Abstract
Across the periodic table the trans-influence operates, whereby tightly bonded ligands selectively lengthen mutually trans metal–ligand bonds. Conversely, in high oxidation state actinide complexes the inverse-trans-influence operates, where normally cis strongly donating ligands instead reside trans and actually reinforce each other. However, because the inverse-trans-influence is restricted to high-valent actinyls and a few uranium(V/VI) complexes, it has had limited scope in an area with few unifying rules. Here we report tetravalent cerium, uranium and thorium bis(carbene) complexes with trans C=M=C cores where experimental and theoretical data suggest the presence of an inverse-trans-influence. Studies of hypothetical praseodymium(IV) and terbium(IV) analogues suggest the inverse-trans-influence may extend to these ions but it also diminishes significantly as the 4f orbitals are populated. This work suggests that the inverse-trans-influence may occur beyond high oxidation state 5f metals and hence could encompass mid-range oxidation state actinides and lanthanides. Thus, the inverse-trans-influence might be a more general f-block principle.
Similar content being viewed by others
Introduction
The trans-influence is a long-established, well-documented concept of broad relevance across inorganic chemistry1,2. This thermodynamic ground-state phenomenon classically occurs in square-planar and pseudo-octahedral d-block complexes where tightly bonded ligands selectively lengthen mutually trans metal–ligand bonds. The trans-influence is fundamentally important and underpins the trans-effect3, a kinetic rate effect where the order of substitution of ligands at a metal centre can be controlled; this is a key parameter to control, for example, the syntheses of cis- or trans-[PtCl2(NH3)2], whose isomerism is important regarding cancer treatment4. Although the bonding of lanthanide(III) and low/mid oxidation state early actinide ions is considered more ionic than in the d-block, there are crystallographic, and in some instances computationally supported, examples of complexes where metrical parameters are consistent with the presence of the trans-influence5,6,7,8,9,10,11,12,13,14,15,16.
In high oxidation state actinide complexes the opposite phenomenon of the inverse-trans-influence (ITI) can be found17,18,19,20,21. Here, strongly donating ligands that normally adopt cis orientations to avoid destabilizing the respective metal–ligand bonds via the trans-influence in fact reside trans to one another and even mutually reinforce each other. The classical, dominant example of the ITI is the uranyl(VI) dication, {UO2}2+, that adopts a trans-linear geometry and is chemically robust because of strong, ITI-strengthened uranium–oxygen bonds20. Indeed, linear trans-dioxo actinyls {AnO2}n+ (An=U, Np, Pu) are well known and prevalent, but trans-dioxos in the d-block are unusual and require strong equatorial σ-donor ligands to weaken the metal-oxo linkages sufficiently to enable them to reside mutually trans. Two isostructural complexes that demonstrate the trans-influence and the ITI are [MoO2Cl2(OPPh3)2] (I)22 and [UO2Cl2(OPPh3)2] (II)23, respectively (Fig. 1); in the former the oxos are cis, whereas in the latter they are trans. The ITI often plays a structure-dictating role, but this is not a criterion that must be met to make invoking the ITI valid; there are examples of high valent uranium complexes where ligands are constrained by their own architecture such that they have no choice but to place donor groups trans to a strongly donating ligand like a nitride or oxo, but despite this they present very short metal–ligand distances despite their unfavourable bonding situation. For example, in the complexes [U(TrenTIPS)(E)] [TrenTIPS=N(CH2CH2NSiPri3)3; E=N (III), O (IV)] (Fig. 1)24,25 the U–Namine distances are short at 2.465(5) and 2.482(6) Å, respectively, despite being trans to nitride and oxo ligands, whereas such U–Namine distances are normally 2.5–2.7 Å (ref. 26). In these systems there is no ITI structure-dictating role, and as part of a polydentate Tren-ligand with minimal reorganization energy27 the Tren amines are forced to be unfavourably trans to a nitride or oxo28, but the U–Namine distances are short, not long, that credibly invokes the ITI.
Complexes I and II are identical except for the metal ion and the molybdenum complex exhibits cis-oxos whereas the uranium complex has trans-oxos, consistent with the trans-influence and inverse-trans-influence, respectively. Complexes III and IV both exhibit short uranium–amine distances despite being trans to hard, charge-loaded nitride and oxo ligands that is not consistent with the trans-influence but conversely invokes the inverse-trans-influence.
The origin of the ITI is complex, but is in part rationalized on the basis that in high oxidation state early actinides 6p orbitals are semi-core and transfer electron density to vacant 5f orbitals, creating an electron hole that is compensated for by additional donation of electron density from trans ligands17,18,19,20,21. For many years the ITI was limited to uranyl(VI) complexes20 or structurally analogous complexes such as [UOCl5]− (ref. 29), but in recent years a limited number of uranium(V) and (VI) ITI complexes have emerged24,25,30,31,32,33. The unifying theme has been high oxidation state (V or VI) metal complexes combined with hard, polarizing, charge-loaded oxo, imide and nitride ligands. Because it is limited to high oxidation state early actinides, the question of whether the ITI is a niche concept or in fact has a broader underpinning role for the f-block has remained unanswered for around a quarter of a century in an area with few unifying rules.
When considering if the ITI could have a broader basis, it would have to be demonstrated to operate over a larger range of oxidation states, and be expanded beyond actinides to include the lanthanides. The IV oxidation state is the logical next step to take in terms of the general synthetic availability of uranium and thorium complexes, as the only two actinides that can be routinely handled without specialist facilities, and also because a IV oxidation state opens the door to extend this concept to the lanthanides; cerium has an accessible IV oxidation state under normal conditions, presenting the opportunity to compare cerium, uranium and thorium together34,35. Although hard, formally di- and trianionic oxygen and nitrogen ligands have so far exclusively supported the ITI with high oxidation state metals, by moving to a mid-range oxidation state a softer, isoelectronic dianionic carbon-based ligand might be arguably desirable to approximately maintain the relative energy matching of frontier metal and ligand orbitals, and we note that the only examples of uranium and even thorium in the +2 oxidation state under ambient conditions are stabilized by carbon-based ligands36,37,38. Indeed, carbon should be a good ligand for the ITI more generally because of its generally high-lying frontier orbitals compared with uranium31. However, a paucity of synthetically accessible families of complexes where the metal can be varied in a common mid-range oxidation state has limited testing the above hypothesis.
Here, we report the realization of our aim by the synthesis of cerium, uranium and thorium bis(carbene) complexes that exhibit linear C=M=C cores supported by the BIPMTMS ligand [BIPMTMS={C(Ph2PNSiMe3)2}2−]. Although the C=M=C units are trans and thus would conventionally be expected to present long M=C distances, they in fact exhibit exceedingly short M=C distances, and for cerium among the shortest experimental Ce-C distance on record. Theoretical calculations reveal that when the pseudo-core 5p (cerium) or 6p (uranium or thorium) orbitals are isolated from the valence manifold the M=C distances increase. Taken together with the short M=C distances, and considering that they are disposed trans, this suggests that the ITI may extend beyond high oxidation state 5f metals to operate in mid-range oxidation state f-element metal complexes with appropriate ligand matching. Investigations of hypothetical praseodymium(IV) and terbium(IV) analogues suggest similar but increasingly diminished ITI phenomena.
Results
Synthesis
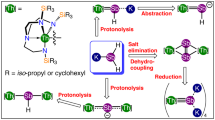
Treating the cerium(III) carbene-methanide complex [Ce(BIPMTMS)(BIPMTMSH)] (1Ce)39 with benzyl potassium and 18-crown-6 ether (18C6) in tetrahydrofuran (THF) gives the yellow cerium(III) bis(carbene) complex [Ce(BIPMTMS)2][K(18C6)(THF)2] (2Ce) in 52% yield (Fig. 2). Although cerium(IV) is regarded as a difficult oxidation state to access in an organometallic context because cerium(IV) is oxidizing and organometallic ligands are reducing, we find that oxidation of 2Ce can be straightforwardly accomplished by AgBPh4 to give the green cerium(IV) bis(carbene) [Ce(BIPMTMS)2] (3Ce) in 43% yield after work-up and recrystallization (Fig. 2). The oxidation of 2Ce to 3Ce is so favourable that even small traces of dry air will effect oxidation. This suggests that the two carbenes together are well suited to stabilizing cerium(IV) and producing a robust C=Ce=C unit, cf, the stability of the ITI-stabilized uranyl O=U=O dication. The corresponding uranium and thorium bis(carbene) complexes [M(BIPMTMS)2] (M=U, 3U; Th, 3Th) were prepared by a different methodology (Fig. 2). The mono(carbene) dichloride complexes [M(BIPMTMS)Cl3Li(THF)2] (M=U, 4U; Th, 4Th)40,41 were converted to the corresponding dialkyls [M(BIPMTMS)(CH2SiMe3)2] (M=U, 5U; Th, 5Th)42; subsequent thermolysis with BIPMTMSH2 gave 3U and 3Th in 75% and 52% yields as brown and colourless crystals, respectively, after work-up.
The cerium(III) carbene–methanide complex 1Ce reacts with benzyl potassium and 18C6 in THF to give the cerium(III) bis(carbene) separated ion pair complex 2Ce with elimination of toluene. Complex 2Ce is readily oxidized by silver tetraphenylborate to give the cerium(IV) bis(carbene) complex 3Ce with elimination of elemental silver and potassium 18-crown-6 ether bis(THF) tetraphenylborate. The uranium and thorium carbene complexes 4U and 4Th are converted to the corresponding carbene dialkyl complexes 5U and 5Th by salt elimination with two equivalents of trimethylsilylmethyl lithium. Complexes 5U and 5Th react with BIPMTMSH2 by alkane elimination to give the uranium and thorium bis(carbene) complexes 3U and 3Th, respectively. Attempts to prepare the praseodymium(IV) and terbium(IV) analogues of 3Ce resulted in elimination of potassium 18-crown-6 bis(THF) tetraphenylborate and insertion of silver into the bis(carbene) complexes to give 6Pr and 6Tb.
Characterization data
The 1H nuclear magnetic resonance (NMR) spectra of 3Ce and 3Th span 0–10 p.p.m. and are characteristic of diamagnetic complexes, whereas that of paramagnetic 3U spans ±33 p.p.m. The 31P NMR spectra of 3Ce, 3U and 3Th exhibit resonances at −13.7, −219.7 and 6.3 p.p.m., respectively; the 31P NMR resonance for 3Ce compares well with that of [Ce(BIPMTMS)(ODipp)2] (−10.2 p.p.m., Dipp=2,6-diisopropylphenyl)43, and those of 3U and 3Th are typical of such complexes41,44. The 13C{1H} NMR carbene resonances for 3U and 3Th could not be located, even utilizing 13C-31P 2D NMR techniques, but the equivalent carbenes in 3Ce were observed at 343.5 p.p.m. (JPC=170 Hz); this is more deshielded than that of [Ce(BIPMTMS)(ODipp)2] (324.6 p.p.m., JPC=149 Hz)43, possibly suggesting that the carbenes in 3Ce are donating more strongly to cerium than in [Ce(BIPMTMS)(ODipp)2] despite their trans arrangement. This might be expected for an ITI, and is also within the 200–400 p.p.m. range of covalent transition metal carbenes rather than that observed for ionic yttrium(III) analogues (10–40 p.p.m.)44.
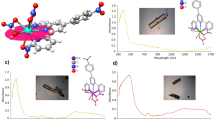
As expected for a 1S0 4f0 cerium(IV) ion, the ultraviolet/visible/near-infrared (UV/Vis/NIR) spectrum of 3Ce exhibits no absorbances in the NIR region where f–f or transitions associated with multiconfigurational character might occur (Supplementary Methods). Two absorptions (Fig. 3a), the broadness of which is a defining feature in many cerium(IV) complexes45, are observed in the visible region at 17,000 and 24,700 cm−1 (ɛ=4,895 and 14,387 M–1 cm–1, respectively), and the latter absorbance is responsible for the green colour of 3Ce. In order to understand the electronic transitions responsible for the green colour of 3Ce we modelled the spectrum using time-dependent density functional theory (TD-DFT) calculations at the statistical average of orbital potentials (SAOP)/zeroth order regular approximation (ZORA)/triple zeta basis set with one polarization function (TZP) level and the profile of the experimental spectrum is reproduced well by these calculations (Supplementary Fig. 1); the calculated absorption bands at 17,000 and 24,700 cm−1 are principally composed of ligand-to-metal charge-transfer from Ce=C π- and σ-combinations to cerium(IV) 4f orbitals. The UV/Vis/NIR spectrum of 3U is characterized by weak (ɛ<80 M–1 cm–1) absorptions over the range 5,000–20,000 cm−1 that are characteristic of the 3H4 electronic manifold of the 5f2 uranium ion (Supplementary Fig. 2), whereas for 3Th the spectrum is featureless in the visible and NIR regions as expected for its 6d05f0 nature.
(a) The UV/Vis/NIR spectrum of 3Ce, illustrating the broad nature of the two principal transitions in the visible region. (b) Cyclic voltammogram of 0.2 mM 3Ce in THF at selected sweep rates (0.1 M [N(Pr)4][BArF4] supporting electrolyte, BArF4=tetrakis(3,5-trifluoromethylphenyl)borate) versus [Fe(Cp)2]0/+ showing a single quasi-reversible redox process assigned to the CeIV/CeIII redox couple. (c) Variable temperature magnetic susceptibility data for 3Ce, 3U and 3Th indicative of populated low-lying paramagnetic states for 3U and diamagnetic, closed-shell assignments for 3Ce and 3Th. (d) Cerium LIII-edge XANES spectrum of the cerium(IV) complex 3Ce (red trace) in comparison with its cerium(III) precursor 2Ce (black trace). As references, spectra of 0.01 M cerium(III) nitrate in water (green trace) and of cerium(IV) dioxide (blue trace) are given. The XANES spectra of 2Ce and 3Ce were recorded at 15 K and the references were recorded at 298 K.
The room temperature cyclic voltammogram of 3Ce reveals a well-resolved, quasi-reversible single redox process at E1/2=−1.63 V versus [Fe(η5-C5H5)2]0/+ assigned as the CeIV/CeIII redox couple (Fig. 3b; for full scan see Supplementary Fig. 3). The CeIV/CeIII redox couple is known to vary widely as a function of ligand environment46, and the reduction potential observed for 3Ce is towards the negative end of reported values, suggesting that the bis(carbene) environment stabilizes cerium(IV), reflecting the facile oxidation of 2Ce to 3Ce. Under the same conditions, no redox processes were observed for 3U or 3Th in the accessible solvent window.
Powdered samples of 3Ce, 3U and 3Th were studied by variable temperature SQUID (superconducting quantum interference device) magnetometry (Fig. 3c and Supplementary Fig. 4). Complex 3U has a χT value of 0.95 cm3 K mol−1 at 298 K (equivalent to 2.77 μB, in agreement with an Evans method solution magnetic moment of 2.61 μB) that decreases below 30 K to a value of 0.4 cm3 K mol−1 at 2 K. Unlike typical magnetization behaviour of uranium(IV) complexes generally (Supplementary Fig. 5), complex 3U retains a high χT value over most of the temperature range and the low-temperature χT value is much higher than expected from temperature-independent paramagnetism alone. This suggests that the crystal field of two strongly donating axial ligands is sufficient to stabilize low-lying paramagnetic states (consistent with low-temperature magnetization data) separated widely from higher energy states (assuming a 3H4 ground term, a strongly axial field would stabilize the |mJ|=4 non-Kramers doublet), and hence even at low temperatures the complex is paramagnetic. This phenomenon has been observed before in uranium(IV) complexes with strongly donating, multiply bonded axial ligands47,48,49,50,51,52. Complexes 3Ce and 3Th are diamagnetic, and the essentially nil or small negative χT slope for the latter rule out any temperature-independent paramagnetism behaviour, suggesting there is no multiconfigurational ground character in 3Ce, and this is also consistent with the absence of low energy absorptions in the optical spectrum. At low temperatures, complex 3Ce has an insignificant χT value (0.003 cm3 K mol−1 at 2 K) and X- and Q-band electron paramagnetic resonance (EPR) spectra are completely silent that contrasts with 4f1 2Ce that exhibits strong EPR features characteristic of cerium(III) (Supplementary Figs 6 and 7).
The characterization data for 3Ce, 3U and 3Th support their IV oxidation state formulations. To unequivocally confirm that 3Ce is a 1S0 complex we subjected it to X-ray absorption spectroscopy techniques. The X-ray photoemission spectroscopy (XPS) of 3Ce is weak because the cerium is a small component of the 151-atom structure, but it exhibits a spectrum characteristic of cerium(IV) in the energy range 870–930 eV (Supplementary Fig. 8). The X-ray absorption near-edge spectroscopy (XANES) of 3Ce reveals two absorptions characteristic of cerium(IV)53, (Fig. 3d), and when all characterization data are taken together the self-consistent picture that emerges is that 3Ce is a closed-shell singlet cerium(IV).
Solid-state structures
The solid-state structures of 3Ce, 3U and 3Th were determined by single crystal X-ray diffraction and were found to be essentially isostructural. Complex 3Ce is illustrated in Fig. 4 and further details of that and all other structurally determined complexes in this study can be found in Supplementary Figs 9–14 and Supplementary Tables 1 and 2. In each case, pseudo-octahedral metal centres present with mutually trans carbenes (3Ce, 176.98(7)°; 3U, 177.5(2)°; 3Th, 176.21(8)°) with deviations from the octahedral ideal because of the BIPMTMS N-M-N bite angles resulting in the imino nitrogen atoms residing above and below the hypothetical equatorial plane. The carbenes adopt essentially planar T-shaped geometries with sum of angles spanning the range 357.23(18) to 360.00(15)° and the metal-BIPMTMS 6-membered chelate rings are essentially planar contrasting with some BIPMTMS complexes where the carbene can be distinctly pyramidal44, including [Ce(BIPMTMS)(ODipp)2]43. In 3Ce, 3U and 3Th, the two BIPMTMS ligands are disposed essentially orthogonally to one another, with dihedral angles between the two N-M-N planes of 92.5(2), 92.2(4) and 91.4(2)°, respectively. The Ce1-C1 and Ce1-C32 distances in 3Ce (2.385(2) and 2.399(3), respectively) are exceedingly short, >0.2 Å shorter than the corresponding distances in 2Ce, and ∼0.04 Å shorter than the Ce=C distances in [Ce(BIPMTMS)(ODipp)2]43 and the special case of cerium confined within an endohedral fullerene54. Significantly, considering they are trans, the Ce-C bond distances in 3Ce are the shortest experimentally determined Ce-C distances to date in discrete molecular compounds, being surpassed only by short (2.247(17)-2.334(15) Å) cerium–carbon distances in periodic ethynediide–halide clusters55,56,57,58,59. Short molecular Ce-C distances have been found in theoretical models of experimentally unknown CeCH2+ and [Cp2CeCH2] that are sterically unimpeded and, in the case of the former, benefit from the reduced electronic repulsion from a net positive charge60,61.
Hydrogen atoms and lattice solvent are omitted for clarity. The structures of 3U and 3Th are very similar. Selected bond lengths (Å) and angles (°): C1-P1 1.664(2), C1-P2 1.664(2), C32-P3 1.665(5), C32-P4 1.663(4), P1-N1 1.6128(18), P2-N2 1.6174(19), P3-N3 1.6247(18), P4-N4 1.6202(18), Ce1-C1 2.385(2), Ce1-C32 2.399(3), Ce1-N1 2.4766(17), Ce1-N2 2.5122(17), Ce1-N3 2.4726(18), Ce1-N4 2.4966(16), P1-C1-P2 164.31(15), P3-C32-P4 163.61(14), N1-Ce1-N2 127.16(6), N3-Ce1-N4 127.36(6), C1-Ce1-C32 176.98(7).
The U1-C1 and U1-C32 distances in 3U (2.410(6) and 2.421(6) Å, respectively) are statistically equivalent to the corresponding distances in 3Ce (ionic radii of CeIV=0.87 Å versus UIV=0.89 Å)62. However, the Th1-C1 and Th1-C32 distances in 3Th (2.514(3) and 2.516(3) Å, respectively) are ∼0.05 Å longer than would be predicted purely based on the increase in ionic radius of ThIV (0.94 Å)62. The short M=C bond lengths in 3Ce, and to some extent 3U, are all the more notable for the fact they are trans, and suggest that an ITI, rather than trans-influence, may be operating.
Reactivity
To give experimental support to the formulation of 3Ce, 3U and 3Th as carbene complexes, we examined their reactivity towards PhCHO. In all cases the Wittig-alkene product PhC(H)=C(PPh2NSiMe3)2 was formed in essentially quantitative yield. Although 3Th reacts too quickly to be monitored (reaction complete in <5 min), and 3U reacts quickly (>80% consumed in 15 min) and is paramagnetic, hence reliable data could not be extracted from questionable NMR integrations, was found to be amenable to a full study (Supplementary Figs 15–17). The reaction of 3Ce with two equivalents of PhCHO was fitted to second-order kinetics overall (first order with respect to both 3Ce and PhCHO) with k (298 K)=1.28 × 10−4±0.255 × 10−4 mol−1 dm3 s−1. Eyring and Arrhenius analyses yielded ΔH‡=+37.2±2 kJ mol–1 and ΔS‡=–194.4 ±6 J mol–1 K–1, affording ΔG‡=+95.2±2 kJ mol–1 (298 K) and Ea=+40.0±2 kJ mol–1. These data are consistent with the reaction conditions and the metallaoxetane intermediates formed from a double [2+2]-cycloaddition/bond metathesis reaction to produce CeO2 and PhC(H)=C(PPh2SiMe3)2. This reactivity is characteristic of covalent early metal carbenes44 and contrasts with more ionic yttrium(III)-methanediide BIPMTMS complexes that activate aryl C–H bonds of carbonyl compounds63. We note that the order of reactivity of 3Ce, 3U and 3Th with PhCHO is 3Th>3U>3Ce; this is principally consistent with the ionic radii of the metals but is also consistent with the increasing level of covalency in the M=C bonds of 3Ce>3U>3Th suggested by our calculations.
Theoretical calculations
To probe the electronic structures of 3Ce, 3U and 3Th, we undertook DFT calculations on their full structures. The geometry-optimized calculations match closely the experimentally determined structures within 0.05 Å and 2° (Supplementary Tables 3–11), and the TD-DFT calculations model the experimentally determined electronic absorption spectrum of 3Ce well. We thus conclude that the theoretical models provide a qualitative description of the electronic manifolds of 3Ce, 3U and 3Th. Although the analysis that follows shows that the dominant feature of the metal–carbon interactions in 3Ce, 3U and 3Th is electrostatic bonding, covalent contributions are present and the discussion focusses on this latter aspect. For 3Ce, 3U and 3Th, the calculated MDCq metal charges are 1.99, 2.77 and 2.48, respectively, and the MDCq carbene charges are −1.79/−1.80, −2.03/−2.06 and −1.95/−2.01, respectively. Although care must be taken when analysing calculated charges, they are indicative of metal(IV) ions44,64 and that perhaps the cerium ion in 3Ce is better matched to receiving electron donation from the ligands than the uranium and thorium ions in 3U and 3Th. However, the calculated MDCm spin density of −2.27 for the uranium ion in 3U is certainly consistent with charge donation from the BIPMTMS ligand to uranium and also with its 5f2 uranium(IV) formulation.
Inspection of the Kohn Sham frontier orbitals of 3Ce, 3U and 3Th (Supplementary Figs 18–20) clearly shows twofold bonding interactions between each carbene and the respective metal. Each M=C bond is polarized, however, as evidenced by M=C Nalewajski–Mrozek bond orders averaging 1.10, 1.30 and 0.73 for 3Ce, 3U and 3Th, respectively. For comparison, the Ce=C bond order in [Ce(BIPMTMS)(ODipp)2] is also 1.1 (ref. 43), whereas uranium(IV)-BIPM complexes typically have U=C double and U–C single bond orders of ∼1.4 and ∼0.5, respectively44,64, and thorium is polarized with Th=C bond orders of ∼0.7 (ref. 64). Therefore, the data presented here fit the overall literature trends.
A clearcut view of the M=C bonding in 3Ce, 3U and 3Th from the Kohn Sham frontier orbitals is precluded because of the delocalized nature of molecular orbital calculations. We therefore turned to natural bond orbital (NBO) analysis (Supplementary Figs 21 and 22) that is suited to the treatment of chemical bonding in molecular complexes. For 3Ce, the Ce=C σ-bonds are composed of ∼13% cerium and 87% carbon character; in each case the cerium contribution is ∼46% 5d and ∼53% 4f, with the remaining ∼1% being 6s. For the Ce=C π-bonds, cerium contributes only ∼8% to these bonds and the cerium component is predominantly 4f character (∼80%) with a modest 5d component (∼19%), with the remainder being of 5p character. These data compare well with those of [Ce(BIPMTMS)(ODipp)2]42, and indeed there is growing evidence that cerium(IV) is suited to utilizing its 4f orbitals in bonding to ligands43,53,65,66. This may be important, because a study on lanthanide(III) chalcogenide complexes found a trans-influence where the lanthanide utilizes predominantly d orbitals12; in contrast the metal(IV) complexes, certainly for cerium and uranium, are deploying predominantly f orbitals, in line with the general theory of the ITI17,18,19,20,21. For 3U, the U=C σ-bonds are composed of ∼14.5% uranium character and ∼85.5% carbon character. As for 3Ce, the 5f:6d contributions of 3U are well balanced at ∼51:47%, with the remainder of 7s character. The U=C, π-bond is ∼14:86% uranium/carbon and, like for cerium, is within the uranium component principally 5f (∼86%) with modest 6d (∼13%), with the remaining ∼1% being 6p character. For 3Th, NBO does not return any Th=C interactions, suggesting that the Th=C bonding is highly ionic.
To better understand the nature of the M=C interactions in these compounds we turned to complete/restricted active space self-consistent field (CASSCF/RASSCF) methodologies that allows us to consider electron correlation through a rigorous configuration-interaction-based approach to directly compare open- and closed-shell compounds. The computational cost of such calculations required truncation and symmetrization of the experimentally determined structures to produce tractable models, but by retaining key structural motifs near identical electronic structures are obtained (Supplementary Tables 12–14).
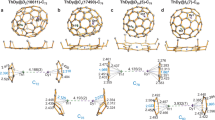
RASSCF calculations were employed in order to identify an appropriate active space for each system. Because of the computational expense of such calculations, RAS1, RAS2 and RAS3 subspaces were constrained to consist of 12, 7 and 12 orbitals, respectively. The 7 RAS2 orbitals comprise the 4f/5f manifold, whereas the RAS1 and RAS3 orbitals account for orbitals with significant C/N 2s and 2p character and natural orbitals whose occupation numbers most deviate from integer values. This active space ensured that all M=C and M–N interactions were accurately modelled. State-averaged RASSCF calculations indicated a degenerate 3B1/3B2 ground state in the 3U model complex, corresponding to a state of E symmetry in the full idealized D2d point group. The natural orbital occupation numbers resulting from these calculations allowed complete active spaces to be defined. Subsequent CASSCF calculations correlated 8 electrons in 8 orbitals in the cases of 3Ce and 3Th model complexes, and 10 electrons in 12 orbitals in the case of the U complex.
CASSCF calculations revealed that all complexes are dominated by M(IV) configurations, (Fig. 5), contributing 96.0%, 96.0% and 95.4% to the ground state wavefunctions of the 3Ce, 3U and 3Th model complexes, respectively. Maximum deviations from integer values in natural orbital occupations were 0.026, 0.021 and 0.026 for the 3Ce, 3U and 3Th model complexes, respectively, indicating at most weak multiconfigurational character. The resultant electronic structure is almost identical to that obtained from the RASSCF simulations, and the dominant M(IV) character in all systems is commensurate with all experimental measures. Notably, as shown in Fig. 5, 3Ce, 3U and 3Th show σ-bonding combinations that are strongly reminiscent of uranyl, a feature that also emerges from the DFT analysis that is consistent with an ITI in these complexes.
Only strongly occupied M–C bonding orbitals and 5f orbitals are shown. Pronounced multiconfigurational character is present among the 5f orbitals in the truncated U complex, but the rest of the electronic manifold comprises orbitals with occupations close to integer values showing at best weak multiconfigurational character. All orbitals rendered at an isosurface of 0.02 atomic units (a.u.).
To further probe the covalent contribution to bonding in these compounds, we directly analyse the resultant electron densities via the quantum theory of atoms in molecules (QTAIM) and focus on two parameters: the delocalization index between two bonded atomic centres (δ) and the magnitude of the electron density at the bond critical point between the centres (ρBCP). The delocalization index, formally a two-electron property of the system, is a measure of the number of electrons shared between two atoms, and is large when orbital mixing because of energetic near degeneracy between the two atoms is pronounced. In this sense, it probes similar properties to those given by orbital decomposition via NBO analysis or, experimentally, by X-ray absorption spectroscopy that probes orbital energy near degeneracy. On the other hand, ρBCP quantifies electronic charge concentration in the bond between two atoms. Combined, these measures give an indication as to whether orbital mixing leads to charge accumulation in the bonding region (and hence bond stabilization) and hence provide a more complete method for assessing bond covalency than orbital analysis alone (Supplementary Table 15)53.
QTAIM-derived atomic charges are more consistent than those obtained via NBO analysis, with q(Ce)<q(U)<q(Th). Carbene charges reflect this trend that therefore provides some evidence for a greater covalent interaction in the 3Ce complex. Stronger evidence is provided by the delocalization indices, δ(M,C), that are notably larger for 3Ce than either 3U or 3Th. Consideration of ρBCP demonstrates that this electron sharing corresponds to charge accumulation in the M=C bonding region: ρBCP follows a similar trend to δ(M,C), with the Ce and U complexes exhibiting significantly larger values than that of Th. Inspection of the ellipticity parameter ɛ for the M=C bonds reveals values that are consistent with an asymmetric distribution of electron density around the M=C bond comparable to those found for alkenes and [M(BIPMTMS)(ODipp)2]43, confirming that in 3Ce, 3U and 3Th there is a M=C bonding interaction involving two electron pairs donated from a carbene to a metal.
Probing the inverse-trans-influence
To probe whether the ITI is operating in 3Ce, 3U and 3Th, we adopted the method of O’Grady and Kaltsoyannis29. Here, 3Ce, 3U and 3Th were geometry optimized with a frozen core, up to 4d for 3Ce and 5d for 3U and 3Th and with the pseudo-core 5 or 6p orbitals, respectively, either explicitly included as valence orbitals or placed in the frozen core. Although the ITI is a complex phenomenon that involves several factors, it is clear that p orbitals are involved in the ITI and that this method isolates the contributions that the pseudo-core 5/6p orbitals have on the bonding. Although the carbene ligands in 3Ce, 3U and 3Th clearly exhibit polarized M=C bonds, the ITI is predominantly dependent on the charge and polarizing nature of the coordinated ligands33. To rule out coincidental systematic errors from a particular method, we examined the effect of varying the functional (BP86 versus PBE) and the basis set (normal all-electron basis set versus a normal frozen core up to 4 or 5d for 3Ce, 3U, and 3Th, respectively) separately or simultaneously, and found no significant changes in equilibrium geometries. However, when the 5 or 6p orbitals for 3Ce, 3U and 3Th, respectively, were additionally also placed in the frozen core, significant changes to the equilibrium geometries were observed in all cases. Specifically, the M=C distances lengthen by ∼0.05 Å when the relevant p orbitals are placed in the frozen core that are very similar shifts to those found previously for [MOX5]− anions (M=U, Np; X=F, Cl, Br)29, and this represents the p orbital contributions to the ITI. It should be noted that, on inclusion of the relevant p orbitals into the frozen cores, the M–Nimino distances also elongate; however, the latter lengthen by only ∼0.02 Å, less than half that of the change to the M=C linkages. As expected, with p orbitals in the frozen cores the metal and carbene charges increase, indicating more polarized and presumably weakened interactions. For example, in 3Ce the cerium and carbene charges rise from 1.83 and −1.71 when 5p orbitals are included in the valence region to 2.54 and −1.82 when 5p orbitals are placed in the frozen core. Inspection of the differences of the total energies of the geometry-optimized structures of 3Ce, 3U and 3Th with the p orbitals in the frozen core or in the valence region yields energy differences of 12.5, 20.5 and 18.2 kcal mol−1 for 3Ce, 3U and 3Th, respectively. This provides a qualitative bracketing of the stabilizing energy that the inclusion of the pseudo-core p orbitals in the bonding to a bis(carbene) ligand set provides, and compares well with the ITI of 6 kcal mol−1 calculated for a uranium(VI)-mono(oxo) unit in a tris(aryloxide) triazacyclononane complex30.
As 3Ce, 3U and 3Th all appear to exhibit the ITI, we investigated the synthesis of the analogous praseodymium(IV) and terbium(IV) bis(BIPM) complexes 3Pr and 3Tb, respectively. We targeted these complexes because after cerium they have the next two lowest fourth ionization energies of all lanthanides34,35. Nevertheless, the fourth ionization energies of these two elements are still considerable, and we could not access 3Pr and 3Tb experimentally. Attempting AgBPh4-mediated oxidations of 2Pr and 2Tb (Fig. 1) results not in oxidation to give 3Pr and 3Tb but instead elimination of [K(18C6)(THF)2][BPh4] and isolation of [M(BIPMTMS)2Ag] (M=Pr, 6Pr; M=Tb, 6Tb); photolysis or electrochemistry experiments on 6Pr and 6Tb resulted in intractable decomposition products. However, although we could not prepare 3Pr and 3Tb, 3Ce, 3U, 3Th, 2Pr and 2Tb provide experimentally calibrated benchmarks with which to provide confidence in the calculated hypothetical geometry-optimized structures of 3Pr and 3Tb. Inspection of the equilibrium geometries of 3Pr and 3Tb calculated with their respective 5p orbitals in-core and included in the valence regions reveals that the ITI persists but diminishes on moving from Ce to Pr to Tb (Supplementary Tables 16–19). Specifically, the Pr=C distances elongate by 0.02 Å when the 5p orbitals are placed in the frozen core, and the Pr-N distances elongate by only 0.006 Å. For Tb, the effect is significantly reduced, with a 0.007 Å elongation of the Tb=C distance when the 5p orbitals are placed in the frozen core and the Tb-N distances elongate by 0.006 Å. We conclude from these data that the ITI may, in principle, apply across the lanthanide(IV) series, but as 4f electron occupancy increases the ITI diminishes. This is consistent with the theory of the ITI as donation of (n)p electron density into the (n−1)f orbital manifold will become less favourable as the (n−1)f occupancy increases because of interelectronic repulsion19. Furthermore, the appearance of a trend suggests greater levels of f-orbital covalency that is supported by the characterization data more widely, whereas for lanthanide(III) systems when the trans-influence has been studied and principally d-orbital participation has been invoked then the trans-influence trend is uniform, suggestive of mainly ionic bonding character12. Thus, the observations of uniform trans-influence with d-orbital bonding for lanthanide(III) ions12 versus diminishing ITI for lanthanide(IV) ions where f-orbital bonding is principally invoked is internally consistent.
The delocalized nature of the molecular orbital approach makes the identification of key molecular orbitals involved in the ITI difficult29,30. Thus, inspection of individual molecular orbitals would not be expected to provide clear-cut information, as has proven to be the case even in highly symmetric complexes29. However, an examination of the electronic manifolds of 3Ce, 3U and 3Th reveals a common molecular orbital that may be significant. All three complexes exhibit a molecular orbital at ∼−16.4 eV (3Ce, −16.483; 3U, −16.485; 3Th, −16.494 eV) ∼12 eV below the highest occupied molecular orbital (−2 for 5f2 3U). In each case contributions from the 2s orbitals of each carbene (normalized to 34%) and 5 or 6p orbitals for cerium or uranium and thorium, respectively (normalized to 5%) are found. Interestingly, a similar orbital is computed for 3Pr at −16.476 eV, although here the carbon (normalized to 32%) and praseodymium (normalized to 2.5%) contributions are notably less well matched. In contrast, for 3Tb the closest match is now a molecular orbital at −20.455 eV with normalized carbon and terbium contributions of 9% and 3%, respectively. We suggest that this molecular orbital that is common to 3Ce, 3U, 3Th and 3Pr but not 3Tb may represent a signature in this instance of the ITI19,20,29.
Discussion
We have prepared three new metal bis(carbene) complexes that contain linear C=M=C cores and the characterization data show these complexes to be unequivocally metal(IV) complexes and thus valid to compare with one another. For the cerium and uranium derivatives the M=C bonds are short, and for the former one of the shortest Ce-C bonds on record; that they are so short despite the fact that they are strongly donating dianions disposed trans with respect to one another suggests that the ITI is operating instead of the more classical trans influence. Theoretical calculations suggest the presence of an ITI in cerium, uranium and thorium derivatives, as removal of pseudo-core (n)p orbitals from the valence region in calculations consistently results in elongation of the M=C bonds by over twice that of the elongation of the M-imino bonds. Interestingly, the characterization data and theoretical data taken together suggest a consistent trend of covalency of 3Ce∼3U>3Th. This work suggests that the ITI concept, first established a quarter of a century ago, may extend beyond high oxidation state 5f metals to now encompass mid-range oxidation state 5f actinides and 4f lanthanides. Calculations also suggest, however, that the ITI may diminish on moving from left to right in the lanthanide series and with increasing (n−1)f occupation number. Although an opposite trend may operate for the actinides29, it may be that the more diffuse 5f orbitals could tolerate occupancy more; however, thus far the radioactivity of those elements has precluded any detailed body of experimental work from being compiled and hence further work will be required to provide the necessary benchmarks with which to investigate this. The observations of uniform trans-influence with d-orbital bonding for lanthanide(III) ions12 versus the trend reported here of diminishing ITI for the lanthanide(IV) ions investigated, where f-orbital bonding is principally invoked, is gratifyingly internally consistent. Thus, the ITI might be a more general f-block principle.
Methods
General
Experiments were carried out under a dry, oxygen-free dinitrogen atmosphere using Schlenk-line and glove-box techniques. All solvents and reagents were rigorously dried and deoxygenated before use. Compounds were variously characterized by elemental analyses, electrochemistry, NMR, Fourier transform infrared spectroscopy (FTIR), EPR, XANES and UV/Vis/NIR electronic absorption spectroscopies, single crystal X-ray diffraction studies (Supplementary Data 1–11), Evans methods and SQUID magnetometry, and DFT, NBO, QTAIM, CASSCF and RASSCF computational methods.
Synthesis of [Ce(BIPMTMS)2][K(18C6)(THF)2] (2Ce)
THF (15 ml) was added to a precooled (−78 °C) mixture of 1Ce (1.41 g, 1.17 mmol) and [K(CH2Ph)] (0.15 g, 1.17 mmol). The resulting orange suspension was allowed to slowly warm to room temperature with stirring over 16 h to afford an orange solution. 18C6 (0.31 g, 1.17 mmol) in THF was added and stirred for a further 2 h. The solvent was removed in vacuo to afford an orange solid. The solid was washed with toluene to afford 2Ce as a yellow powder. Yield: 2.31 g, 52%. Recrystallization of a small portion from toluene afforded yellow crystals of 2Ce on storing at room temperature. Anal. Calcd for C82H116CeKN4O8P4Si4: C, 57.91; H, 6.87; N, 3.29%. Found: C, 57.65; H, 6.89; N, 3.09%. 31P{1H} NMR (C6D6, 298 K): δ 7.22 (CeCP2). FTIR v/cm−1 (Nujol): 1,350 (w) 1,302 (w), 1,077 (s), 961 (m), 846 (m), 771 (m), 743 (s), 695 (m), 633 (m), 522 (s). Magnetic moment (Evans method, THF, 298 K): μeff=2.18 μB.
Synthesis of [Ce(BIPMTMS)2] (3Ce)
Toluene (15 ml) was added to a precooled (−78 °C) mixture of 2Ce (1.64 g, 0.96 mmol) and [Ag(BPh4)] (0.41 g, 0.96 mmol). The resulting yellow suspension was allowed to warm to room temperature with stirring over 16 h to afford a green suspension. The suspension was filtered and volatiles were removed in vacuo to afford a green solid. Recrystallization from toluene (2 ml) afforded 3Ce as green crystals. Yield: 0.52 g, 43%. Recrystallization of a small portion from pentane (3 ml) afforded green crystals suitable for single crystal X-ray diffraction analysis. Anal. Calcd for C62H76CeN4P4Si4: C, 59.42; H, 6.11; N, 4.47%. Found: C, 59.63; H, 6.15; N, 4.43%. 1H NMR (C6D6, 298 K): δ 0.95 (36H, s, NSi(CH3)3), 7.08 (24H, d, p/o-Ar-H), 7.24 (16H, m, o-Ar-H) p.p.m. 13C{1H} NMR (C6D6, 298 K): δ 6.41 (NSi(CH3)3), 127.26 (m-Ar-C), 129.78 (o-Ar-C), 132.52 (p-Ar-C), 140.50 (i-Ar-C), 343.48 (t, JPC=170.22 Hz, CeCP2) p.p.m. 31P{1H} NMR (C6D6, 298 K): δ −13.65 (CeCP2) p.p.m. 29Si{1H} NMR (C6D6, 298 K): δ −2.17 (NSi(CH3)3) p.p.m. FTIR v/cm−1 (Nujol): 1,294 (w), 1,246 (w), 1,064 (s), 843 (m), 757 (w), 693 (w), 656 (m), 597 (w), 509 (m).
Preparation of [U(BIPMTMS)(CH2SiMe3)2] (5U)
THF (10 ml) was added to a precooled (−78 °C) mixture of [U(BIPMTMS)(Cl)3(Li)(THF)2] (1.09 g, 1 mmol) and LiCH2SiMe3 (0.19 g, 2 mmol). The mixture was then allowed to slowly warm to room temperature with stirring over 6 h to afford a brown solution. Volatiles were removed in vacuo and the resulting brown solid was extracted with toluene (20 ml). Volatiles were removed in vacuo and the resulting brown solid was recrystallized from hexane (3 ml) and stored at −30 °C to afford 5U as brown crystals. Yield 0.67 g (69%). Anal Calcd for C38H60N2P2Si4U: C, 47.22; H, 6.00; N, 2.98. Found: C, 46.53; H, 5.92; N, 2.97. The carbon is consistently low over five independently synthesized batches that we attribute to carbide formation. 1H NMR (C6D6, 298 K): δ −7.76 (s, 18H, Si(CH3)3), 0.41 (s, 18H, Si(CH3)3), 5.21 (m, 4H, Ar-H), 5.49 (br, 8H, Ar-H), 5.85 (br, 8H, Ar-H), 7.15 (br, 4H). IR v/cm−1 (Nujol): 1,403 (w), 1,305 (w), 1,248 (m, br), 1,108 (m), 1,019 (m, br), 836 (s, br). (Evans method, C6D6, 298 K): 2.45 μB.
Preparation of [U(BIPMTMS)2] (3U)
Toluene (15 ml) was added to a mixture of [U(BIPMTMS)(CH2SiMe3)2] (0.97 g, 1.00 mmol) and BIPMTMSH2 (0.56 g, 1.00 mmol) at room temperature. The resulting brown solution was stirred at 90 °C for 18 h, and then was allowed to cool to ambient temperature and filtered. All volatiles were removed from the filtrate to afford a brownish black solid that was washed by 5 ml of pentane at 0 °C to afford the product as a brown solid. Yield 0.81 g, 60%. Recrystallization of a small portion from a toluene/hexane mixture at 5 °C afforded brown crystals suitable for a single crystal X-ray diffraction study. Anal. Calcd for C62H76N4P4Si4U·C6H14: C 56.81; H 6.16; N 3.90. Found: C 56.44; H 6.16; N 3.89. 1H NMR (C6D6, 298 K): δ −33.88 (br, 36 H, NSi(CH3)3), 12.04 (s, 9 H, Ar-H), 13.76 (s, 17 H, Ar-H), 30.42 (br, 14 H, Ar-H). 31P{1H} NMR (C6D6, 298 K): δ −219.70 (br, UCP2) p.p.m. FTIR ν/cm−1 (Nujol): 1,958 (w), 1,450 (s), 1,334 (s), 1,281 (m), 1,245 (m), 1,178 (w), 1,155 (w), 1,103 (s), 1,035 (m), 745 (m), 669 (m), 628 (m), 514 (w), 489 (w). Magnetic moment (Evans method, C6H6 298 K): μeff=2.61 μB.
Preparation of [Th(BIPMTMS)(CH2SiMe3)2] (5Th)
Li2BIPM (1.71 g, 3 mmol) in THF (10 ml) was added to a precooled (−78 °C) suspension of ThCl4(THF)3.5 (1.88 g, 3 mmol) in THF (10 ml). The pale yellow reaction mixture was stirred at −78 °C for 30 min and at room temperature for 2 h. After which the mixture was cooled to −78 °C again, and LiCH2SiMe3 (0.56 g, 6 mmol) in THF (10 ml) was added. The resulted pale yellow solution was kept at −35 °C for 12 h and then all volatiles were removed in vacuo to afford a yellow oil that was extracted by i-hexane (3 × 10 ml). All volatiles were removed in vacuo from the filtrate to afford the product as a pale yellow solid. Yield: 2.17 g, 75%. Recrystallization of a small portion from i-hexane (3 ml) at −30 °C afforded colourless crystals suitable for single crystal X-ray diffraction analysis. Anal. Calcd for C39H60N2P2Si4Th: C, 48.63; H, 6.28; N, 2.91%. Found: C, 48.70; H, 6.39; N, 2.65%. 1H NMR (C6D6, 298 K): δ −0.01 (s, 4 H, ThCH2), 0.22 (s, 18 H, CH2SiMe3 or NSiMe3), 0.43 (s, 18 H, CH2SiMe3 or NSiMe3), 6.98 (br, 12 H, p- and o-Ar-H), 7.59 (br, 8 H, m-Ar-H) p.p.m. 13C{1H} NMR (C6D6, 298 K) δ 2.74 (s, CH2SiMe3 or NSiMe3), 4.54 (s, CH2SiMe3 or NSiMe3), 73.75 (t, 1JPC=159 Hz, ThCP2), 92.59 (s, ThCH2), 130.18 (s, o- and p-ArC), 131.13 (t, 3JPC=5.6 Hz, m-ArC), 137.79 (t, 1JPC=50.3 Hz, ipso-ArC) p.p.m. 31P NMR (C6D6, 298 K): δ 5.81 (s) p.p.m. 29Si{1H} NMR (C6D6, 298 K): δ −6.85 (t, 2JPSi=3.10 Hz, NSiMe3), −1.17 (s, ThCH2SiMe3) p.p.m. FTIR v/cm−1 (Nujol): 1,591 (w), 1,403 (s), 1,302 (m), 861 (s), 695 (w), 608 (w), 588 (w), 551 (w).
Preparation of [Th(BIPMTMS)2] (3Th)
Toluene (10 ml) was added to a mixture of [Th(BIPMTMS)(CH2SiMe3)2] (0.87 g, 0.90 mmol) and BIPMTMSH2 (0.51 g, 0.90 mmol). The resulting pale yellow solution was stirred at 50 °C for 20 h, and then was dried in vacuo to afford sticky yellow solid. The crude product was washed with hexanes (2 × 10 ml) and then dried in vacuo to afford 3Th as a colourless powder. Yield: 0.63 g, 52%. Recrystallization of a small portion from toluene at 5 °C afforded colourless crystal suitable for single crystal X-ray diffraction. Anal. Calcd for C62H76N4P4Si4Th: C 55.34; H 5.69; N 4.16%. Found: C 55.02; H 5.78; N 3.91%. 1H NMR (C6D6, 298 K): δ 0.30 (s, 36 H, NSi(CH3)3), 7.03–7.12 (m, 25 H, Ar-H), 7.59–7.66 (m, 15 H, Ar-H). 13C{1H} NMR (C6D6, 298 K): δ 5.25 (s, NSi(CH3)3), 127.37 (s, Ar-C), 129.75 (s, Ar-C), 132.31 (s, Ar-C), 141.27 (t, 3JPC=47.0 Hz, i-Ar-C). The carbene centre in 3Th was not observed in the 13C{1H} NMR spectrum. 31P{1H} NMR (C6D6, 298 K): δ 6.25 (ThCP2). 29Si{1H} NMR (C6D6, 298 K): δ −6.83 (NSi(CH3)3). FTIR ν/cm−1 (Nujol): 1,305 (m), 1,244 (w), 1,154 (m), 1,051 (s), 804 (s), 741 (w), 722 (s), 696 (w), 634 (s), 492 (m).
Preparation of [Tb(BIPMTMS)(BIPMTMSH)] (1Tb)
BIPMTMSH2 (4.47 g, 8 mmol) in toluene (10 ml) was added dropwise to a precooled (−78 °C) suspension of [Tb(CH2Ph)3(THF)3] (2.32 g, 4 mmol) in toluene (15 ml). The resulting orange suspension was warmed to room temperature with stirring over 16 h and then refluxed for 10 min to afford a yellow solution. Volatiles were removed in vacuo and the resulting yellow residue recrystallized from hot toluene (4 ml) to afford colourless crystals of 1Tb on cooling to room temperature. Yield: 1.81 g, 36%. Anal. Calcd for C62H77N4P4Si4Tb: C, 58.50; H, 6.10; N, 4.40%. Found: C, 58.61; H, 6.06; N, 4.33%. FTIR v/cm−1 (Nujol): 1,367 (s), 1,242 (m), 1,219 (m), 1,105 (s), 1,031 (m), 841 (s), 696 (m), 638 (w), 610 (w), 553 (m), 522 (m). Magnetic moment (Evans method, C6D6, 298 K): μeff=9.90 μB.
Preparation of [Pr(BIPMTMS)2][K(18C6)(THF)2] (2Pr)
THF (15 ml) was added to a precooled (−78 °C) mixture of 1Pr (5.25 g, 4.18 mmol) and [K(CH2Ph)] (0.55 g, 4.18 mmol). The resulting orange suspension was allowed to slowly warm to room temperature with stirring over 16 h to afford an orange solution. 18C6 (1.11 g, 4.18 mmol) in THF was added and stirred for a further 2 h. The solvent was removed in vacuo to afford an orange solid. The solid was washed with toluene to afford 2Pr as a yellow powder. Yield: 4.50 g, 63%. Recrystallization of a small portion from toluene afforded yellow crystals of 2Pr on storing at room temperature. Anal. Calcd for C82H116KN4O8P4PrSi4: C 57.89; H, 6.87; N, 3.29%. Found: C, 56.72; H, 6.67; N, 3.29%. FTIR v/cm−1 (Nujol): 1,352 (w), 1,302 (w), 1,105 (m), 964 (w), 826 (w), 695 (w), 634 (w), 522 (m). Magnetic moment (Evans method, THF, 298 K): μeff=3.56 μB.
Preparation of [Tb(BIPMTMS)2][K(18C6)(THF)2] (2Tb)
THF (15 ml) was added to a precooled (−78 °C) mixture of 1Tb (0.94 g, 0.74 mmol) and [K(CH2Ph)] (0.096 g, 0.74 mmol). The resulting orange suspension was allowed to slowly warm to room temperature with stirring over 16 h to afford a yellow solution. 18C6 (0.31 g, 1.17 mmol) in THF was then added and the resulting yellow solution stirred for 2 h. The solution was then reduced in volume to ca. 2 ml that afforded colourless crystals of 2Tb on standing at room temperature. Yield: 0.26 g, 38%. Anal. Calcd for C82H116KN4O8P4Si4Tb: C, 57.27; H, 6.80; N, 3.26%. Found: C, 56.67; H, 6.63; N, 3.34%. FTIR v/cm−1 (Nujol): 1,351 (w), 1,303 (w), 1,071 (s), 960 (m), 848 (m), 760 (m), 743 (s), 700 (m), 634 (m), 523 (s). Magnetic moment (Evans method, THF, 298 K): μeff=10.55 μB.
Preparation of [Pr(BIPMTMS)2Ag] (6Pr)
Toluene (15 ml) was added to a precooled (−78 °C) mixture of 2Pr (1.70 g, 1.00 mmol) and [Ag(BPh4)] (0.43 g, 1.00 mmol). The resulting yellow suspension was allowed to slowly warm to room temperature with stirring over 16 h. The suspension was filtered and volatiles removed in vacuo and the resulting solid washed with hexanes (10 ml) to afford 6Pr as a colourless powder. Yield 1.18 g, 87%. Recrystallization of a small portion from toluene afforded colourless crystals of 6Pr. Anal. Calcd for C62H76AgN4P4PrSi4·1.25(C7H8): C 57.54; H, 5.87; N, 3.79%. Found: C, 57.44; H, 5.84; N, 3.69%. 31P{1H} NMR (C6D6, 298 K): δ −98.83 (1P, PrCP2), −83.25 (1P, PrCP2), 33.99 (2P, PrCP2) p.p.m. FTIR v/cm−1 (Nujol): 1,244 (w), 1,080 (s), 1,026 (s), 831 (m), 768 (m), 732 (m), 657 (m), 606 (w), 575 (w), 526 (w). Magnetic moment (Evans method, d8-THF, 298 K): μeff=2.93 μB.
Preparation of [Tb(BIPMTMS)2Ag] (6Tb)
Toluene (15 ml) was added to a precooled (−78 °C) mixture of 2Tb (1.72 g, 1.00 mmol) and [Ag(BPh4)] (0.43 g, 1.00 mmol). The resulting yellow suspension was allowed to slowly warm to room temperature with stirring over 16 h. The brown suspension was filtered and volatiles reduced in volume to 2 ml to afford colourless crystals of 6Tb upon storage at room temperature. Yield 0.42 g, 31%. Anal. Calcd for C62H76AgN4P4Si4Tb·0.8(C7H8): C, 55.84; H, 5.71; N, 3.85%. Found: C, 55.85; H, 5.81; N, 3.83%. FTIR v/cm−1 (Nujol): 1,435 (m), 1,243 (w), 1,167 (m), 1,106 (s), 1,060 (s), 833 (s), 769 (m), 715 (m), 693 (m), 611 (w), 527 (w). Magnetic moment (Evans method, d8-THF, 298 K): μeff=9.12 μB.
Reaction of [Ce(BIPMTMS)2] with PhCHO
Benzaldehyde (2.5 mg, 24 μmol) was added to a solution of [Ce(BIPMTMS)2] (15 mg, 12 μmol) in d6-benzene (0.4 ml). The reaction mixture was shaken vigorously, forming a green reaction mixture. The reaction mixture was stored at room temperature for 16 h and then analysed by multinuclear NMR that revealed a small amount of conversion to (Me3SiNPPh2)2C=C(H)Ph. The reaction mixture was then heated to 60 °C for 48 h. Analysis of the crude mixture showed quantitative conversion to (Me3SiNPPh2)2C=C(H)Ph. All spectroscopic data matched previously reported data67.
Reaction of [Ce(BIPMTMS)2] with ArCHO
d6-benzene (0.4 ml) was added to a mixture of 9-anthracene carboxaldehyde (4.9 mg, 24 μmol) and [Ce(BIPMTMS)2] (15 mg, 12 μmol). The reaction mixture was shaken vigorously, forming a green reaction mixture. The reaction mixture was stored at room temperature for 16 h, heated to 60 °C for 48 h and heated to 80 °C for 48 h. Analysis of the crude mixture showed no conversion to (Me3SiNPPh2)2C=C(H)Ar.
Reaction of [U(BIPMTMS)2] with PhCHO
Benzaldehyde (6.4 mg, 60 μmol) was added to a solution of [U(BIPMTMS)2] (40.5 mg, 30 μmol) in d6-benzene (0.4 ml). The reaction mixture was shaken vigorously, forming a brown reaction mixture. The reaction mixture was stored at room temperature for 48 h and then analysed by multinuclear NMR that revealed 95% conversion to (Me3SiNPPh2)2C=C(H)Ph. All spectroscopic data matched previously reported data67.
Reaction of [U(BIPMTMS)2] with ArCHO
d6-benzene (0.4 ml) was added to a mixture of 9-anthracene carboxaldehyde (12.4 mg, 60 μmol) and [U(BIPMTMS)2] (40.5 mg, 30 μmol). The reaction mixture was shaken vigorously, forming a brown reaction mixture. The reaction mixture was stored at room temperature for 96 h and then analysed by multinuclear NMR that revealed 90% conversion to (Me3SiNPPh2)2C=C(H)Ar. All spectroscopic data matched previously reported data40.
Reaction of [Th(BIPMTMS)2] with PhCHO
Benzaldehyde (6.4 mg, 60 μmol) was added to a solution of [Th(BIPMTMS)2] (40.1 mg, 30 μmol) in d6-benzene (0.4 ml). The reaction mixture was shaken vigorously, forming a colourless reaction mixture. The reaction mixture was stored at room temperature for 36 h and then analysed by multinuclear NMR that revealed 95% conversion to (Me3SiNPPh2)2C=C(H)Ph. All spectroscopic data matched previously reported data67.
Reaction of [Th(BIPMTMS)2] with ArCHO
d6-benzene (0.4 ml) was added to a mixture of 9-anthracene carboxaldehyde (12.4 mg, 60 μmol) and [Th(BIPMTMS)2] (40.1 mg, 30 μmol). The reaction mixture was shaken vigorously, forming a yellow reaction mixture. The reaction mixture was stored at room temperature for 48 h and then analysed by multinuclear NMR that revealed 95% conversion to (Me3SiNPPh2)2C=C(H)Ar. All spectroscopic data matched previously reported data40.
Data availability
The X-ray crystallographic coordinates (cif format) for structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers 1500929-1500939. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. All other data are available from the corresponding authors on request.
Additional information
How to cite this article: Gregson, M. et al. The inverse-trans-influence in tetravalent lanthanide and actinide bis(carbene) complexes. Nat. Commun. 8, 14137 doi: 10.1038/ncomms14137 (2017).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
05 February 2018
The original version of this Article contained a typographical error in Fig. 2, where reagent H2C(Ph2PNSiMe3)2 was incorrectly given as H2C(PNSiMe3)2. This has now been corrected in both the PDF and HTML versions of the Article.
References
Basolo, F. & Pearson, R. G. The trans effect in metal complexes. Prog. Inorg. Chem. 4, 381–453 (1962).
Pidcock, A., Richards, R. E. & Venanzi, L. M. 195Pt-31P nuclear spin coupling constants and nature of the trans-effect in platinum complexes. J. Chem. Soc. A 1707–1710 (1966).
Coe, B. J. & Glenwright, S. J. Trans-effects in octahedral transition metal complexes. Coord. Chem. Rev. 203, 5–80 (2000).
Wilson, J. J. & Lippard, S. J. Synthetic methods for the preparation of platinum anticancer complexes. Chem. Rev. 114, 4470–4495 (2014).
Deacon, G. B., Gatehouse, B. M., Shen, Q. & Ward, G. N. Organoamido- and aryloxo-lanthanides-VII. The X-ray structure of five-coordinate [La(OC6H3Ph2-2,6)3(THF)2]·THF. Polyhedron 12, 1289–1294 (1993).
Cosgriff, J. E., Deacon, G. B. & Gatehouse, B. M. Organoamido- and aryloxo-lanthanoids. IX Preparations and structures of tris(η2-3,5-diphenylpyrazolato)lanthanoid(III) complexes with triphenylphosphine oxide and tetrahydrofuran. Aust. J. Chem. 46, 1881–1896 (1993).
Deacon, G. B., Feng, T., Skelton, B. W. & White, A. H. Organoamido- and aryloxo-lanthanoids. XI Synthesis and crystal structures of Nd(Odpp)3, Nd(Odpp)3(thf) and [Nd(Odpp)3(thf)2.2(thf) (Odpp−=2,6-Diphenylphenolate): variations in intramolecular π-Ph-Nd interactions. Aust. J. Chem. 48, 741–756 (1995).
Freedman, D., Melman, J. H., Emge, T. J. & Brennan, J. G. Cubane clusters containing lanthanide ions: (py)8Yb4Se4(SePh)4 and (py)10Yb6S6(SPh)6 . Inorg. Chem. 37, 4162–4163 (1998).
Rabe, G. W., Strissel, C. S., Liable-Sands, L. M., Concolino, T. E. & Rheingold, A. L. Terphenyl ligand systems in lanthanide chemistry: synthesis and structural characterization of two 2,6-dimesitylphenyl derivatives of trivalent ytterbium. Inorg. Chem. 38, 3446–3447 (1999).
Deacon, G. B. et al. Manipulation of reaction pathways in redox transmetallation-ligand exchange synthesis of lanthanoid(II/III) aryloxide complexes. Dalton Trans. 2006, 802–812 (2006).
Panda, T. K. et al. Imidazolin-2-iminato complexes of rare earth metals with very short metal−nitrogen bonds: experimental and theoretical studies. Inorg. Chem. 48, 5462–5472 (2009).
Krogh-Jespersen, K., Romanelli, M. D., Melman, J. H., Emge, T. J. & Brennan, J. G. Covalent bonding and the trans influence in lanthanide compounds. Inorg. Chem. 49, 552–560 (2010).
Gardner, B. M., Lewis, W., Blake, A. J. & Liddle, S. T. Halide, amide, cationic, manganese carbonylate and oxide derivatives of triamidosilylamine uranium complexes. Inorg. Chem. 50, 9631–9641 (2011).
Hamidi, S., Deacon, G. B., Junk, P. C. & Neumann, P. Direct reaction of iodine-activated lanthanoid metals with 2,6-diisopropylphenol. Dalton Trans. 41, 3541–3552 (2012).
Gholivand, K. & Mahzouni, H. R. Trans influence and covalent bonding in a new octahedral lanthanum(III) complex of diphenylmorpholinyl phosphinamide. Inorg. Chim. Acta 386, 8–12 (2012).
Deacon, G. B., Hamidi, S., Junk, P. C., Kelly, R. P. & Wang, J. Direct reactions of iodine-activated rare-earth metals with phenols of varying steric bulk. Eur. J. Inorg. Chem. 2014, 460–468 (2014).
Walch, P. F. & Ellis, D. E. Effects of secondary ligands on the electronic structure of uranyls. J. Chem. Phys. 65, 2387–2392 (1976).
Tatsumi, K. & Hoffmann, R. Bent Cis d0 MoO22+ vs. linear trans d0f0 UO22+: a significant role for nonvalence 6p orbitals in uranyl. Inorg. Chem. 19, 2656–2658 (1980).
Denning, R. G. Electronic structure and bonding in actinyl ions. Struct. Bonding (Berlin) 79, 215–276 (1992).
Denning, R. G. Electronic structure and bonding in actinyl ions and their analogs. J. Phys. Chem. A 111, 4125–4143 (2007).
La Pierre, H. S. & Meyer, K. Uranium–ligand multiple bonding in uranyl analogues, [L=U=L]n+, and the inverse trans influence. Inorg. Chem. 52, 529–539 (2013).
Butcher, R. J., Penfold, B. R. & Sinn, E. Crystal structures of cis-dibromodioxobis(triphenylphosphine oxide)molybdenum(VI), cis-dichlorodioxobis(triphenylphosphine oxide)molybdenum(VI), and cis-bis(butane-2,3-diolato)dioxomolybdenum(VI)-butane-2,3-diol (1/2): a comparison of co-ordination spheres and the general stereochemistry of molybdenum(VI) oxo-complexes. J. Chem. Soc. Dalton Trans. 1979, 668–675 (1979).
Bombieri, G., Forsellini, E., Day, J. P. & Azeez, W. I. Crystal and molecular structure of dichlorodioxobis(triphenylphosphine oxide)uranium(VI). J. Chem. Soc. Dalton Trans. 1978, 677–680 (1978).
King, D. M. et al. Isolation and characterisation of a uranium(VI)-nitride triple bond. Nat. Chem. 5, 482–488 (2013).
King, D. M. et al. Single-molecule magnetism in a single-ion triamidoamine uranium(V) terminal mono-oxo complex. Angew. Chem. Int. Ed. 52, 4921–4924 (2013).
Gardner, B. M. & Liddle, S. T. Uranium triamidoamine chemistry. Chem. Commun. 51, 10589–10607 (2015).
Gardner, B. M. et al. Homologation and functionalization of carbon monoxide by a recyclable uranium complex. Proc. Natl Acad. Sci. USA 109, 9265–9270 (2012).
Eikey, R. A. & Abu-Omar, M. M. Nitrido and imido transition metal complexes of groups 6-8. Coord. Chem. Rev. 243, 83–124 (2003).
O’Grady, E. & Kaltosyannis, N. On the inverse trans influence. Density functional studies of [MOX5]n−_ (M=Pa, n=2; M=U, n=1; M=Np, n=0; X=F, Cl or Br). J. Chem. Soc. Dalton Trans. 2002, 1233–1239 (2002).
Kosog, B., La Pierre, H. S., Heinemann, F. W., Liddle, S. T. & Meyer, K. Synthesis of uranium(VI) terminal oxo complexes: molecular geometry driven by the inverse trans-influence. J. Am. Chem. Soc. 134, 5284–5289 (2012).
Lewis, A. J., Carroll, P. J. & Schelter, E. J. Stable uranium(VI) methyl and acetylide complexes and the elucidation of an inverse trans influence ligand series. J. Am. Chem. Soc. 135, 13185–13192 (2013).
Lewis, A. J., Mullane, K. C., Nakamaru-Ogiso, E., Carroll, P. J. & Schelter, E. J. The inverse trans influence in a family of pentavalent uranium complexes. Inorg. Chem. 53, 6944–6953 (2014).
La Pierre, H. S. et al. Charge control of the inverse trans-influence. Chem. Commun. 51, 16671–16674 (2015).
Sroor, F. M. A. & Edelmann, F. T. in The Rare Earth Elements: Fundamentals and Applications (ed. Atwood D. A. 313John Wiley & Sons Ltd (2012).
Sroor, F. M. A. & Edelmann, F. T. in The Rare Earth Elements: Fundamentals and Applications (ed. Atwood D. A. 321John Wiley & Sons Ltd (2012).
MacDonald, M. R. et al. Identification of the +2 oxidation state for uranium in a crystalline molecular complex, [K(2.2.2-cryptand)][(C5H4SiMe3)3U]. J. Am. Chem. Soc. 135, 13310–13313 (2013).
La Pierre, H. S., Scheurer, A., Heinemann, F. W., Hieringer, W. & Meyer, K. Synthesis and characterization of a uranium(II) monoarene complex supported by δ backbonding. Angew. Chem. Int. Ed. 53, 7158–7162 (2014).
Langeslay, R. R., Fieser, M. E., Ziller, J. W., Furche, F. & Evans, W. J. Synthesis, structure, and reactivity of crystalline molecular complexes of the {[C5H3(SiMe3)2]3Th}1− anion containing thorium in the formal +2 oxidation state. Chem. Sci. 6, 517–521 (2015).
Wooles, A. J., Mills, D. P., Lewis, W., Blake, A. J. & Liddle, S. T. Lanthanide tri-benzyl complexes: structural variations and useful precursors to phosphorus-stabilised lanthanide carbenes. Dalton Trans. 39, 500–510 (2010).
Cooper, O. J. et al. Uranium-carbon multiple bonding: facile access to the pentavalent uranium carbene [U{C(PPh2NSiMe3)2}(Cl)2(I)] and comparison of UV=C and UIV=C double bonds. Angew. Chem. Int. Ed. 50, 2383–2386 (2011).
Ma, G., Ferguson, M. J., McDonald, R. & Cavell, R. G. Actinide metals with multiple bonds to carbon: synthesis, characterization, and reactivity of U(IV) and Th(IV) bis(iminophosphorano)methandiide pincer carbene complexes. Inorg. Chem. 50, 6500–6508 (2011).
Lu, E. et al. Uranium-carbene-imido metalla-allenes: ancillary-ligand-controlled cis-/trans-isomerisation and assessment of trans-influence in the R2C=UIV=NR' unit (R=Ph2PNSiMe3; R'=CPh3). Chem. Eur. J. 22, 11559–11563 (2016).
Gregson, M. et al. A cerium(IV)-carbon multiple bond. Angew. Chem. Int. Ed. 52, 13016–13019 (2013).
Gregson, M., Wooles, A. J., Cooper, O. J. & Liddle, S. T. Covalent uranium carbene chemistry. Comm. Inorg. Chem. 35, 262–294 (2015).
Levin, J. R., Dorfner, W. L., Carroll, P. J. & Schelter, E. J. Control of cerium oxidation state through metal complex secondary structures. Chem. Sci. 6, 6925–6934 (2015).
Piro, N. A., Robinson, J. R., Walsh, P. J. & Schelter, E. J. The electrochemical behavior of cerium(III/IV) complexes: thermodynamics, kinetics and applications in synthesis. Coord. Chem. Rev. 260, 21–36 (2014).
Patel, D. et al. A formal high oxidation state inverse-sandwich diuranium complex: a new route to f-block-metal bonds. Angew. Chem. Int. Ed. 50, 10388–10392 (2011).
Brown, J. L., Fortier, S., Lewis, R. A., Wu, G. & Hayton, T. W. A complete family of terminal uranium chalcogenides, [U(E)(N{SiMe3}2)3]− (E=O, S, Se, Te). J. Am. Chem. Soc. 134, 15468–15475 (2012).
Patel, D. et al. An actinide-zintl cluster: a tris(triamidouranium)μ3-η2:η2:η2-heptaphosphanortricyclane and its diverse synthetic utility. Angew. Chem. Int. Ed. 52, 13334–13337 (2013).
King, D. M. et al. Synthesis and characterization of an f-block terminal parent imido [U=NH] complex: a masked uranium(IV)-nitride. J. Am. Chem. Soc. 136, 5619–5622 (2014).
Halter, D. P., La Pierre, H. S., Heinemann, F. W. & Meyer, K. Uranium(IV) halide (F−, Cl−, Br−, and I−) monoarene complexes. Inorg. Chem. 53, 8418–8424 (2014).
Gardner, B. M. et al. Triamidoamine uranium(IV)-arsenic complexes containing one-, two-, and three-fold U–As bonding interactions. Nat. Chem. 7, 582–590 (2015).
Gregson, M. et al. Emergence of comparable covalency in isostructural cerium(IV)- and uranium(IV)-carbon multiple bonds. Chem. Sci. 7, 3286–3297 (2016).
Yamada, M. et al. Positional control of encapsulated atoms inside a fullerene cage by exohedral addition. J. Am. Chem. Soc. 127, 14570–14571 (2005).
Mattausch, H., Hoch, C. & Simon, A. Crystal structure of dodecacerium heptadecaiodide triethanide, Ce12I17(C2)3 . Z. Kristallogr. New Cryst. Struct. 220, 301–302 (2005).
Mattausch, H., Hoch, C. & Simon, A. Three new ethanide iodides of La: La5I9(C2), La6I10(C2) and La10I15(C2)2 . Z. Anorg. Allg. Chem. 631, 1423–1429 (2005).
Mattausch, H. et al. EYPHKAMEN: Ln octahedron triples in Ln14(C2)3I20 with Ln=La, Ce. Z. Anorg. Allg. Chem. 632, 1661–1670 (2006).
Mattausch, H., Schaloske, M. C., Hoch, C., Zheng, C. & Simon, A. Rare earth halides Ln4X5Z. Part 1: C and/or C2 in Ln4X5Z. Z. Anorg. Allg. Chem. 634, 491–497 (2008).
Mattausch, H., Schaloske, M. C., Hoch, C. & Simon, A. Rare earth halides Ln4X5Z. Part 2: an orthorhombic variant of Ln4X5Z structure. Z. Anorg. Allg. Chem. 634, 498–502 (2008).
Roos, B. O. & Pyykkö, P. Bonding trends in molecular compounds of lanthanides: the double-bonded carbene cations LnCH2+, Ln=Sc, Y, La-Lu. Chem. Eur. J. 16, 270–275 (2010).
Clark, D. L., Gordon, J. C., Hay, P. J. & Poli, R. Existence and stability of lanthanide−main group element multiple bonds. new paradigms in the bonding of the 4f elements. A DFT study of Cp2CeZ (Z=F+, O, NH, CH−, CH2) and the ligand adduct Cp2Ce(CH2)(NH3). Organometallics 24, 5747–5758 (2005).
Shannon, R. D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. Sect. A 32, 751–767 (1976).
Mills, D. P., Soutar, L., Lewis, W., Blake, A. J. & Liddle, S. T. Regioselective C-H activation and sequential C-C and C-O bond formation reactions of aryl ketones promoted by an yttrium carbene. J. Am. Chem. Soc. 132, 14379–14381 (2010).
Cooper, O. J. et al. The nature of the U=C double bond: pushing the stability of high-oxidation state uranium carbenes to the limit. Chem. Eur. J. 19, 7071–7083 (2013).
Bogart, J. A. et al. Homoleptic cerium(III) and cerium(IV) nitroxide complexes: significant stabilization of the 4+ oxidation state. Inorg. Chem. 52, 11600–11607 (2013).
Löble, M. W. et al. Covalency in lanthanides. An X-ray absorption spectroscopy and density functional theory study of LnCl6x− (x=3,2). J. Am. Chem. Soc. 137, 2506–2523 (2015).
Mills, D. P. et al. Synthesis of a uranium(VI)-carbene: reductive formation of uranyl(V)-methanides, oxidative preparation of a [R2C=U=O]2+ analogue of the [O=U=O]2+ uranyl ion (R=Ph2PNSiMe3), and comparison of the nature of UIV=C, UV=C and UVI=C double bonds. J. Am. Chem. Soc. 134, 10047–10054 (2012).
Acknowledgements
We are grateful to the Royal Society, the UK Engineering and Physical Sciences Research Council, the Marie Curie IIF Scheme, the European Research Council, The University of Nottingham, The University of Manchester, the UK National Nuclear Laboratory and COST Action CM1006 for generous support and funding, the National Service for Computational Chemistry Software for access to the Columbus HPC facility and the UK EPSRC National EPR Facility. We thank Dr Emily Smith (University of Nottingham) for recording the XPS spectrum of 3Ce.
Author information
Authors and Affiliations
Contributions
M.G., E.L. and D.P.M. synthesized and characterized the compounds. F.T. and E.J.L.M. recorded and analysed the EPR and SQUID data. C.H. and A.C.S. recorded and analysed the XANES data. J.M. and S.T.L. conducted and analysed the DFT and NBO calculations. W.L. and A.J.B. carried out the single crystal X-ray diffraction work. A.K. conducted and analysed the CASSCF/RASSCF and QTAIM data. S.T.L. originated the central idea, supervised the work, analysed the data and wrote the manuscript with contributions from all co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures, Supplementary Tables, Supplementary Methods and Supplementary References (PDF 10623 kb)
Supplementary Data 1
Crystallographic Information File for 1TB (CIF 961 kb)
Supplementary Data 2
Crystallographic Information File for 2Ce (CIF 1353 kb)
Supplementary Data 3
Crystallographic Information File for 2Pr (CIF 1123 kb)
Supplementary Data 4
Crystallographic Information File for 2Tb (CIF 1169 kb)
Supplementary Data 5
Crystallographic Information File for 3Ce (CIF 915 kb)
Supplementary Data 6
Crystallographic Information File for 3U (CIF 1578 kb)
Supplementary Data 7
Crystallographic Information File for 3Th (CIF 851 kb)
Supplementary Data 8
Crystallographic Information File for 5Th (CIF 1683 kb)
Supplementary Data 9
Crystallographic Information File for 5U (CIF 1143 kb)
Supplementary Data 10
Crystallographic Information File for 6Pr (CIF 979 kb)
Supplementary Data 11
Crystallographic Information File for 6Tb (CIF 861 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Gregson, M., Lu, E., Mills, D. et al. The inverse-trans-influence in tetravalent lanthanide and actinide bis(carbene) complexes. Nat Commun 8, 14137 (2017). https://doi.org/10.1038/ncomms14137
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms14137
This article is cited by
-
A terminal neptunium(V)–mono(oxo) complex
Nature Chemistry (2022)
-
Evidence for ligand- and solvent-induced disproportionation of uranium(IV)
Nature Communications (2021)
-
Isolation and characterization of a covalent CeIV-Aryl complex with an anomalous 13C chemical shift
Nature Communications (2021)
-
Creation of an unexpected plane of enhanced covalency in cerium(III) and berkelium(III) terpyridyl complexes
Nature Communications (2021)
-
Emergence of the structure-directing role of f-orbital overlap-driven covalency
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.