: the frequency of association of carcinoma in the body or tail of the pancreas with multiple venous thrombosis. Amer J Cancer 34: 566–585
Pinzon R et al. (1986) Pancreatic carcinoma and Trousseau's syndrome: experience at a large cancer center. J Clin Oncol 4: 509–514
Virchow RLK (1856) Gesammelte Abhandlungen zur Wissenschaftlichen Medicin. Frankfurt, Meidinger: Sohn & Co. (Reprint edition: Virchow R (1998) Thrombosis and Emboli (1846–1856). AC Matzdorff, WR Bell, transl, Canton, MA: Science History Publications
Brotman DJ et al. (2004) Virchow's triad revisited. South Med J 97: 213–214
Haas SL et al. (2006) Expression of tissue factor in pancreatic adenocarcinoma is associated with activation of coagulation. World J Gastroenterol 12: 4843–4849
Khorana AA et al. (2007) Tissue factor expression, angiogenesis, and thrombosis in pancreatic cancer. Clin Cancer Res 13: 2870–2875
Wojtukiewicz MZ et al. (2000) Expression of prothrombin fragment 1 + 2 in cancer tissue as an indicator of local activation of blood coagulation. Thromb Res 97: 335–342
Rudroff C et al. (2002) Expression of the thrombin receptor PAR-1 correlates with tumour cell differentiation of pancreatic adenocarcinoma in vitro. Clin Exp Metastasis 19: 181–189
Wojtukiewicz M.Z et al. (2001) Localization of blood coagulation factors in situ in pancreatic carcinoma. Thromb Haemost 86: 1416–1420
Kakkar A et al. (1995) Tissue factor expression correlates with histological grade in human pancreatic cancer. Br J Surg 82: 1101–1104
Nitori N et al. (2005) Prognostic significance of tissue factor in pancreatic ductal adenocarcinoma. Clin Cancer Res 11: 2531–2539
Ueda C et al. (2001) Pancreatic cancer complicated by disseminated intravascular coagulation associated with production of tissue factor. J Gastroenterol 36: 848–850
Lindahl AK et al. (1992) Coagulation inhibition and activation in pancreatic cancer. Changes during progress of disease. Cancer 70: 2067–2072
Wojtukiewicz M et al. (2003) Immunohistochemical localization of tissue factor pathway inhibitor-2 in human tumor tissue. Thromb Haemost 90: 140–146
Chaturvedi P et al. (2007) MUC4 mucin potentiates pancreatic tumor cell proliferation, survival, and invasive properties and interferes with its interaction to extracellular matrix proteins. Mol Cancer Res 5: 309–320
Lindahl AK et al. (1993) Increased plasma thrombomodulin in cancer patients. Thromb Haemost 69: 112–114
Marguerie GA et al. (1984) The platelet–fibrinogen interaction. Evidence for proximity of the A alpha chain of fibrinogen to platelet membrane glycoproteins IIb/III. Eur J Biochem 139: 5–11
Isenberg JS et al. (2008) Thrombospondin-1 stimulates platelet aggregation by blocking the antithrombotic activity of nitric oxide/cGMP signaling. Blood 111: 613–623
Tobita K et al. (2002) Thrombospondin-1 expression as a prognostic predictor of pancreatic ductal carcinoma. Int J Oncol 21: 1189–1195
Wahrenbrock M (2003) Selectin–mucin interactions as a probable molecular explanation for the association of Trousseau syndrome with mucinous adenocarcinomas. J Clin Invest 112: 853–862
Kakkar AK (1998) A characterization of the coagulant and fibrinolytic profile of human pancreatic carcinoma cells. Haemostasis 28: 1–6
Andren-Sandberg A et al. (1992) Peaks in plasma plasminogen activator inhibitor-1 concentration may explain thrombotic events in cases of pancreatic carcinoma. Cancer 69: 2884–2887
Jesnowski R et al. (2007) Genes and proteins differentially expressed during in vitro malignant transformation of bovine pancreatic duct cells. Neoplasia 9: 136–146
Yu JL et al. (2005) Oncogenic events regulate tissue factor expression in colorectal cancer cells: implications for tumor progression and angiogenesis. Blood 105: 1734–1741
Redston MS et al. (1994) p53 mutations in pancreatic carcinoma and evidence of common involvement of homocopolymer tracts in DNA microdeletions. Cancer Res 54: 3025–3033
Allgayer H et al. (1999) Targeted disruption of the K-ras oncogene in an invasive colon cancer cell line down-regulates urokinase receptor expression and plasminogen-dependent proteolysis. Br J Cancer 80: 1884–1891
Fernandez PM et al. (2004) Tissue factor and fibrin in tumor angiogenesis. Semin Thromb Hemost 30: 31–44
Ruf W and Mueller BM (2006) Thrombin generation and the pathogenesis of cancer. Semin Thromb Hemost 32 (Suppl 1): S61–S68
Tsopanoglou NE and Maragoudakis ME (2004) Role of thrombin in angiogenesis and tumor progression. Semin Thromb Hemost 30: 63–69
Hobbs JE et al. (2007) Alternatively spliced human tissue factor promotes tumor growth and angiogenesis in a pancreatic cancer tumor model. Thromb Res 120 (Suppl 2): S13–S21
Alvarez RH et al. (2006) Biology of platelet-derived growth factor and its involvement in disease. Mayo Clin Proc 81: 1241–1257
Hwang RF et al. (2003) Inhibition of platelet-derived growth factor receptor phosphorylation by STI571 (Gleevec) reduces growth and metastasis of human pancreatic carcinoma in an orthotopic nude mouse model. Clin Cancer Res 9: 6534–6544
Löhr M et al. (2001) Transforming growth factor-beta1 induces desmoplasia in an experimental model of human pancreatic carcinoma. Cancer Res 61: 550–555
Kleeff J et al. (2007) Pancreatic cancer microenvironment. Int J Cancer 121: 699–705
Fukunaga A et al. (2004) CD8+ tumor-infiltrating lymphocytes together with CD4+ tumor-infiltrating lymphocytes and dendritic cells improve the prognosis of patients with pancreatic adenocarcinoma. Pancreas 28: e26–e31
Albo D et al. (1999) Thrombospondin-1 and transforming growth factor beta-1 upregulate plasminogen activator inhibitor type 1 in pancreatic cancer. J Gastrointest Surg 3: 411–417
Fareed D et al. (2004) Blood levels of nitric oxide, C-reactive protein, and tumor necrosis factor-alpha are upregulated in patients with malignancy-associated hypercoagulable state: pathophysiologic implications. Clin Appl Thromb Hemost 10: 357–364
Scarpati EM and Sadler JE (1989) Regulation of endothelial cell coagulant properties. Modulation of tissue factor, plasminogen activator inhibitors, and thrombomodulin by phorbol 12-myristate 13-acetate and tumor necrosis factor. J Biol Chem 264: 20705–20713
Esposito I et al. (2004) Inflammatory cells contribute to the generation of an angiogenic phenotype in pancreatic ductal adenocarcinoma. J Clin Pathol 57: 630–636
Rudroff C et al. (2001) Thrombin enhances adhesion in pancreatic cancer in vitro through the activation of the thrombin receptor PAR 1. Eur J Surg Oncol 27: 472–476
Gunji Y and Gorelik E (1988) Role of fibrin coagulation in protection of murine tumor cells from destruction by cytotoxic cells. Cancer Res 48: 5216–5221
Palumbo JS et al. (2000) Fibrinogen is an important determinant of the metastatic potential of circulating tumor cells. Blood 96: 3302–3309
He Y et al. (2007) Interaction between cancer cells and stromal fibroblasts is required for activation of the uPAR-uPA-MMP-2 cascade in pancreatic cancer metastasis. Clin Cancer Res 13: 3115–3124
Qian X et al. (1997) Thrombospondin-1 modulates angiogenesis in vitro by up-regulation of matrix metalloproteinase-9 in endothelial cells. Exp Cell Res 235: 403–412
Qian X et al. (2001) Expression of thrombospondin-1 in human pancreatic adenocarcinomas: role in matrix metalloproteinase-9 production. Pathol Oncol Res 7: 251–259
Palumbo JS et al. (2005) Platelets and fibrin(ogen) increase metastatic potential by impeding natural killer cell-mediated elimination of tumor cells. Blood 105: 178–185
Monreal, M et al. (1991) Occult cancer in patients with deep venous thrombosis. A systematic approach. Cancer 67: 541–545
Prandoni P et al. (1992) Deep-vein thrombosis and the incidence of subsequent symptomatic cancer. N Engl J Med 327: 1128–1133
Monreal M et al. (1997) Occult cancer in patients with venous thromboembolism: which patients, which cancers. Thromb Haemost 78: 1316–1318
White RH et al. (2005) Incidence of venous thromboembolism in the year before the diagnosis of cancer in 528,693 adults. Arch Intern Med 165: 1782–1787
Sorensen HT et al. (2000) Prognosis of cancers associated with venous thromboembolism. N Engl J Med 343: 1846–1850
Iodice S et al. (2008) Venous thromboembolic events and organ-specific occult cancers: a review and meta-analysis. J Thromb Haemost 6: 781–788
Mandala M et al. (2007) Venous thromboembolism predicts poor prognosis in irresectable pancreatic cancer patients. Ann Oncol 18: 1660–1665
Khorana AA et al. (2006) Thromboembolism in hospitalized neutropenic cancer patients. J Clin Oncol 24: 484–490
Chew HK et al. (2006) Incidence of venous thromboembolism and its effect on survival among patients with common cancers. Arch Intern Med 166: 458–464
Hirsh J et al. (2003) American Heart Association/American College of Cardiology Foundation guide to warfarin therapy. Circulation 107: 1692–1711
Hirsh J et al. (2001) Heparin and low-molecular-weight heparin: mechanisms of action, pharmacokinetics, dosing, monitoring, efficacy, and safety. Chest 119 (Suppl 1): S64–S94
Norrby K (2006) Low-molecular-weight heparins and angiogenesis. Apmis 114: 79–102
Miller GJ et al. (2004) Increased incidence of neoplasia of the digestive tract in men with persistent activation of the coagulant pathway. J Thromb Haemost 2: 2107–2114
Folkman J et al. (1983) Angiogenesis inhibition and tumor regression caused by heparin or a heparin fragment in the presence of cortisone. Science 221: 719–725
Pross M et al. (2004) Effect of low molecular weight heparin on intra-abdominal metastasis in a laparoscopic experimental study. Int J Colorectal Dis 19: 143–146
Schwalke MA et al. (1990) Effects of prostacyclin on hepatic metastases from human pancreatic cancer in the nude mouse. J Surg Res 49: 164–167
Schulman S and Lindmarker P (2000) Incidence of cancer after prophylaxis with warfarin against recurrent venous thromboembolism. Duration of Anticoagulation Trial. N Engl J Med 342: 1953–1958
Schulman S et al. (2006) Post-thrombotic syndrome, recurrence, and death 10 years after the first episode of venous thromboembolism treated with warfarin for 6 weeks or 6 months. J Thromb Haemost 4: 734–742
Taliani MR et al. (2003) Incidence of cancer after a first episode of idiopathic venous thromboembolism treated with 3 months or 1 year of oral anticoagulation. J Thromb Haemost 1: 1730–1733
Zacharski LR et al. (1984) Effect of warfarin anticoagulation on survival in carcinoma of the lung, colon, head and neck, and prostate. Final report of VA Cooperative Study #75. Cancer 53: 2046–2052
Zacharski LR and Henderson WG (2002) A rebuttal: vitamin K antagonists and cancer survival. Thromb Haemost 88: 173–175
Maurer LH et al. (1997) Randomized trial of chemotherapy and radiation therapy with or without warfarin for limited-stage small-cell lung cancer: a Cancer and Leukemia Group B study. J Clin Oncol 15: 3378–3387
Kuderer NM et al. (2007) A meta-analysis and systematic review of the efficacy and safety of anticoagulants as cancer treatment: impact on survival and bleeding complications. Cancer 110: 1149–1161
Nakchbandi W et al. (2006) Effects of low-dose warfarin and regional chemotherapy on survival in patients with pancreatic carcinoma. Scand J Gastroenterol 41: 1095–1104
Nakchbandi W et al. Prospective study on warfarin and regional chemotherapy in patients with pancreatic carcinoma. J Gastrointestin Liver Dis, in press
Lee AY et al. (2003) Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med 349: 146–153
Ruf W (2007) Tissue factor and PAR signaling in tumor progression. Thromb Res 120 (Suppl 2): S7–S12
Jung SP (2001) Inhibition of human angiogenesis with heparin and hydrocortisone. Angiogenesis 4: 175–186
Kiricuta I et al. (1973) Prophylaxis of metastases formation by unspecific immunologic stimulation associated with heparintherapy. Cancer 31: 1392–1396
Owen CA Jr (1982) Anticoagulant treatment of rats with Walker 256 carcinosarcoma. J Cancer Res Clin Oncol 104: 191–193
Green D et al. (1992) Lower mortality in cancer patients treated with low-molecular-weight versus standard heparin. Lancet 339: 1476
Khorana AA et al. (2003) Heparin inhibition of endothelial cell proliferation and organization is dependent on molecular weight. Arterioscler Thromb Vasc Biol 23: 2110–2115
Lee AY et al. (2005) Randomized comparison of low molecular weight heparin and coumarin derivatives on the survival of patients with cancer and venous thromboembolism. J Clin Oncol 23: 2123–2129
Icli F et al. (2007) Low molecular weight heparin (LMWH) increases the efficacy of cisplatinum plus gemcitabine combination in advanced pancreatic cancer. J Surg Oncol 95: 507–512
von Delius S et al. (2007) Effect of low-molecular-weight heparin on survival in patients with advanced pancreatic adenocarcinoma. Thromb Haemost 98: 434–439
Pelzer U et al. (2006) A prospective randomized trial of simultaneous pancreatic cancer treatment with enoxaparin and chemotherapy (PROSPECT CONKO 004). 2006 ASCO Annual Meeting Proceedings. J Clin Oncol 24 (Suppl 18): S4110
Maraveyas A. Gemcitabine and dalteparin or gemcitabine alone for advanced pancreatic cancer (FRAGEM). [http://www.cancerhelp.org.uk/trials/trials/] (to find, search for pancreatic cancer trials; accessed 16 May 2008)
Acknowledgements
This work was supported in part by the Max-Planck Society (IAN), the German Cancer Research Center (JML), and a research fund from the Dr Hans & Lore Graf Stiftung (JML).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Nakchbandi, I., Löhr, JM. Coagulation, anticoagulation and pancreatic carcinoma. Nat Rev Gastroenterol Hepatol 5, 445–455 (2008). https://doi.org/10.1038/ncpgasthep1184
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncpgasthep1184
This article is cited by
-
Gadofullerene nanoparticles extend survival rate and down-regulate thrombin expression in orthotopic pancreatic cancer
Science China Materials (2022)
-
Subclinical Inflammation and Endothelial Dysfunction in Patients with Chronic Pancreatitis and Newly Diagnosed Pancreatic Cancer
Digestive Diseases and Sciences (2016)
-
Pulmonary Embolism and Stroke as the Initial Manifestation of Advanced Metastatic Pancreatic Adenocarcinoma
Journal of Gastrointestinal Cancer (2015)
-
Clinical Significance of Coagulation Assays in Metastatic Pancreatic Adenocarcinoma
Journal of Gastrointestinal Cancer (2013)
-
Rationale and design of PROSPECT-CONKO 004: a prospective, randomized trial of simultaneous pancreatic cancer treatment with enoxaparin and chemotherapy)
BMC Cancer (2008)