Abstract
In patients who are receiving prolonged antiretroviral treatment (ART), HIV can persist within a small pool of long-lived resting memory CD4+ T cells infected with integrated latent virus. This latent reservoir involves distinct memory subsets. Here we provide results that suggest a progressive reduction of the size of the blood latent reservoir around a core of less-differentiated memory subsets (central memory and stem cell-like memory (TSCM) CD4+ T cells). This process appears to be driven by the differences in initial sizes and decay rates between latently infected memory subsets. Our results also suggest an extreme stability of the TSCM sub-reservoir, the size of which is directly related to cumulative plasma virus exposure before the onset of ART, stressing the importance of early initiation of effective ART. The presence of these intrinsic dynamics within the latent reservoir may have implications for the design of optimal HIV therapeutic purging strategies.
Similar content being viewed by others
Introduction
Although combined antiretroviral therapy (ART) generally suppresses HIV replication to undetectable plasma levels for prolonged periods, it fails to eradicate the virus. HIV can persist within a small pool of long-lived resting memory CD4+ T cells infected with integrated latent virus1,2,3,4. This latent reservoir appears to involve several memory CD4+ T-cell subsets at distinct differentiation stages with different phenotypic and functional properties, forming distinct ‘sub-reservoirs’5,6. Precise immunological characterization of the latent CD4+ T-cell reservoir, including the size of each sub-reservoir, is crucially important for the complex challenge of ‘therapeutic purging’. The relative size of each sub-reservoir may depend on its decay rate and may therefore vary according to the time on ART.
Here we show the existence of a dynamic process that progressively reduces the size of the latent reservoir around a core of less differentiated memory CD4 T-cell subsets (for example, central memory CD4+ T cells and the recently identified stem cell-like memory CD4+ T cells). Our results also stress the importance of early initiation of effective ART to limit the size of the TSCM sub-reservoir, which appears directly related to cumulative plasma virus exposure.
Results
Study design
We examined the decay rates of resting memory subsets latently infected by HIV in highly selected patients with consistently undetectable plasma virus on ART. Cell sorting of CD4+ T-cell memory subsets requires the use of fresh peripheral blood mononuclear cells (PBMC), as cryopreservation alters the expression of markers, such as CD62L, which is required to sort the different memory subsets7,8 (see also Supplementary Fig. 1). This ruled out a retrospective study on frozen cells. Furthermore, a longitudinal prospective study can take more than a decade. We therefore chose to conduct a cross-sectional analysis on strictly selected patients from a cohort of 360 HIV-1-infected patients (see methods). The characteristics of patients who fulfilled the selection criteria and were enrolled are shown in Supplementary Table 1. The recently identified TSCM subset consists of rare CD4+ memory T cells with stem cell-like features9. In response to recall antigens, TSCM exhibited the strongest proliferation among the tested memory cell subsets (Supplementary Fig. 2). Figure 1a shows the gating strategy used to sort highly purified resting TSCM (see also Supplementary Fig. 3 for purity). Resting central memory (TCM) and effector memory CD4+ T cells (TEM) were sorted on the basis of stringent criteria9. An additional resting CD4+ T-cell memory subset with an intermediate CCR7− CD62L+ phenotype, designated TIM (intermediate memory), was also sorted (see Methods). Infectious virus was recovered from the four resting memory cell subsets following in vitro activation (see Supplementary Fig. 4).
(a) The gating strategy used to sort the memory cell subsets: CD4-enriched PBMC were stained with a cocktail of antibodies (see Methods) and doublets were excluded on the basis of both forward scatter (FSC) and side scatter (SSC). Resting CD4+ T cells were gated after exclusion of CD19+, CD14+, CD8+, HLADR+, CD69+, CD25+ cells. Resting TSCM were sorted on the basis of the following phenotype: CD45RA+ CD45RO− CCR7+ CD62L+ CD27+ CD95+. CXCR3 and CD122 expression by TSCM is also shown. (b) The absolute number of cells in each memory subset (TSCM, TCM, TEM and TIM) was determined in HIV-infected patients with undetectable plasma viral load on ART and with CD4 cell counts above 500 per mm3, who were tested for integrated virus (UND, n=38), as well as in age- and sex-matched viremic patients (VIR, n=18) and HIV-seronegative healthy donors (HD, n=20). The Kruskal–Wallis and Dunn tests were used for statistical analysis (*P<0.05, **P<0.01, ****P<0.0001). Boxes represent the median and the 25th and 75th percentiles; whiskers represent the 10th and 90th percentiles. (c) Shows the percentage of each memory subset within the compartment formed by the four memory subsets analysed. The χ2 test was used to analyse differences in the distribution of the four memory subsets (****P<0.0001).
Tight homeostatic regulation of TSCM
Cell numbers of the different memory subsets per mm3 of blood were similar in the patients with undetectable plasma virus and in a group of HIV-seronegative healthy donors (Fig. 1b). In contrast, in a group of patients with chronic active viral replication, the TSCM and TCM CD4+ subsets were significantly depleted (Fig. 1b). Within the pool of memory CD4+ T cells, the distribution of the different memory subsets was markedly different between patients with chronic active HIV replication and HIV-seronegative donors (Fig. 1c). This was mainly due to a significant increase in the proportions of TEM and TIM, to the detriment of TCM (P<0.01 each for the percentages of TCM, TEM and TIM within the pool of memory CD4+ T cells; Kruskal–Wallis and Dunn’s tests; see Supplementary Fig. 5). Possible explanations for these differences include increased differentiation of TCM towards memory effector subsets, higher susceptibility of TCM to infection-induced cell death and altered TCM trafficking between secondary lymphoid tissues and blood. In the patients with undetectable plasma virus on ART, the percentages of TCM and TEM within the pool of memory CD4+ T cells were significantly different when compared with healthy donors (P<0.01; Kruskal–Wallis and Dunn’s tests), but were not different when compared with patients with active virus replication (see Supplementary Fig. 5). The percentage of TIM was not different between patients on ART with undetectable virus and both healthy donors and viremic patients. As shown in Fig. 1c, the distribution of the different subsets within memory CD4+ T cells differed between the patients with undetectable plasma virus on ART and both the patients with active virus replication and the healthy donors. This suggested that recovery of the CD4+ T-cell memory pool during prolonged effective ART was incomplete. Interestingly, there was no significant difference in the percentage of TSCM within memory CD4+ T cells between the patients on prolonged effective ART, the patients with active virus replication and the healthy donors (see Supplementary Fig. 5). This suggests tight homeostatic regulation maintaining the equilibrium between TSCM and the rest of the memory CD4+ T cells despite active virus replication. In each group of patients and healthy donors analysed, there was no significant relation between the age and the absolute numbers of TSCM per mm3 of blood (P=0.12, 0.53 and 0.69, for patients with chronic active virus replication, patients on prolonged effective ART and healthy donors, respectively, Spearman Rank Test) or the percentage of TSCM within memory CD4+ T cell (P=0.83, 0.11 and 0.11, respectively, Spearman Rank Test).
A highly stable Core of latently infected TSCM and TCM
Integrated HIV DNA was quantified in sorted CD4 memory subsets with a well-established Alu-gag PCR method10,11 (see Methods and Supplementary Fig. 6). Sorted naive CD4+ T cells (CD45RA+ CD45RO− CCR7+ CD62L+ CD27+ CD95−) were also tested, but in our series integrated HIV DNA was detected in truly naive cells only in 18.4% of patients (see Supplementary Fig. 7). The results obtained for memory subsets are shown in Fig. 2a. The best linear regression of our data suggested that the TSCM sub-reservoir had the longest half-life among the latently infected memory subsets analysed (not shown). However, to take possible inter-patient variability into account and to validate this cross-sectional analysis, the results were analysed with the Monte Carlo (MC) algorithm (see Methods). Briefly, we performed 106 computer simulations with random samplings for each latently infected memory subset, based on the mean values and variance of our experimental data. For each simulation, we fitted the number of integrated HIV DNA copies per 105 cells at the time of undetectable plasma virus following ART initiation (y0) and the slope of decay (λ). The mean values of y0 and λ obtained from the 106 simulations were used to plot the decay curves shown in Fig. 2a. The distribution of y0 and λ for each latently infected memory subset is indicated in Supplementary Fig. 8. The mean value of λ was used to estimate the half-life (−ln2/λ) of each latent sub-reservoir. The mean y0, λ and half-life values were as follows: 16.54 integrated copies per 105 cells, −0.0025 month−1 and 277 months for TSCM; 55.54 integrated copies per 105 cells, −0.0048 month−1 and 144 months for TCM; 42.92 integrated copies per 105 cells, −0.0052 month−1 and 133 months for TIM and 59.88 integrated copies per 105 cells, −0.0079 month−1 and 88 months for TEM. Analysis of the MC results (two-way analysis of variance and t-test (significance at P<0.05), with a Bonferroni post-test) confirmed that the TSCM latent reservoir had a slower decay rate than the other studied sub-reservoirs (P<0.05), whereas no significant difference was found between the decay rates of TCM, TIM and TEM (see also Supplementary Fig. 8a). The same analysis also showed that TSCM had a lower number of integrated copies than the other sub-reservoirs at the time of undetectable plasma virus following ART initiation (y0; P<0.05), whereas no significant difference was found between TCM, TIM and TEM (see also Supplementary Fig. 8b).
(a) Integrated HIV DNA was quantified in TSCM, TCM, TEM and TIM isolated from 38 strictly selected patients in whom viral load was undetectable for 24–189 months on ART. Recovery of TEM and TSCM was suboptimal in three patients. TIM could not be isolated from nine patients. The results are expressed as the integrated HIV DNA copy number per 105 cells. Taking inter-patient variability into account, the data shown in a were analysed with the Monte Carlo algorithm with 106 computer simulations with random samplings for each latently infected memory subset (see Methods). For each simulation, the number of integrated HIV DNA copies per 105 cells at the time of undetectable plasma virus following ART initiation (y0) and the slope of decay (λ) were fitted. The distributions of y0 and λ obtained from the 106 simulations are shown in Supplementary Fig. 8. The mean values of y0 and λ were used to plot the decay curves shown in a. The mean half-life of decay (−ln2/λ) is indicated for each latently infected memory subset. Statistical significance is indicated as * (for statistical analysis, see the Monte Carlo section of Methods). (b) Extends the data shown in a to the entire blood compartment. To analyse the decay in absolute numbers of latently infected memory subsets in the whole blood compartment, we used the 106 simulation values of y0 and slopes of decay obtained for each memory subset following Monte Carlo analysis. The predictions shown in b correspond to the average values of y0 and slopes. For the other parameters required to calculate the absolute numbers of latently infected cells (body weight, CD4 T-cell count and the percentages of the memory subsets among total CD4 T cells), we used the median values from our cohort of 38 patients. Total blood volume was estimated to represent 7% of total body weight29. Note that the predictions in b remained consistent when random sampling (Monte Carlo approach) was used for body weight, the CD4+ T-cell count and the percentages of memory subsets among total CD4+ T cells, based on the median values and variance for the patients analysed. We considered 50 time points from 0 to 200 years (a total of 50 × 106 values for each memory subset). Comparison between the memory subsets was performed as in a. (See in Methods the Monte Carlo section). Statistical significance is indicated *, #. (c) The immunological dynamics of the CD4+ T-cell latent reservoir, corresponding to changes, at chosen time-points, in the relative size of each memory subset within the compartment formed by the four latently infected memory subsets in the entire blood compartment (see above). The surface areas are proportional to the numbers of integrated HIV DNA copies in each memory CD4 subset in the entire blood volume (predictions shown in b).
We then predicted the individual decay of each latently infected memory subset within the entire blood volume (Fig. 2b), as well as long-term changes in the relative proportions of the different memory subsets forming the latent CD4+ T-cell reservoir (Fig. 2c). For this purpose, we took into account the y0 and decay rate values of each sub-reservoir obtained by MC analysis, together with the absolute number of cells composing each CD4+ memory subset in the entire blood volume (see Methods and legend to Fig. 2b,c). We found the following hierarchy in the total sizes of the different memory sub-reservoirs in the entire blood volume at the time of undetectable plasma virus following ART initiation: TCM>TEM>TIM>TSCM (P>0.05; Fig. 2b). The relative size of the TSCM sub-reservoir within the latent memory reservoir then increased over time, owing to the longer half-life of these cells (Fig. 2b,c). The data predicted that the size difference between TSCM, TEM and TIM sub-reservoirs would become non-significant after 50 years on effective ART, whereas the TCM sub-reservoir would remain larger than the other three sub-reservoirs (Fig. 2b). The decay of the whole-blood latent CD4+ T-cell memory reservoir appeared to be driven mainly by the decay rate of the TCM sub-reservoir (Fig. 2b). The results also imply that it would take more than 100 years for TEM, the most labile latent sub-reservoir, to be eradicated by ART, without additional therapeutic approach (Fig. 2b). Altogether, TSCM and TCM appear to form the most stable part of the latent CD4+ T-cell memory reservoir, mainly owing to the extremely long half-life of TSCM and to the larger size of the TCM reservoir at the initiation of effective ART (Fig. 2b,c).
TSCM reservoir size is related to cumulative HIV exposure
We then examined the influence of several parameters on the frequency of integrated HIV in each memory CD4+ T-cell subset at the time when plasma virus became undetectable (see legend of Fig. 3), including age and sex, the CD4+ T-cell nadir, the duration of detectable HIV viremia and cumulative HIV plasma viremia. Cumulative plasma viremia, expressed in log10 virus copy-years, is a measure of cumulative plasma viral load exposure during a given time period12. As shown in Fig. 3a–c, the frequency of integrated HIV DNA in TSCM correlated positively with cumulative HIV plasma viremia, over a period up to 7 years before plasma virus becoming undetectable, whereas no such relation was found for the other memory subsets (Fig. 3b,c). None of the other parameters significantly influenced the size of the latently infected memory subsets (P values ranged to from 0.18–0.9, Spearman rank test). Together, the results suggested that the size of the TSCM sub-reservoir is more directly influenced by the level and duration of plasma virus exposure than the other sub-reservoirs.
(a,b) Cumulative viremia, expressed as log10 virus copy-years during periods of 36, 60 and 84 months before plasma virus became undetectable, was determined in patients (n=9–13) for whom virological follow-up was available for a period of 84 months. Associations were sought between cumulative viremia and the frequency of latent infection of each memory subset at the time when plasma virus became undetectable after ART initiation. For each patient, the frequency of latent infection of each memory subset at the time when plasma virus became undetectable after ART initiation was calculated from the slopes of the decay curves shown in Fig. 2a. The number of patients for whom virological follow-up was available over a period longer than 84 months before undetectable plasma virus, was insufficient for statistical analysis. In c, the same analysis was performed for all patients (n=16–22) for whom virological follow-up was available over a period of 36 months before plasma virus became undetectable. The Spearman rank test was used for statistical analysis (*P<0.05, **P<0.01).
Discussion
Together, these results reveal the existence of an intrinsic dynamic process within the latent reservoir that contracts around a core of less-differentiated memory subsets (TCM and TSCM). This slow process is driven by differences in initial sizes and decay rates of the latently infected memory subsets. The half-life of each latently infected subset may be related to the intrinsic capacity of its component cells to survive and to self-renew. Several lines of evidence suggest that homeostatic proliferation, including in response to IL-7, may not necessarily lead to efficient reactivation of latent HIV and subsequent cell destruction, thus allowing self-renewing cells and their progeny to persist in the latent CD4+ T-cell reservoir5,13,14. Long-term survival and self-renewal are key properties of TSCM and, to a lesser extent, of TCM9. These features may explain the greater stability of the TSCM reservoir as compared with the more highly differentiated CD4+ T-cell memory subsets. However, the half-lives of latently infected memory subsets may also be influenced by cell transfer from other compartments. For example, in response to homeostatic signals, TSCM can generate more highly differentiated cells such as TCM and TEM while maintaining their own pool9,15, thereby fueling downstream compartments.
A recent work suggested that TSCM harbour the highest level of HIV DNA among memory T-cell subsets regardless of the time on ART, with a considerable inter-individual variations16. Our results do not support this finding. We found that the TSCM sub-reservoir has great stability but at the time when plasma virus became undetectable on ART, it harbours the lowest level of stable infection among latently infected memory subsets. Noteworthy, the authors used a PCR procedure that quantifies total HIV DNA including integrated and unintegrated labile virus. The latter may reflect residual virus expression17,18 that may increase inter-individual variability of total HIV DNA.
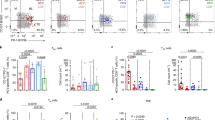
Why does cumulative plasma virus exposure influence the formation of the latent TSCM sub-reservoir more strongly than the formation of other memory sub-reservoirs? One possibility is the existence of additional factors that influence the formation of latently infected CD4+ T cells in addition to viral exposure and that are more active in TEM, TIM and TCM. These might include immunological factors that negatively regulate cell activation, such as cell-surface inhibitory receptors. The level of PD-1 expression has been shown to influence the level of latent infection in CD4 T cells in patients on ART5. As shown in Fig. 4a,b, a lower PD-1 expression was found on TSCM as compared with other CD4 memory subsets in viremic patients and patients on ART with undetectable plasma virus. This was consistent with previous reports suggesting that in CD4 and CD8 T cells the expression of the inhibitory receptor PD-1 could be related to the stage of differentiation5,19,20. PD-1 could represent a potential target to facilitate HIV eradication5,21. However, the low PD-1 expression on TSCM argues against an effectiveness of PD-1 targeting therapies in reducing the size of the TSCM sub-reservoir. Alternatively, the presence within a memory subset of latently infected cells generated by indirect infection, for example, through cell transfer from the TSCM pool towards more highly differentiated memory subsets, might also alter the direct relationship between the size of the sub-reservoir and cumulative viral exposure.
Surface PD-1 expression on blood memory CD4+ T-cell subsets was analysed in 15 patients with CD4+ T-cell counts >500 per mm3 and undetectable plasma virus for at least 24 months on ART (a) and in 15 viremic patients (median CD4 T-cell count=477 per mm3, median viral load=3.3 log10, b). Data are expressed as the difference in PD-1 mean fluorescence intensity (ΔMFI) relative to naive CD4+ T cells. Boxes represent the median and the 25th and 75th percentiles; whiskers represent the 10th and 90th percentiles. Representative PD-1 staining is shown. The data were analysed with the Friedman test for paired data and with Dunn’s multiple comparison test (*P<0.05, **P<0.01, ***P<0.001, ****P<0.0001).
Our results show that it is unrealistic to expect ART alone to purge the latent HIV reservoir. PCR procedures for detecting integrated HIV may overestimate the size of the latent CD4+ T-cell reservoir. Indeed, a significant proportion of integrated viruses is assumed to be defective because it cannot be induced by in vitro activation. Importantly, the stimuli used in vitro to activate cells and to recover latent virus in outgrowth assays may not fully reproduce the complex activation conditions existing in vivo, particularly given the marked immunological heterogeneity of the latent reservoir cells. Indeed, this reservoir comprises different CD4+ T-cell subsets with distinct activation requirements (for instance, see Fig. 4a,b, showing variable expression of the inhibitory receptor PD-1 by different cell subsets). This implies that some latent viruses capable of generating infective virions in vivo may not be recoverable in vitro. Also, a recent elegant study showed that the latent reservoir contains proviruses with no deletions or inactivating mutations that are not induced after in vitro activation, but might possibly be recoverable in vivo22. The authors of this latter study forwarded a model of stochastic induction of intact proviruses following cell activation22. Clearly, although PCR procedures for detecting integrated HIV may overestimate the size of the latent reservoir, viral outgrowth assays can underestimate it22. In practice, this means that a patient cannot be said to be cured of HIV infection unless integrated virus is undetectable by PCR.
The part of the latent CD4 T-cell reservoir formed by TCM and TSCM should be a priority target for therapeutic strategies addressing the latent reservoir. Therapeutic approaches targeting key memory and effector differentiation pathways could also help to drive latently infected memory cells, beyond the effector memory state, towards short-lived terminally differentiated effector states, thereby accelerating the decay of the latent reservoir. Our results also stress the importance of early initiation of effective ART to limit the size of the TSCM reservoir, which appears directly related to cumulative plasma virus exposure.
Methods
Patients
Patients were selected from a cohort of 360 HIV-1-infected patients in whom plasma viral load was determined every 3 months. To be eligible for the study, patients had to have undetectable plasma virus on combined antiretroviral therapy (ART) for at least 24 months. Detection of a single viral blip was a formal exclusion criterion. Patients also had to have a CD4+ T-cell count higher than 500 per mm3 of blood, indicating effective immune recovery on ART, at the time when their cell subsets were tested for integrated HIV or for viral production. Forty-five eligible patients (plasma virus load undetectable (<20 copies per ml) for 24–189 months) agreed to participate in this cross-sectional study. Integrated HIV DNA was quantified in 38 patients. Characteristics of the patients are indicated in Supplementary Table 1. Cells from seven patients with undetectable plasma virus for 34–148 months were used for inducing virus production in vitro. Written informed consent was obtained from each patient and the study was approved by the local ethics committee (Comité pour la Protection des Personnes Ile-de-France VII, (CPP IDF VII) Bicêtre, France). Cumulative HIV plasma viremia up to the time when plasma virus became undetectable (from 12–84 months before than plasma virus became undetectable) was determined as previously described13 in patients for whom plasma viral load data were available.
Flow cytometry
A blood sample of 200 μl was used for CD4+ memory T-cell phenotyping with the following antibodies: anti-CD8-FITC (1/10, clone RPA-T8), anti-PD1-FITC (1/5, clone MIH4), anti-CD122-PE (1/10, clone Mik-B3), anti-CD62L-V450 (1/10, clone DREG-56), anti-CD4-V500 (1/20, clone RPA-T4), anti-CD95-APC (1/10, clone DX2), anti-CD45RA-PE-Cy7 (1/20, clone HI100), anti-CD45RO-PerCPCy5.5 (1/10, clone UCHL1), anti-CCR7-PE-CF594 (1/10, clone 150503), anti-CXCR3-Alexa 700 (1/10, clone 1C6/CXCR3), anti-CD27-APC-H7 (1/10, clone M-T271) (all from BD Biosciences), and anti-CD3-eFluor 650NC (1/10, clone OKT3, eBioscience). After staining, the blood sample was fixed (fix/lyse solution, BD Biosciences) and cells were acquired on a BD LSR Fortessa cytometer (BD Biosciences). Data were analysed with Flow Jo software.
Cell sorting
CD4+ T cells were enriched from PBMC by negative selection with magnetic beads (CD4 BD Imag, BD Biosciences). The cells were then stained with a cocktail of the following antibodies: anti-HLA-DR-FITC (1/15, clone G46-6), anti-CD8 FITC (1/15, clone RPA-T8), anti-CD19-FITC (1/15, clone HIB19), anti-CD14 FITC (1/15, clone M5E2), anti-CD25-FITC (1/6, clone M-A251), anti-CD69-FITC (1/6, clone FN50), anti-CD3-Alexa 700 (1/15, clone OKT3), anti-CD122-PE (1/12, clone Mik-B3), anti-CD62L-V450 (1/15, clone DREG-56), anti-CD4-V500 (1/30, clone RPA-T4), anti-CD95-APC (1/15, clone DX2), anti-CD45RA-PE-Cy7 (1/30, clone HI100), anti-CD45RO-PerCP-Cy5.5 (1/15, clone UCHL1), anti-CCR7-PE-CF594 (1/15, clone 150503) and anti-CD27-APC-H7 (1/30, clone M-T271) (all from BD Biosciences), then sorted with a BD FacS ARIA III cytometer (BD Biosciences). Non-CD4+ T cells and possibly activated CD4+ T cells were gated out by fluorescein isothiocyanate staining. After gating on the live-cell gate and doublet exclusion, highly purified memory CD4+ T-cell subsets were sorted on the basis of the following phenotypes: (i) stem cell memory CD4+ T cells (TSCM): CD3+ CD4+ CD45RA+ CD45RO− CCR7+ CD62L+ CD27+ CD95+9; (ii) central memory CD4+ T cells (TCM): CD3+ CD4+ CD45RA− CD45RO+ CCR7+ CD62L+ and (iii) effector memory CD4 T cells (TEM): CD3+ CD4+ CD45RA− CD45RO+ CCR7− CD62L−. An additional subset with an intermediate phenotype (TIM) was also sorted: CD3+ CD4+ CD45RA− CD45RO+ CCR7− CD62L+.
Latent HIV quantification
Integrated HIV DNA was quantified by two-step ALU-gag PCR as previously described10,11 (see also Supplementary Fig. 6). Serial dilutions of the 8E5 cell line (American Type Culture Collection) in PBMC from HIV-seronegative donors were used to generate a standard curve with which to quantify integrated HIV DNA (Supplementary Fig. 6a). The 8E5 cell line harbours a single integrated HIV copy per genome. The standard curve spanned 2,000 to 1 HIV copies per PCR. The PCR method was able to detect one integrated HIV DNA copy (one 8E5 cell) among 100,000 cellular genomes (Supplementary Fig. 6a). The same numbers of resting TSCM, TCM, TEM and TIM, corresponding to the lowest cell number recovered among the four cell subsets after sorting, were pelleted for DNA extraction. In all patients, glyceraldehyde 3-phosphate dehydrogenase quantitative PCR performed on 10% of the extracted DNA showed that similar amounts of DNA were effectively recovered from the cell subsets. Duplicate cell aliquots were used whenever possible. The product of the first Alu-gag PCR was split into five 10-μl aliquots, and each aliquot was submitted to nested PCR10,11. We used the mean value obtained for the five aliquots. When duplicate cellular aliquots were available, we used the mean of the ten values. Additional information about Latent HIV quantification is provided in Supplementary Methods.
Statistical analysis
Statistical analysis was performed using Prism software (GraphPad). Differences between groups were analysed using the Kruskal–Wallis and Dunn’s tests for unpaired continuous variables, and the Friedman and Dunn tests for paired continuous variables. The χ2 test was used for categorical variables. The Spearman rank test was used to examine associations between the size of the latently infected memory subsets at the time when plasma virus became undetectable and parameters including age and sex, the CD4+ T-cell nadir, the duration of HIV viremia and cumulative HIV viremia before plasma virus becoming undetectable.
Estimation of decay rates of CD4+ T-cell sub-reservoirs
Bootstrapping is a statistic method for assigning measures of accuracy to estimates23,24. A standard choice for approximating the distribution is to use the empirical distribution of the observed data. This can be implemented by constructing a number of resamples of the data set, each of which is obtained by random sampling with replacement from the original data set.
In this paper, for each CD4+ T-cell memory reservoir, we consider the following model:

where y0 is the integrated HIV DNA copy number at the time of undetectable plasma virus following ART initation, λ is the slope of decay for each latent sub-reservoir and t is the duration of plasma virus undetectability. The half-life is computed with the term −ln2/λ. Note that we considered nonlinear mathematical models, however we could not find any statistically significant improvement.
Therefore, given a vector observation (measurement) yi at time ti, the parameter set φ=[λ,y0] is computed for each CD4+ T-cell memory sub-reservoir (inverse problem23).
The parameter fitting is performed by minimizing the root mean square difference on a log scale between the model predictive output (xi) and the experimental measurement (yi), as follows:

where n is the total number of measurements. The minimization of root mean square is performed using the differential evolution algorithm25. Several optimization solvers were tested, including both deterministic (fmincon Matlab routine, Threshold Acceptance algorithm and Pattern Search algorithm) and stochastic methods (Genetic algorithm and Annealing algorithm). We found that the differential evolution global optimization algorithm was robust to initial guesses of parameters and converged faster with more certainty than the other methods.
As inter-patient variability is non-negligible in this work, we considered the bootstrap method to analyse mathematical models in stochastic environments. Bootstrap is useful when there is no form to estimate the distribution of the statistic of interest24. One way of performing case resampling is to use the MC algorithm. This methodology has been successfully used to address different biological problems24,26.
MC Analysis
The MC algorithm24,27 is a large dimensional-space method that uses repeated random samplings to obtain numerical results in a stochastic environment when experiments cannot be repeated several times and when variability is present.
To settle down the simulations, we resample the data with replacements. The size of the resample must be equal to the size of the original set. To this end, based on the variance of our raw data for each time point in Fig. 2a, we generated a vector with random samplings using a geometric distribution (results were consistent using normal or exponential distribution). Using equation (1) we again refitted the parameters (λ, y0), and repeated this process 106 times for each CD4+ T-cell subset. This large number of repetitions provides a more precise estimate of the parameter distribution. Histograms showing the distribution of y0 and λ obtained by bootstrapping can be found in Supplementary Fig. 8. The 95% confidence interval of parameter estimates is computed using the outcome of the bootstrap method28. For each constant parameter, we select the 2.5 and 97.5% quantiles of the 106 estimates to form the 95% confidence interval. The results are presented in Supplementary Fig. 8.
Analyses of the MC outcome were implemented with two-way analysis of variance and a t-test. Significance was assumed at P<0.05, and the data were further analysed with a Bonferroni post-test.
Additional information
How to cite this article: Jaafoura, S. et al. Progressive contraction of the latent HIV reservoir around a core of less-differentiated CD4+ memory T-Cells. Nat. Commun. 5:5407 doi: 10.1038/ncomms6407 (2014).
References
Chun, T. W. et al. Quantification of latent tissue reservoirs and total body viral load in HIV-1 infection. Nature 387, 183–188 (1997).
Chun, T. W. et al. Presence of an inducible HIV-1 latent reservoir during highly active antiretroviral therapy. Proc. Natl Acad. Sci. USA 94, 13193–13197 (1997).
Finzi, D. et al. Latent infection of CD4+ T cells provides a mechanism for lifelong persistence of HIV-1, even in patients on effective combination therapy. Nat. Med. 5, 512–517 (1999).
Finzi, D. et al. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science (New York, NY) 278, 1295–1300 (1997).
Chomont, N. et al. HIV reservoir size and persistence are driven by T cell survival and homeostatic proliferation. Nat. Med. 15, 893–900 (2009).
Tran, T. A. et al. Resting regulatory CD4 T cells: a site of HIV persistence in patients on long-term effective antiretroviral therapy. PLoS ONE 3, e3305 (2008).
Costantini, A. et al. Effects of cryopreservation on lymphocyte immunophenotype and function. J. Immunol. Methods 278, 145–155 (2003).
Weinberg, A. et al. Optimization and limitations of use of cryopreserved peripheral blood mononuclear cells for functional and phenotypic T-cell characterization. Clin. Vaccine Immunol. 16, 1176–1186 (2009).
Gattinoni, L. et al. A human memory T cell subset with stem cell-like properties. Nat. Med. 17, 1290–1297 (2011).
Liszewski, M. K., Yu, J. J. & O'Doherty, U. Detecting HIV-1 integration by repetitive-sampling Alu-gag PCR. Methods (San Diego, Calif) 47, 254–260 (2009).
Yu, J. J. et al. A more precise HIV integration assay designed to detect small differences finds lower levels of integrated DNA in HAART treated patients. Virology 379, 78–86 (2008).
Cole, S. R. et al. Copy-years viremia as a measure of cumulative human immunodeficiency virus viral burden. Am. J. Epidemiol. 171, 198–205 (2010).
Kim, H. & Perelson, A. S. Viral and latent reservoir persistence in HIV-1-infected patients on therapy. PLoS Comput. Biol. 2, e135 (2006).
Vandergeeten, C. et al. Interleukin-7 promotes HIV persistence during antiretroviral therapy. Blood 121, 4321–4329 (2013).
Lugli, E. et al. Superior T memory stem cell persistence supports long-lived T cell memory. J. Clin. Invest. 123, 594–599 (2013).
Buzon, M. J. et al. HIV-1 persistence in CD4 T cells with stem cell-like properties. Nat. Med. 20, 139–142 (2014).
Mexas, A. M. et al. Concurrent measures of total and integrated HIV DNA monitor reservoirs and ongoing replication in eradication trials. AIDS (London, England) 26, 2295–2306 (2012).
Agosto, L. M. et al. Patients on HAART often have an excess of unintegrated HIV DNA: implications for monitoring reservoirs. Virology 409, 46–53 (2011).
Adekambi, T. et al. Distinct effector memory CD4+ T cell signatures in latent Mycobacterium tuberculosis infection, BCG vaccination and clinically resolved tuberculosis. PLoS ONE 7, e36046 (2012).
Sauce, D. et al. PD-1 expression on human CD8 T cells depends on both state of differentiation and activation status. AIDS (London, England) 21, 2005–2013 (2007).
Porichis, F. & Kaufmann, D. E. Role of PD-1 in HIV pathogenesis and as target for therapy. Curr. HIV/AIDS Rep. 9, 81–90 (2012).
Ho, Y. C. et al. Replication-competent noninduced proviruses in the latent reservoir increase barrier to HIV-1 cure. Cell 155, 540–551 (2013).
Chernick, M. R. Bootstrap Methods: A Practitioner's Guide Wiley (1999).
Davison, A. C. & Hinkley, D. V. Bootstrap Methods and Their Application Cambridge Univ. (1997).
Storn, R. & Price, K. Differential evolution a simple and efficient heuristic for global optimization over continuous spaces. J. Global Optim. 11, 341–359 (1997).
Hernandez-Vargas, E. A. et al. The effects of aging on influenza virus infection dynamics. J. Virol. 88, 4123–4131 (2014).
Anderson, H. L. Metropolis, Monte Carlo and the MANIAC. Los Alamos Science 14, 96–108 (1986).
Xue, H., Miao, H. & Wu, H. Sieve estimation of constant and time varying coefficients in nonlinear ordinary differential equation models by considering both numerical error and measurement error. Ann. Stat. 38, 2351–2387 (2010).
Cameron, J. R., Skofronick, J. G. & Grant, R. M. Physics of the Body 2nd ednMedical Physics Publishing (1999).
Acknowledgements
This work was supported by ANRS (Agence Nationale de Recherche sur le SIDA, 75013 Paris, France). S.J. has a fellowship grant from ANRS. We thank K Bourdic, M Mole, MJ Dulucq and all the nurses at the HIV Daycare Unit, Department of Internal Medicine, Bicêtre Hospital, 94276 le Kremlin-Bicêtre, France.
Author information
Authors and Affiliations
Contributions
S.J. designed and performed experiments and analysed data. M.G.deG.deH. designed and performed experiments, analysed data, performed statistical analysis and wrote the paper. E.A.H.-V. analysed data, performed mathematical modelling and statistical analysis and wrote the paper. M.A. performed experiments and analysed data. M.C.M. performed experiments and analysed data. R.K. analysed data. M.M. selected patients and analysed data. R.S. performed statistical analysis. M.T. designed experiments and analysed data. J.F.D. designed experiments and analysed data. C.G. selected patients, designed experiments and analysed results. Y.T. designed experiments, analysed data, wrote the paper and supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-8, Supplementary Table 1 and Supplementary Methods. (PDF 510 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Jaafoura, S., de Goër de Herve, M., Hernandez-Vargas, E. et al. Progressive contraction of the latent HIV reservoir around a core of less-differentiated CD4+ memory T Cells. Nat Commun 5, 5407 (2014). https://doi.org/10.1038/ncomms6407
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms6407
This article is cited by
-
More than the Infinite Monkey Theorem: NHP Models in the Development of a Pediatric HIV Cure
Current HIV/AIDS Reports (2024)
-
Early intervention with 3BNC117 and romidepsin at antiretroviral treatment initiation in people with HIV-1: a phase 1b/2a, randomized trial
Nature Medicine (2022)
-
Longitudinal analysis of subtype C envelope tropism for memory CD4+ T cell subsets over the first 3 years of untreated HIV-1 infection
Retrovirology (2020)
-
Viral diversity is an obligate consideration in CRISPR/Cas9 designs for targeting the HIV reservoir
BMC Biology (2018)
-
Measuring integrated HIV DNA ex vivo and in vitro provides insights about how reservoirs are formed and maintained
Retrovirology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.