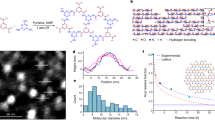
Abstract
Aligning polymeric nanostructures up to macroscale in facile ways remains a challenge in materials science and technology. Here we show polymeric self-assemblies where nanoscale organization guides the macroscopic alignment up to millimetre scale. The concept is shown by halogen bonding mesogenic 1-iodoperfluoroalkanes to a star-shaped ethyleneglycol-based polymer, having chloride end-groups. The mesogens segregate and stack parallel into aligned domains. This leads to layers at ~10 nm periodicity. Combination of directionality of halogen bonding, mesogen parallel stacking and minimization of interfacial curvature translates into an overall alignment in bulk and films up to millimetre scale. Upon heating, novel supramolecular halogen-bonded polymeric liquid crystallinity is also shown. As many polymers present sites capable of receiving halogen bonding, we suggest generic potential of this strategy for aligning polymer self-assemblies.
Similar content being viewed by others
Introduction
Self-assembly of polymers has received significant attention aiming at simple and low-cost methods for new technologies1,2,3,4,5. Supramolecular principles further allow hierarchical polymeric structures, modularity, complexity and functionalities6,7,8,9,10. Self-assembly of polymers results in microphase-separated domains at a length scale of ca. 10–100 nm. However, typically the structures are local, that is, they lack common alignment and therefore the materials are macroscopically disordered. To achieve overall alignment in bulk matter, large electric, magnetic and flow fields have been used11,12,13,14. In films, graphoepitaxy and surface templating allow long-range order15,16,17,18,19. Also extensive thermal or solvent annealing20 classically improve the overall order. However, achieving aligned and globally ordered self-assembled polymer systems organized up to millimetre length scale through simple, rapid and technologically relevant ways remains a fascinating challenge, in order to exploit the properties of the nanometre structures at macroscopic scale.
Towards tackling such a problem, we designed self-assemblies involving polymers and low-molecular-weight rod-like mesogens where the latter ones would pack in strictly parallel fashion leading to an overall broken symmetry, where the interface curvature in the local and global scale between the rods and the polymer matrix is minimized, and where plasticization by supramolecular binding of low-molecular-weight units improves dynamics to reach the equilibrium. Such features may synergistically result in an overall spontaneous organization up to macroscale without the need of external stimuli. The aimed strong repulsion of the mesogenic rods from the organic polymeric domains could be achieved by the use of perfluorinated molecules. To this purpose, we used iodoperfluoroalkanes (IPFAs) that can be halogen-bonded to polymers possessing electron-donor sites21,22,23,24,25. However, in spite of its strength, specificity and directionality, halogen bonding has seldom been used for directing polymer self-assembly26,27.
Here we report, as a model material, the amine hydrochloride derivative of a 4-arm polyethylene glycol, that is, C-[CH2-(OCH2CH2)29-NH3+Cl−]4 (molecular weight of 5 kDa, a branched semicrystalline polymer, see Methods), halogen-bonded to mesogenic IPFAs (Fig. 1). A star-shaped polymer is selected aiming to reduce coiling and entanglement tendency, which we expect to promote ordering and to allow a dense packing of the noncovalently bonded IPFA chains. In IPFAs, the fluorine atoms inductively boost the electron accepting ability of the terminal iodine, which promotes halogen bonding. The long-chain IPFAs adopt all-trans conformations, showing rod-like behaviour and exceptionally high repulsion from hydrocarbons (fluorophobic effect)28. However, selection of proper perfluoroalkyl chain lengths of IPFA turned out to be subtle: too short chains lead to excessive volatility thus reducing the stability of the complexes, whereas too long chains do not allow solution processing to prepare the complexes. Therefore, 1-iodoperfluorodecane (I-C10F21) and 1-iodoperfluorododecane (I-C12F25) are here selected. Halogen bonds are expected to occur between the chloride anions and the iodine atoms (Fig. 1). The simplicity and rapidity of the method described here demonstrate that the synergistic use of halogen bonding and fluorophobic effect constitutes a major advancement to obtain nanostructured polymeric materials aligned up to the macroscopic scale.
Results
Stoichiometry of the halogen-bonded complexes
In small-molecule co-crystals, the chloride anions may behave as either mono- or bi-dentate halogen bond acceptors (see Supplementary Note 1). Therefore, to establish the nominal halogen bonding stoichiometry in the present polymeric case, we first used a facile process, where powders of the polymer and I-C10F21 were ground together in a mortar and pestle for 10 min using 1:1 and 1:2 molar ratios of Cl−:I (see Methods). Fourier transform infrared spectroscopy (FTIR) showed that the C-F stretching mode at 1,198 cm−1 and the I-CF2 deformation mode at 631 cm−1 of I-C10F21 exhibit a blue shift of around 8 and 11 cm−1 upon complexation to the polymer (Fig. 2a). Similar blue shift is also observed for the C–O–C bending at 1,100 cm−1, whereas a smaller blue shift of ca. 2 cm−1 involves the stretching mode of the polymer at 2,880 cm−1. An intensity decrease in the complex is observed in the region 3,700–3,300 cm−1, potentially associated with the modified interaction environment of the ammonium chloride group. The spectral changes support supramolecular interaction. Importantly, the 1:1 and 1:2 molar ratios of Cl−:I yielded almost indistinguishable FTIR spectra. Therefore, we suggest that each chloride nominally binds one IPFA molecule, and in the process of grinding the Cl−:I 1:2 composition, the additional IPFA sublimes off the powder owing to its high vapour pressure. Similar FTIR spectra are observed in the complex with I-C12F25 (see Supplementary Fig. 1).
(a) Attenuated total reflectance (ATR) FTIR spectra in the relevant regions. (b) XPS spectra showing the shifts in I 3d and (c) Cl 2p orbital binding energies upon halogen bonding. (d) DSC thermographs upon the first heating cycle at 10 °C min−1 of the starting compounds and the C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21 complex prepared by grinding.
Further evidence of the halogen bond between C-[CH2-(OCH2CH2)29-NH3+Cl−]4 and I-C10F21 at the nominal 1:1 Cl−:I molar ratio is given by X-ray photoelectron spectroscopy (XPS) (Fig. 2b,c). The complexes were prepared upon mixing the starting materials in isopropanol (see Methods). We expected that upon halogen bonding the electron density of the I atom (halogen bond donor) would increase while that of the Cl− anion (halogen bond acceptor) would decrease, resulting in a downshift of the binding energies for the I 3d doublet of I-C10F21 and an upshift of the Cl 2p doublet in C-[CH2-(OCH2CH2)29-NH3+Cl−]4. Measuring the XPS spectrum of highly volatile I-C10F21 posed challenges in our setup under its high-vacuum conditions and focused beam, and therefore the binding energy for the I 3d doublet was measured using the less volatile I-C12F25. As XPS only probes the nearest atomic neighbourhood of the iodine, the slightly different alkyl tail length is not expected to be relevant in this case. Indeed, the peaks at 620.4 and 632 eV, corresponding to I 3d, shift to 618.7 and 630.2 eV, respectively, while the Cl 2p doublet in C-[CH2-(OCH2CH2)29-NH3+Cl−]4 shifts to higher energies (see Supplementary Note 2). These energy shifts agree with literature values for related interactions27, supporting that chloride ion and the IPFA are halogen bonded.
Liquid-crystalline behaviour
Differential scanning calorimetry (DSC) shows a single sharp endothermic peak at 67 °C for I-C10F21 upon heating (Fig. 2d). As polarized optical microscopy (POM) (Fig. 3b) shows transition from an optically anisotropic state to an isotropic state upon heating, we assign this as melting. C-[CH2-(OCH2CH2)29-NH3+Cl−]4 polymer shows the main melting at 41 °C as well as a small exothermic peak at around 33 °C (Fig. 2d) due to cold crystallization and related melting endotherm at 37 °C, signalling polymorphism. The corresponding POM images (Fig. 3a) show growth of spherulitic crystalline domains upon cooling after having been heated above its melting temperature. On the other hand, the complex C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21 shows a main endothermic peak at 44 °C (Fig. 2d) corresponding to the transition from crystalline solid to the liquid-crystalline phase (smectic A phase, SmA), as supported by POM at 47 °C, Fig. 3c. POM (Fig. 3d) agrees with the DSC data as the optical anisotropy is suppressed upon heating from 48 to 54 °C, and becomes again observable upon cooling. No sign of phase-separated I-C10F21 or C-[CH2-(OCH2CH2)29-NH3+Cl−]4 polymer was observed. The present finding shows the first observation of supramolecular halogen-bonded liquid crystals involving polymers.
(a) C-[CH2-(OCH2CH2)29-NH3+Cl−]4 after heating to 38 and 47 °C, subsequent cooling and resting at room temperature for about 5 min. The growth of crystalline spherulitic domains is observed over time upon cooling below the polymer’s melting temperature. (b) I-C10F21. (c) C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21 complex at 47 °C, suggesting smectic A-type liquid crystallinity (SmA), as prepared by grinding. (d) C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21 complex after heating to 39, 48 and 54 °C, and subsequent cooling to 48 °C. The complex is prepared from isopropanol solution. Scale bars, 200 μm.
Halogen-bonded mesogen directed self-assembly
The structures were elucidated by using small-angle X-ray scattering (SAXS), X-ray diffraction (XRD) and transmission electron microscopy (TEM) (Fig. 4). As a reference, the pristine C-[CH2-(OCH2CH2)29-NH3+Cl−]4 shows a nanoscale periodicity due to the charge clustering of the end-group ammonium salts, as SAXS shows a broad reflection at q2=0.067 Å−1 (periodicity of ca. 9.4 nm) with a faint second-order reflection (Fig. 4a, plot 2). Two-dimensional (2D) SAXS confirms that the material is macroscopically isotropic with a poor overall alignment. Peaks q5 and q6 (Fig. 4b, plot 2) are signatures of the crystalline structure within C-[CH2-(OCH2CH2)29-NH3+Cl−]4. Pure I-C10F21 shows a reflection at q1=0.38 Å−1 (periodicity of ca. 1.6 nm) (see Fig. 4a, plot 1) and its higher-order peaks in the wide-angle region (Fig. 4b, plot 1). On the other hand, the complex C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21, as prepared from isopropanol solution, shows a highly ordered lamellar structure with the primary SAXS peak at q3=0.055 Å−1 and a series of higher-order reflections 2q3, 3q3, 4q3 and 5q3 (Fig. 4a, plot 3). Compared with the pure polymer, the periodicity has increased to 11.4 nm, due to the intercalation of the halogen-bonded I-C10F21 mesogens (Fig. 4d). XRD shows narrow reflections at q4=1.4 Å−1, 2q4 and 3q4 corresponding to a well-defined spacing of 0.45 nm (Fig. 4b, plot 3), which suggests tight lateral packing driven by the perfluoroalkyl rod-like chains. Peak q5 (Fig. 4b, plot 3) indicates crystallization within the C-[CH2-(OCH2CH2)29-NH3+Cl−]4 domains. 2D SAXS shows improved overall alignment in comparison with the pure polymer, which is also confirmed by TEM (Fig. 4c). As C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21 shows no SAXS reflections of the starting materials, macroscopic phase separation is not suggested.
(a) SAXS patterns of I-C10F21 (1), C-[CH2-(OCH2CH2)29-NH3+Cl−]4 (2, indicating poor self-assembly by segregation of the ionic end-groups from the polyethylene glycol-core), and supramolecular complex C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21 (3, showing highly ordered self-assembly). Corresponding 2D SAXS (right) shows poor overall order for C-[CH2-(OCH2CH2)29-NH3+Cl−]4 and high overall order for C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21. (b) XRD shows (1) fluoroalkyl chain order for pure I-C10F21, (2) Polyethylene glycol-crystallization for pure C-[CH2-(OCH2CH2)29-NH3+Cl−]4 and (3) lateral packing due to the fluoroalkyl chains in the supramolecular complex C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21. (c) TEM micrograph of C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21, showing lamellar smectic-like order with periodicity of 11.4 nm (scale bar, 50 nm). (d) Cartoon showing the self-assembly mechanism of the halogen-bonded complex.
The high order in the complex C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21 observed by X-ray scattering and TEM can be explained by the interplay between various interactions. First, I-C10F21 interacts through a halogen bond with the chloride ion of the ammonium chloride end-capped polyethylene glycol polymer chains. Then, the terminal perfluoroalkyl rod-like chains pack tightly into fluorous-segregated layers, resulting in a long-range lamellar order, which can even be improved by annealing at mild conditions. Indeed, in Fig. 5 we show that vacuum treatment at sufficiently low temperature does not cause removal of I-C10F21 from the complexes. For that, C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C10F21 was kept in high vacuum for 2 days at 33 oC, which is below the melting point of the crystalline domains within the C-[CH2-(OCH2CH2)29-NH3+Cl−]4 phase. In these conditions the complex remains stable, as seen from the C-F (1,206 cm−1) stretching and I-CF2 (643 cm−1) deformation bands in Fig. 5a as compared with those shown in Fig. 2a. Such a vacuum annealing at low temperature even slightly improves the structural order and overall alignment of the system, compared with non-annealed sample (see Fig. 5b), showing primary SAXS peak at q1=0.052 Å−1 and a series of higher-order reflections, as shown in Fig. 5b.
However, I-C10F21 can be completely removed upon thermal treatment at sufficiently high temperatures, as demonstrated by the thermogram obtained in the second heating cycle (Supplementary Fig. 2), which coincides with that of pure C-[CH2-(OCH2CH2)29-NH3+Cl−]4 (Fig. 2d). It appears, therefore, that all of I-C10F21 has been removed during the heating to 90 °C in the first cycle. It is important to note that at mild conditions, that is, vacuum and mild temperatures, the complex remains stable.
We next aimed to further reduce the interfacial curvature and undulations. Therefore, we incorporated a longer fluorous tail by using I-C12F25 to increase the repulsion to the organic domains. However, since 1-iodoperfluorododecane is poorly soluble in common organic solvents, we could not use solution methods to prepare the complexes. Here we identified a vapour-phase procedure as a facile and new processing method to prepare the complex by exposing drop-cast films of C-[CH2-(OCH2CH2)29-NH3+Cl−]4 to vapours of I-C12F25 at ca. 100 °C for 3 h. This allowed the volatile I-C12F25 to penetrate the melt polymer and become absorbed by forming halogen bonds. The films can even be relatively thick, even a fraction of millimetre. After the reaction, the material was returned back to room temperature and analysed by XPS (Fig. 2b,c) and FTIR (Supplementary Fig. 1; Supplementary Note 3), which showed occurrence of halogen bonding of I-C12F25 to C-[CH2-(OCH2CH2)29-NH3+Cl−]4.
Such longer perfluoroalkyl chains led to increased thickness of the fluorous layer, as SAXS showed the main reflection at q2≈0.05 Å−1 (Fig. 6a, plot 2), indicating that periodicity has increased from 9 nm (Fig. 6a, plot 1) to 12.5 nm. This agrees with the halogen bond-driven intercalation of I-C12F25 upon complexing with the polymer, followed by interdigitation and packing of the perfluoroalkyl chains, which result into a highly ordered and oriented lamellar structure, as demonstrated by higher-order reflections 2q2, 3q2 and 4q2 (Fig. 6a, plot 2) and 2D SAXS (Fig. 6b). However, the most striking observation was based on TEM. TEM micrographs on several positions provided direct evidence that there is an overall alignment of the C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C12F25 complex, which extends to the millimetres (Fig. 6d), that is, six orders of magnitude higher than the nanometric-scale interactions and assembly, as driven by the halogen bonding and fluorophobic effect. The starting polymer (Fig. 6c) or complexes with IPFAs shorter than 10 carbon atoms do not show such an overall alignment. The I-C12F25 can also be completely removed upon combined vacuum/thermal treatment, as confirmed by FTIR analysis (Supplementary Fig. 3; Supplementary Note 4) and SAXS, which showed a shrinking of the periodicity from 12.5 nm to 9.5 nm (Fig. 6a,b plot 3).
(a,b) SAXS patterns showing weak nanometer-scale order of the pristine C-[CH2-(OCH2CH2)29-NH3+Cl−]4 (1); halogen bonding leads to an increased periodicity by complexation of I-C12F25 from vapours at ca. 100 °C to C-[CH2-(OCH2CH2)29-NH3+Cl−]4 and allows highly ordered self-assembly with equidistant reflections based on SAXS (2) and high overall alignment. By heating and vacuum treatment, the halogen-bonded I-C12F25 is removed and the original periodicity is approximately recovered, however, showing better residual alignment of C-[CH2-(OCH2CH2)29-NH3+Cl−]4 (3). (c) TEM micrographs at various points of pure C-[CH2-(OCH2CH2)29-NH3+Cl−]4 showing lack of macroscale alignment. (d) TEM micrographs of the complex C-[CH2-(OCH2CH2)29-NH3+Cl−]4·4 I-C12F25, denoted as its nominal composition, after 3 h of exposure to I-C12F25 vapours. The micrographs were taken at spots ~0.5 mm apart from each other, and show exceptionally well-ordered lamellar nanostructures with overall macroscale order at the millimetre scale. Scale bar, 100 nm (c,d).
Discussion
In summary, the halogen bond-driven self-assembly of a model polymer with IPFAs resulted in a very efficient spontaneous organization up to millimetre length scale without applying external aligning fields. The concept is based on the guiding effect of halogen bonding, owing to its strength and directionality, coupled with the packing of perfluoroalkyl mesogenic rods, whose extremely high repulsion from the organic polymeric phase promotes straight interfaces, and high dynamics due to their low molecular weight, which promotes plasticization and favours progress towards energy minimum. We believe that this approach is broad and not limited to the materials reported here, as many polymers possess halogen bond-accepting groups. The facile reversibility of the binding to the IPFAs opens up a plethora of new possibilities for the processing, structure control and molecular imprinting of polymeric materials, as well as for the tuning of materials properties, such as directional wetting, transport, optical, electrical and ionic properties.
Methods
Materials
C-[CH2-(OCH2CH2)29-NH3+Cl−]4 with Mn=5,000 g mol−1 was purchased from Jenkem Technology USA Inc. and used without additional purification. Iodoperfluorodecane (I-C10F21) and iodoperfluorododecane (I-C12F25) were purchased from Apollo Scientific Ltd. and used without additional purification. The solvents in this study were used as received from commercial suppliers.
FTIR
Attenuated total reflectance-FTIR spectra were recorded on a Nicolet Nexus FTIR spectrometre equipped with a UATR unit. The values were given in wavenumbers and were rounded to 1 cm−1 upon automatic assignment.
DSC
DSC analyses were performed on a Mettler Toledo DSC823e differential scanning calorimeter, using aluminium 40 μl sample pans and Mettler STARe software for calculation. The measurements were carried out from 25 to 100 °C, with a heating rate of 10 °C min−1 (the first cycle); after this, the sample was cooled down to −20 °C with a cooling rate of 10 °C min−1 and heated again to 100 °C (the second cycle), all at 10 °C min−1. Calibration was carried out using an indium standard and an empty pan, sealed in the same way as the sample. Sample weights of about 1–4 mg were used in the measurements.
POM
Liquid crystal textures (Fig. 3) were studied with an Olympus BX51 POM equipped with a Linkam Scientific LTS 350 heating stage and a Sony CCD-IRIS/RGB colour video camera connected to a Sony video monitor CMA-D2. POM images were also taken as a function of temperature (Fig. 3) using a Leica DM4500 P polarization microscope with a Leica DFC420 digital camera.
XPS
XPS spectra were recorded by Surface Science Instruments SSX-100 spectrometre using monochromated Al Kα X-rays and operated at 100 W.
SAXS
SAXS measurements were performed with a setup consisting of a Bruker Microstar microfocus X-ray source with a rotating anode (λ=1.54 Å) and Montel optics. The beam from the X-ray source was further adjusted by four sets of four-blade slits, which resulted in an ~1 × 1-mm beam at the sample position. The scattered beam was detected with the Hi-Star 2D area detector (Bruker). For measurements, the sample to detector distance was set to 0.59 m to capture the desired length scale in the measurements. The measured 2D scattering data is azimuthally averaged to obtain one-dimensional SAXS data.
XRD
XRD spectra were recorded using XRD; Panalytical X’Pert PRO MPD. CuKα1 radiation (45 kV, 40 mA, with a wavelength of 0.154060, nm) was used.
TEM
TEM specimens were prepared by cryo-microtoming samples below −80 °C with a Leica EM UC7 ultramicrotome to 70–150 nm thick sections. During preliminary TEM measurements, it was noticed that sections introduced from room temperature to the high vacuum (~10−6 bar) melted and formed micrometre-sized droplets when exposed under the beam. Therefore for the actual TEM investigations, the specimens were cooled down to −187 oC after sectioning and cryotransferred to the TEM device in order to keep the sections relatively intact.
Preparation of the solid mixtures by grinding
In a typical preparation procedure, C-[CH2-(OCH2CH2)29-NH3+Cl−]4 (20 mg, 4 × 10−3 mmol) and I-C10F21 (10 mg, 1.6 × 10−2 mmol or 20 mg, 3.2 × 10−2 mmol, respectively) were ground in an agate mortar for 5 min. The mixture was allowed to stand overnight at room temperature, then ground for further 5 min and finally characterized by DSC, attenuated total reflectance-FTIR and POM.
Preparation of the iodoperfluorodecane complex from solution
I-C10F21 allowed preparation of complexes with C-[CH2-(OCH2CH2)29-NH3+Cl−]4 from solution, as isopropanol was found to be a common solvent for both compounds. The complexes were prepared by dissolving I-C10F21 and C-[CH2-(OCH2CH2)29-NH3+Cl−]4 in isopropanol in a final concentration of 10 mg ml−1 and stirred overnight. After stirring, the solvent was evaporated at room temperature and atmospheric pressure in a glass vial.
Preparation of the complex by exposing to iodoperfluorododecane vapours
No common solvent was found for I-C12F25 and C-[CH2-(OCH2CH2)29-NH3+Cl−]4. The latter compound was dissolved in isopropanol and cast on a glass substrate to form a film of sub-millimetre thickness by solvent evaporation. Alternatively, the C-[CH2-(OCH2CH2)29-NH3+Cl−]4 solution was placed on a Teflon plate and inserted on a glass substrate or into a flask. In both cases the melted material was exposed to I-C12F25 vapours at 100 °C for 3 h. After cooling to room temperature, the material was detached from the substrate, collected and characterized. No separate I-C12F25 or C-[CH2-(OCH2CH2)29-NH3+Cl−]4 XPS peaks (Fig. 2b,c) were detected, which suggests that the halogen-bonded complex is stoichiometric. Note that even relatively thick samples could be complexed.
Additional information
How to cite this article: Houbenov, N. et al. Halogen-bonded mesogens direct polymer self-assemblies over millimetre length scale. Nat. Commun. 5:4043 doi: 10.1038/ncomms5043 (2014).
References
Abetz, V. & Simon, P. F. W. Block copolymers I. Adv. Polym. Sci. 189, 125–212 (2005).
Matyjaszewski, K. & Tsarevsky, N. V. Nanostructured functional materials prepared by atom transfer radical polymerization. Nat. Chem. 1, 276–288 (2009).
Matsushita, Y., Takano, A., Hayashida, K., Asari, T. & Noro, A. Hierarchical nanophase-separated structures created by precisely-designed polymers with complexity. Polymer 50, 2191–2203 (2009).
Bates, F. S. et al. Multiblock polymers: panacea or pandora’s box? Science 336, 434–440 (2012).
Gröschel, A. H. et al. Precise hierarchical self-assembly of multicompartment micelles. Nat. Commun. 3, 710 (2012).
Aida, T., Meijer, E. W. & Stupp, S. I. Functional supramolecular polymers. Science 335, 813–817 (2012).
ten Brinke, G., Ruokolainen, J. & Ikkala, O. Supramolecular materials based on hydrogen-bonded polymers. Adv. Polym. Sci. 207, 113–177 (2007).
Rauwald, U. & Scherman, O. A. Supramolecular block copolymers with cucurbit[8]uril in water. Ang. Chem. Int. Ed. 47, 3950–3953 (2008).
Mugemana, C. et al. Metallo-supramolecular diblock copolymers based on heteroleptic cobalt(III) and nickel(II) bis-terpyridine complexes. Chem. Commun. 46, 1296–1298 (2010).
Houbenov, N. et al. Self-assembled polymeric supramolecular frameworks. Angew. Chem. Int. Ed. 50, 2516–2520 (2011).
Mansky, P. et al. Large-area domain alignment in block copolymer thin films using electric fields. Macromolecules 31, 4399–4401 (1998).
Liedel, C., Pester, C. W., Ruppel, M., Urban, V. S. & Böker, A. Beyond orientation: the impact of electric fields on block copolymers. Macromol. Chem. Phys. 213, 259–269 (2012).
Gopinadhan, M., Majewski, P. W., Beach, E. S. & Osuji, C. O. Magnetic field alignment of a diblock copolymer using a supramolecular route. ACS Macro. Lett. 1, 184–189 (2012).
Chen, Z. R., Kornfield, J. A., Smith, S. D., Grothaus, J. T. & Satkowski, M. M. Pathways to macroscale order in nanostructured block copolymers. Science 277, 1248–1253 (1997).
Segalman, R. A., Yokoyama, H. & Kramer, E. J. Graphoepitaxy of spherical domain block copolymer films. Adv. Mater. 13, 1152–1155 (2001).
Kim, S. O. et al. Epitaxial self-assembly of block copolymers on lithographically defined nanopatterned substrates. Nature 424, 411–414 (2003).
Bita, I. et al. Graphoepitaxy of self-assembled block copolymers on two-dimensional periodic patterned templates. Science 321, 939–943 (2008).
Tang, C., Lennon, E. M., Fredrickson, G. H., Kramer, E. J. & Hawker, C. J. Evolution of block copolymer lithography to highly ordered square arrays. Science 322, 429–432 (2008).
Park, S. et al. Macroscopic 10-terabit–per–square-inch arrays from block copolymers with lateral order. Science 323, 1030–1033 (2009).
Kim, S. H., Misner, M. J. & Russell, T. P. Solvent-induced ordering in thin film diblock copolymer/homopolymer mixtures. Adv. Mater. 16, 2119–2123 (2004).
Cavallo, G. et al. Halogen bonding: a general route in anion recognition and coordination. Chem. Soc. Rev. 39, 3772–3783 (2010).
Metrangolo, P., Meyer, F., Pilati, T., Resnati, G. & Terraneo, G. Halogen bonding in supramolecular chemistry. Angew. Chem. Int. Ed. 47, 6114–6127 (2008).
Metrangolo, P. & Resnati, G. Halogen versus hydrogen. Science 321, 918–919 (2008).
Metrangolo, P. et al. Nonporous organic solids capable of dynamically resolving mixtures of diiodoperfluoroalkanes. Science 323, 1461–1464 (2009).
Xu, J., Liu, X., Lin, T., Huang, J. & He, C. Synthesis and self-assembly of difunctional halogen-bonding molecules: a new family of supramolecular liquid-crystalline polymers. Macromolecules 38, 3554–3557 (2005).
Bertani, R. et al. Supramolecular route to fluorinated coatings: self-assembly between poly(4-vinylpyridines) and haloperfluorocarbons. Adv. Mater. 14, 1197–1201 (2002).
Wang, F., Ma, N., Chen, Q., Wang, W. & Wang, L. Halogen bonding as a new driving force for layer-by-layer assembly. Langmuir. 23, 9540–9542 (2007).
Wilson, C. J., Wilson, D. A., Feiring, A. E. & Percec, V. Disassembly via an environmentally friendly and efficient fluorous phase constructed with dendritic architectures. J. Polym. Sci. A Polym. Chem. 48, 2498–2508 (2010).
Acknowledgements
The European Research Council is acknowledged for the Starting Grant ERC-2012-StG_20111012 FOLDHALO (Grant Agreement Number 307108) (P.M.) and Advanced Grant MIMEFUN (O.I.). This work was partially supported by the Academy of Finland under Center of Excellence in Molecular Engineering of Biosynthetic Hybrid Materials Research. Matti Lehtimäki is acknowledged for his XRD support. This work was supported by the Academy of Finland Centre of Excellence (CoE) programme for 2014–2019.
Author information
Authors and Affiliations
Contributions
N.H. and M.P. performed experimental work, except that J.H. measured the samples by electron microscopy, J.S. performed the XPS experiments, and V.D. contributed with FTIR measurements. N.H., R.M., O.I., G.R. and P.M. conceived the experiments. All authors contributed to results, discussion and manuscript writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
Supplementary Figures 1-3, Supplementary Notes 1-4 and Supplementary References (PDF 882 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Houbenov, N., Milani, R., Poutanen, M. et al. Halogen-bonded mesogens direct polymer self-assemblies up to millimetre length scale. Nat Commun 5, 4043 (2014). https://doi.org/10.1038/ncomms5043
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms5043
This article is cited by
-
High-resolution crystal structure of a 20 kDa superfluorinated gold nanocluster
Nature Communications (2022)
-
Supramolecular macrocycles reversibly assembled by Te…O chalcogen bonding
Nature Communications (2016)
-
Synthesis and characterization of hydrogen bonded liquid crystalline block copolymers based on a Schiff base moiety
Journal of Polymer Research (2015)
-
Synthesis of dispersive iron or iron–silver nanoparticles on engineered capsid pVIII of M13 virus with electronegative terminal peptides
Journal of Nanoparticle Research (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.