Abstract
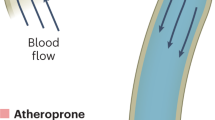
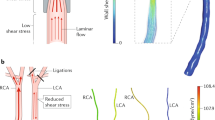
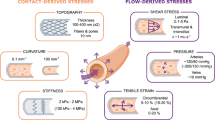
Endothelium lining the cardiovascular system is highly sensitive to hemodynamic shear stresses that act at the vessel luminal surface in the direction of blood flow. Physiological variations of shear stress regulate acute changes in vascular diameter and when sustained induce slow, adaptive, structural-wall remodeling. Both processes are endothelium-dependent and are systemically and regionally compromised by hyperlipidemia, hypertension, diabetes and inflammatory disorders. Shear stress spans a range of spatiotemporal scales and contributes to regional and focal heterogeneity of endothelial gene expression, which is important in vascular pathology. Regions of flow disturbances near arterial branches, bifurcations and curvatures result in complex spatiotemporal shear stresses and their characteristics can predict atherosclerosis susceptibility. Changes in local artery geometry during atherogenesis further modify shear stress characteristics at the endothelium. Intravascular devices can also influence flow-mediated endothelial responses. Endothelial flow-induced responses include a cell-signaling repertoire, collectively known as mechanotransduction, that ranges from instantaneous ion fluxes and biochemical pathways to gene and protein expression. A spatially decentralized mechanism of endothelial mechanotransduction is dominant, in which deformation at the cell surface induced by shear stress is transmitted as cytoskeletal tension changes to sites that are mechanically coupled to the cytoskeleton. A single shear stress mechanotransducer is unlikely to exist; rather, mechanotransduction occurs at multiple subcellular locations.
Key Points
-
Hemodynamic forces, and in particular shear stresses, are regulators of many physiologic and pathologic aspects of endothelial function in the cardiovascular system
-
In vivo and in vitro global endothelial analyses reveal that endothelial phenotypes are heterogeneous over regional and focal length scales, which links flow characteristics to cardiovascular disease protection, susceptibility and development
-
Endothelial responses are sensitive to variations in the characteristics of flow that generate shear stresses; regions with oscillating shear stress and flow reversal correspond with pathologic changes in the artery wall and are a risk factor for atherosclerosis-susceptibility
-
When shear stresses deform the endothelium, a mechanical perturbation is communicated via the cytoskeleton to multiple sites of mechanotransduction, which include cell–matrix adhesion sites, intercellular junctions and the nuclear membrane
-
Endothelial responses that are specific to shear stress offer potential therapeutic pharmacological targets, although a single mechanosensor is unlikely to exist
-
Beneficial systemic effects include maintenance of arterial hemodynamics within normal limits through antihypertensive therapies, regular exercise to promote continuous adaptive remodeling and inhibition of endothelial dysfunction, and (when intervention is required) better design of intravascular devices to optimize flow characteristics
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Pohl U et al. (1986) Crucial role of endothelium in the vasodilator response to increased flow in vivo. Hypertension 8: 37–44
Moncada S (2006) Adventures in vascular biology: a tale of two mediators. Philos Trans R Soc Lond B Biol Sci 361: 735–759
Corson MA et al. (1996) Phosphorylation of endothelial nitric oxide synthase in response to fluid shear stress. Circ Res 79: 984–991
Griffith TM (2002) Endothelial control of vascular tone by nitric oxide and gap junctions: a haemodynamic perspective. Biorheology 39: 307–318
Davies PF (1995) Flow-mediated endothelial mechanotransduction. Physiol Rev 75: 519–560
Langille BL and O'Donnell F (1986) Reductions in arterial diameter produced by chronic decreases in blood flow are endothelium-dependent. Science 231: 405–407
Zhang H et al. (2008). Heparin-binding epidermal growth factor-like growth factor signaling in flow-induced arterial remodeling. Circ Res 102: 1275–1285
Aird WC (Ed.; 2007) Endothelial Biomedicine. New York: Cambridge University Press
Lucitti JL et al. (2007). Vascular remodeling of the mouse yolk sac requires hemodynamic force. Development 134: 3317–3326
Teichert AM et al. (2008) Endothelial nitric oxide synthase gene expression during murine embryogenesis: commencement of expression in the embryo occurs with the establishment of a unidirectional circulatory system. Circ Res 103: 24–33
Glagov S et al. (1988) Hemodynamics and atherosclerosis. Insights and perspectives gained from studies of human arteries. Arch Pathol Lab Med 112: 1018–1031
Passerini AG et al. (2004) Coexisting proinflammatory and antioxidative endothelial transcription profiles in a disturbed flow region of the adult porcine aorta. Proc Natl Acad Sci USA 101: 2482–2487
Volger OL et al. (2007) Distinctive expression of chemokines and transforming growth factor-beta signaling in human arterial endothelium during atherosclerosis. Am J Pathol 171: 326–337
Cornhill JF and Roach MR (1976) A quantitative study of the localization of atherosclerotic lesions in the rabbit aorta. Atherosclerosis 23: 489–499
Mattsson EJ et al. (1997) Increased blood flow induces regression of intimal hyperplasia. Arterioscler Thromb Vasc Biol 17: 2245–2249
Green DJ et al. (2004) Effect of exercise training on endothelium-derived nitric oxide function in humans. J Physiol 561: 1–25
Adams JA (2007) Therapeutic approaches to altering hemodynamic forces. In Endothelial Biomedicine, 1690–1697 (Ed Aird WC) New York: Cambridge University Press
Suo J et al. (2008) Blood flow patterns in the proximal human coronary arteries: relationship to atherosclerotic plaque occurrence. Mol Cell Biomech 5: 9–18
Steinman DA and Taylor CA (2005) Flow imaging and computing: large artery hemodynamics. Ann Biomed Eng 33: 1704–1709
Davies PF et al. (1999) A spatial approach to transcriptional profiling: mechanotransduction and the focal origin of atherosclerosis. Trends Biotechnol 17: 347–351
García-Cardeña G et al (2001) Mechanosensitive endothelial gene expression profiles: scripts for the role of hemodynamics in atherogenesis? Ann NY Acad Sci 947: 1–6
Libby P (2000) Coronary artery injury and the biology of atherosclerosis: inflammation, thrombosis, and stabilization. Am J Cardiol 86: 3J–8J
Folie BJ and McIntire LV (1989) Mathematical analysis of mural thrombogenesis. Concentration profiles of platelet-activating agents and effects of viscous shear flow. Biophys J 56: 1121–1141
Duraiswamy N et al. (2005) Spatial distribution of platelet deposition in stented arterial models under physiologic flow. Ann Biomed Eng 33: 1767–1777
DePaola N et al. (1992) Vascular endothelium responds to fluid shear stress gradients. Arterioscler Thromb 12: 1254–1257
Moore JE and Berry JL (2002) Fluid and solid mechanical implications of vascular stenting. Ann Biomed Eng 30: 498–508
Balakrishnan B et al. (2005) Strut position, blood flow, and drug deposition: implications for single and overlapping drug–eluting stents. Circulation 111: 2958–2965
Kastrati A et al. (2001) Intracoronary stenting and angiographic results: strut thickness effect on restenosis outcome (ISAR-STEREO) trial. Circulation 103: 2816–2821
Loth F et al. (2003) Transitional flow at the venous anastomosis of an arteriovenous graft: potential activation of the ERK1/2 mechanotransduction pathway. J Biomech Eng 125: 49–61
Hajra L et al. (2000) The NF-κB signal transduction pathway in aortic endothelial cells is primed for activation in regions predisposed to atherosclerotic lesion formation. Proc Natl Acad Sci USA 97: 9052–9057
Caro CG et al. (1969) Arterial wall shear and distribution of early atheroma in man. Nature 223: 1159–1161
Fry DL (1973) Atherogenesis: initiating factors. CIBA Found Symposium 12: 96–118
Dewey CF et al. (1981) The dynamic response of vascular endothelial cells to fluid shear stress. J Biomech Eng 103: 177–188
Levesque MJ and Nerem RM (1985) The elongation and orientation of cultured endothelial cells in response to shear stress. J Biomech Eng 107: 341–347
Suo J et al. (2007) Hemodynamic shear stresses in mouse aortas: implications for atherogenesis. Arterioscler Thromb Vasc Biol 27: 346–351
Wolf S and Werthessen NT (1979) Dynamics of arterial flow. Adv Exp Med Biol 115: 1–472
Huo Y et al. (2007) Flow patterns in three-dimensional porcine epicardial coronary arterial tre. Am J Physiol Heart Circ Physiol 293: H2959–H2970
Asakura T and Karino T (1990) Flow patterns and spatial distribution of atherosclerotic lesions in human coronary arteries. Circ Res 66: 1045–1066
Dekker RJ et al. (2002) Prolonged fluid shear stress induces a distinct set of endothelial cell genes, most specifically lung Krüppel-like factor (KLF2). Blood 100: 1689–1698
Dai G et al. (2007) Biomechanical forces in atherosclerosis-resistant vascular regions regulate endothelial redox balance via phosphoinositol 3-kinase/Akt-dependent activation of Nrf2. Circ Res 101: 723–733
Ziegler T et al. (1998) Influence of oscillatory and unidirectional flow environments on the expression of endothelin and nitric oxide synthase in cultured endothelial cells. Arterioscler Thromb Vasc Biol 18: 686–692
Cheng C et al. (2005) Shear stress affects the intracellular distribution of eNOS: direct demonstration by a novel in vivo technique. Blood 106: 3691–3698
Cheng C et al. (2006) Atherosclerotic lesion size and vulnerability are determined by patterns of fluid shear stress. Circulation 113: 2744–2753
Iiyama K et al. (1999) Patterns of vascular cell adhesion molecule-1 and intercellular adhesion molecule-1 expression in rabbit and mouse atherosclerotic lesions and at sites predisposed to lesion formation. Circ Res 85: 199–207
De Nigris F et al. (2003) Beneficial effects of antioxidants and L-arginine on oxidation-sensitive gene expression and endothelial NO synthase activity at sites of disturbed shear stress. Proc Natl Acad Sci USA 100: 1420–1425
Magid R and Davies PF (2005) Endothelial protein kinase C isoform identity and differential activity of PKCζ in an athero-susceptible region of porcine aorta. Circ Res 97: 443–449
Zakkar M et al. (2008) Increased endothelial mitogen-activated protein kinase phosphatase-1 expression suppresses proinflammatory activation at sites that are resistant to atherosclerosis. Circ Res 103: 726–732
Parmar KM et al. (2006) Integration of flow-dependent endothelial phenotypes by Krüppel-like factor 2. J Clin Invest 116: 49–58
García-Cardeña G and Gimbrone MA (2006) Biomechanical modulation of endothelial phenotype: implications for health and disease. Handb Exp Pharmacol 176: 79–95
Davies PF et al. (1984) Influence of hemodynamic forces on vascular endothelial function. In vitro studies of shear stress and pinocytosis in bovine aortic cells. J Clin Invest 73: 1121–1129
Davies PF et al. (1986) Turbulent fluid shear stress induces vascular endothelial cell turnover in vitro. Proc Natl Acad Sci USA 83: 2114–2117
Shyy YJ et al. (1994) Fluid shear stress induces a biphasic response of human monocyte chemotactic protein 1 gene expression in vascular endothelium. Proc Natl Acad Sci USA 91: 4678–4682
Barbee KA et al. (1995) Subcellular distribution of shear stress at the surface of flow aligned and non-aligned endothelial monolayers. Am J Physiol 268: H1765–H1772
Davies PF and Helmke BP (2008) Endothelial mechanotransduction. In Cellular Mechanotransduction: Diverse Perspectives from Molecules to Tissue, (Eds Mofrad RK and Kamm RD) New York: Cambridge University Press [In press]
Olesen S-P et al. (1988) Hemodynamic shear stress activates a K+ current in vascular endothelial cells. Nature 331: 168–170
Gautam M et al. (2006) Flow-activated chloride channels in vascular endothelium. Shear stress sensitivity, desensitization dynamics, and physiological implications. J Biol Chem 281: 36492–36500
Davies PF et al. (1994) Quantitative studies of endothelial cell adhesion: directional remodeling of focal adhesion sites in response to flow forces. J Clin Invest 93: 2031–2038
Shyy JY and Chien S (2002) Role of integrins in endothelial mechanosensing of shear stress. Circ Res 91: 769–775
Fujiwara K et al. (2001) Is PECAM-1 a mechanoresponsive molecule? Cell Struct Funct 26: 11–17
Tzima E et al. (2005) A mechanosensory complex that mediates the endothelial cell response to fluid shear stress. Nature 437: 426–431
Helmke BP et al. (2003) Spatial concentration of intracellular strain induced by hemodynamic shear stress. Biophys J 84: 2691–2699
Mott RE and Helmke BP (2007) Mapping the dynamics of shear stress-induced structural changes in endothelial cells. Am J Physiol Cell Physiol. 293: C1616–C1626
Maniotis AJ et al. (1997) Demonstration of mechanical connections between integrins, cytoskeletal filaments, and nucleoplasm that stabilize nuclear structure. Proc Natl Acad Sci USA 94: 849–854
Dalby MJ et al. (2007) Nanomechanotransduction and interphase nuclear organization influence on genomic control. J Cell Biochem 102: 1234–1244
Weinbaum S et al. (2007) The structure and function of the endothelial glycocalyx layer. Annu Rev Biomed Eng 9: 121–167
Vink H and Duling BR (1996) Identification of distinct luminal domains for macromolecules, erythrocytes, and leukocytes within mammalian capillaries. Circ Res 79: 581–589
Florian JA et al. (2003) Heparan sulfate proteoglycan is a mechanosensor on endothelial cells. Circ Res 93: e136–e142
Yao Y et al. (2007) Glycocalyx modulates the motility and proliferative response of vascular endothelium to fluid shear stress. Am J Physiol Heart Circ Physiol 293: H1023–H1030
Huang H et al. Three-dimensional sub-diffraction-limited single fluorophore imaging of proteins near the cell membrane: application to the endothelial glycocalyx. Proc Natl Acad Sci USA, in press
Secomb TW et al. (2001) Effect of the endothelial surface layer on transmission of fluid shear stress to endothelial cells. Biorheology 38: 143–150
Helmke BP and Davies PF (2002) The cytoskeleton under external fluid mechanical forces: hemodynamic forces acting on the endothelium. Ann Biomed Eng 30: 284–329
Stamatas GN and McIntire LV (2001) Rapid flow-induced responses in endothelial cells. Biotechnol Prog 17: 383–402
Haust MD (1987) Endothelial cilia in human aortic atherosclerotic lesions. Virchows Arch A Pathol Anat Histopathol 410: 317–326
Van der Heiden K et al. (2008) Endothelial primary cilia in areas of disturbed flow are at the base of atherosclerosis. Atherosclerosis 196: 542–550
Iomini C et al. (2004) Primary cilia of human endothelial cells disassemble under laminar shear stress. J Cell Biol 164: 811–817
Malek AM and Izumo S (1996) Mechanism of endothelial cell shape change and cytoskeletal remodeling in response to fluid shear stress. J Cell Sci 109: 713–726
Hu S and Wang N (2006) Control of stress propagation in the cytoplasm by prestress and loading frequency. Mol Cell Biomech 3: 49–60
Desai G et al. (1999) Pressure-jump studies of the folding/unfolding of trp repressor. J Mol Biol 288: 461–475
Ingber D (2007) Vascular control through tensegrity-based integration of mechanics and chemistry. In Endothelial biomedicine, 1786–1792 (Ed Aird WC) Cambridge: Cambridge University Press
Computational imaging of pulsatile flow in a normal human carotid bifurcation [http://www.mie.utoronto.ca/labs/bsl/gallery/carotid.mpg] (accessed 20 October 2008)
Liu Y et al. (2008) Shc coordinates signals from intercellular junctions and integrins to regulate flow-induced inflammation. J Cell Biol 182: 185–196
Tzima E (2006) Role of small GTPases in endothelial cytoskeletal dynamics and the shear stress response. Circ Res 98: 176–185
Acknowledgements
The author's research is supported by grants from the National Heart Lung and Blood Institute of the National Institutes of Health.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Davies, P. Hemodynamic shear stress and the endothelium in cardiovascular pathophysiology. Nat Rev Cardiol 6, 16–26 (2009). https://doi.org/10.1038/ncpcardio1397
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncpcardio1397
This article is cited by
-
Structural and Mechanical Properties of Human Superficial Femoral and Popliteal Arteries
Annals of Biomedical Engineering (2024)
-
Role of albumin infusion in cirrhosis-associated complications
Clinical and Experimental Medicine (2024)
-
A clinician’s guide to understanding aortic 4D flow MRI
Insights into Imaging (2023)
-
Haemodynamic changes in visceral hybrid repairs of type III and type V thoracoabdominal aortic aneurysms
Scientific Reports (2023)
-
Ultrafast longitudinal imaging of haemodynamics via single-shot volumetric photoacoustic tomography with a single-element detector
Nature Biomedical Engineering (2023)