Abstract
β-Lactamases enable resistance to almost all β-lactam antibiotics. Pioneering work revealed that acyclic boronic acids can act as ‘transition state analogue’ inhibitors of nucleophilic serine enzymes, including serine-β-lactamases. Here we report biochemical and biophysical analyses revealing that cyclic boronates potently inhibit both nucleophilic serine and zinc-dependent β-lactamases by a mechanism involving mimicking of the common tetrahedral intermediate. Cyclic boronates also potently inhibit the non-essential penicillin-binding protein PBP 5 by the same mechanism of action. The results open the way for development of dual action inhibitors effective against both serine- and metallo-β-lactamases, and which could also have antimicrobial activity through inhibition of PBPs.
Similar content being viewed by others
Introduction
The β-lactamase-catalysed hydrolysis of β-lactam antibiotics (BLAs) is of central importance in antibiotic resistance1. β-Lactam-based inhibitors (for example clavulanic acid) of the Class A serine-β-lactamases (SBLs) are widely used in combination with penicillins2. Recently, avibactam, an inhibitor of Class A, C and some Class D SBLs, has been introduced for clinical use in combination with a cephalosporin1. Though not a β-lactam, avibactam is susceptible to β-lactamase-catalysed hydrolysis1. In contrast to SBLs, there are no clinically useful inhibitors of the Class B zinc-dependent metallo-β-lactamases (MBLs), which are of growing concern as a cause of antibiotic failure. With the exception of the monobactams, MBLs catalyse the hydrolysis of all β-lactam families including penicillins, cephalosporins, carbapenems and SBL inhibitors3.
SBLs and the penicillin-binding protein (PBP) targets of the β-lactams are evolutionarily and mechanistically related; as a consequence, several β-lactam classes, for example, carbapenems, can inhibit both SBLs and PBPs4. MBLs, however, are mechanistically and structurally distinct, and constitute a heterogeneous group2. The requirement for clinically useful inhibition of a broad spectrum of clinically relevant MBL subfamilies (NDM, IMP, VIM, SPM), which differ in the loops surrounding their active site, makes them challenging medicinal chemistry targets5.
Since many bacteria have acquired both SBL- and MBL-mediated resistance1, we are interested in identifying dual action MBL/SBL inhibitors. Very few potent inhibitors (IC50<1 μM) targeting SBLs, MBLs and/or PBPs have been developed. Since transient oxyanionic species (for example the ‘tetrahedral intermediate’ of SBLs) produced by nucleophilic attack onto the β-lactam carbonyl are likely common to SBL- and MBL-catalysed β-lactam hydrolysis3,6, we reasoned analogues of this intermediate may provide the desired dual action-BL activity. While such ‘tetrahedral intermediate’ analogues are well-characterized for nucleophilic enzymes, including PBPs and SBLs2, they have not been widely described for metallo-hydrolases. The observation of MBL inhibition by trifluoromethyl ketones7 is evidence that mimicking a tetrahedral intermediate may also be useful for the inhibition of MBLs. Since acyclic boronic acids, are established as SBL/PBP inhibitors1 (the SBL inhibitor, RPX7009 (ref. 1), is in clinical trials), we screened various boronic acids, including some reported to be SBL/PBP inhibitors, for inhibition of the NDM-1 MBL. Interestingly, cyclic boronates, but not the acyclic boronic acids, manifested potent MBL inhibition. We therefore synthesized and tested additional boronic acids, including compounds (2, 4 and 5) described in the patent literature as β-lactamase inhibitors8 and novel derivatives 1 and 3 (designed using modeling).
We demonstrate through biochemical, biophysical and cellular evidence that cyclic boronates are potent inhibitors of both SBLs and MBLs. Interestingly, we also found that the cyclic boronates inhibit the PBP targets of the BLAs. High-resolution crystallographic analyses reveal the proposed mechanism of action. The cyclic boronates act as ‘transition state analogues’ for both ‘serine’ and ‘metallo’ enzymes and therefore represent a promising strategy for combating antibiotic resistance.
Results
MBL inhibition by cyclic boronates
Using a fluorogenic assay for MBLs9, we screened the cyclic boronates (Fig. 1) against a representative panel of clinically relevant B1 subfamily MBLs, including IMP-1 (Imipenemase-1), VIM-2 (Verona-Integron-Encoded MBL-2), NDM-1 (New Delhi MBL-1), SPM-1 (São Paulo MBL-1) and the model MBL, BcII from Bacillus cereus2. The results imply that cyclic boronates with an aromatic side chain, positioned analogously to the 6β/7β side chains of the penicillins/cephalosporins, are potent inhibitors of B1 MBLs (for example, pIC50 values for 5 range from 8 to 9 against VIM-2 and NDM-1; Table 1). In support of these findings, 19F-protein NMR studies, employing site-selective labelling of M67C NDM-1 with 1,1,1-trifluoro-3-bromo acetone10, reveal tight binding of 2 to the active site of the NDM-1 MBL (Supplementary Fig. 1). In vitro inhibition of MBLs by the tested cyclic boronates yielded the following rank order of potency: VIM-2>NDM-1>BcII>IMP-1>SPM-1 (Table 1). As SPM-1 (a ‘hybrid’ enzyme with properties of both the B1/B2 MBL subfamilies11) was inhibited least strongly (IC50 13–36 μM), we investigated inhibition of Aeromonas hydrophilia CphA12 as a representative of the mono-Zn(II) B2 MBL subfamily and observed similar inhibition potency (high μM range, Table 1), suggesting that the tested cyclic boronates may be less potent against B2 MBLs. Overall, these data identify 2 and 5 as highly potent inhibitors of VIM-2 and NDM-1, respectively, the most widely distributed members of the clinically important B1 subfamily (Table 1).
SBL and PBP inhibition by cyclic boronates
We then used fluorogenic assays9 to measure the potency of the cyclic boronates against clinically relevant Class A and Class D SBLs, including TEM-1 (Class A) and OXA-10 (Class D)13. All of the compounds tested were potent TEM-1 inhibitors (IC50 6→0.3 nM, Table 1) and compounds with saturated side chains (1 and 3) manifested IC50 values<1 μM against OXA-10 (Table 1). Although our initial objective was to identify and characterize compounds potent against both MBLs and SBLs, the observed potency of the cyclic boronates versus SBLs motivated us to also investigate their potential for inhibition of a mechanistically-related PBP.14 PBP 5 (dacA) from Escherichia coli, which is a non-essential PBP15, was potently inhibited by all tested cyclic boronates (residual activity<1% at 10 μM; IC50 for 2, 1.7 nM). We also tested the essential PBP 3 from Pseudomonas aeruginosa at 100 μM against the cyclic boronates, but no inhibition was detected (Table 1). These results reveal the potential for cyclic boronates to act as broad-spectrum inhibitors of SBLs and MBLs with activity against, at least some, PBPs.
Pathogen susceptibility to cyclic boronate
Since 2 was a potent inhibitor of all three enzyme classes in vitro, we next tested its activity against bacterial cells (the Gram-negative pathogens E. coli and Klebsiella pneumoniae) in antimicrobial susceptibility assays (Table 2). 2 was evaluated against multiple strains of both organisms and, alone and in combination with the carbapenem meropenem, against recombinant K. pneumoniae16 and previously described K. pneumoniae and E. coli strains of clinical origin.17 These strains all carry the NDM-1 MBL, together with multiple SBLs of different classes17. 2 alone did not display antibacterial activity against any of the strains tested at concentrations up to 128 μg ml−1 (for E. coli ATCC 25922 and K. pneumoniae NCTC 5055, data not shown). The minimal inhibitory concentrations (MICs) determined for NDM-1-expressing K. pneumoniae Ecl8 in the presence of compound 2 at concentrations ranging from 0.5 to 64 μg ml−1 demonstrated a dose-dependent increase in susceptibility as the concentration of 2 increased (Supplementary Table 1). For all the strains producing NDM-1, co-administration with 2 reduced the MIC of meropenem. Clear reductions in meropenem MIC were observed at 10 μg ml−1 2. At a concentration of 25 μg ml−1, 2 brought the meropenem MIC into the susceptible range (MIC<8 μg ml−1) for recombinant K. pneumoniae strain Ecl8 and its ΔramR mutant derivative (which has reduced envelope permeability through reduced porin and increased efflux pump production) when both are producing NDM-1. Neither 1 nor 2 showed cytotoxicity in human HEK293 cells when administered at concentrations up to 100 μM (Supplementary Fig. 2).
Structural analysis of cyclic boronate binding to BcII and VIM-2
We then investigated the mechanism of MBL inhibition by 2 using X-ray crystallography. We obtained high-resolution structures for VIM-2 and BcII in complex with 2 to 1.5 Å and 1.9 Å resolution, respectively (Supplementary Figs 3–6). In both these structures, the electron density for the inhibitor clearly defines the geometry of the boron as tetrahedral (sp3).
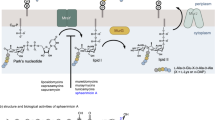
For the B1 subfamily MBLs, β-lactam hydrolysis is proposed to proceed via nucleophilic attack of a hydroxide ion onto the β-lactam carbonyl; in ground state structures, this hydroxide bridges the two active site zinc ions (Zn1 and Zn2)17. A transient tetrahedral oxyanionic species (Fig. 2a) is formed that reacts to give a Zn1-bound carboxylate and a, sometimes detectable, intermediate18 in which the negatively charged β-lactam-derived nitrogen coordinates to Zn2; protonation of this intermediate is required for product release. The mode of binding of 2 observed here, in which both boron-bound oxygen atoms participate in bidentate coordination of the Zn1 ion, most closely resembles that predicted for the tetrahedral oxyanion formed during β-lactam hydrolysis, with the implication that these inhibitors act as mimics of this state (Fig. 2b,c).
(a) Outline mode of action of MBLs showing proposed intermediates. (b,c) Views from structures obtained by co-crystallization of 2 with BcII (b) and VIM-2 (Chain A) (c). Note the relative position of Trp87 is rotated by ∼180° compared with its position in structures of BcII and VIM-2 without inhibitor; in the case of VIM-2, Trp87 interacts with the cyclic boronate via a water molecule. (d) The overlay compares the binding modes of 2 and hydrolysed cefuroxime in complex with NDM-1 (PDB ID: 4RL2)). (e) Key active site interactions made by the cyclic boronates.
The structures of both BcII and VIM-2 in complex with 2 reveal binding modes similar in some key aspects to those observed for complexes of B1 MBLs with hydrolysed β-lactams, in particular for a ring-opened cephalosporin intermediate19 (Fig. 2d). First, one oxygen atom of the inhibitor C-3 carboxylate coordinates to Zn2; the other carboxylate oxygen interacts with Lys224 (NDM-1 and BcII) or Arg228 (VIM-2) by hydrogen-bonding/electrostatic interactions. These interactions are analogous to those with hydrolysed β-lactams where the C-3/C-4 carboxylate (of penicillins/cephalosporins, respectively) binds Zn2 and Lys224 (BcII and NDM-1) or Arg228 (in the VIM family) (Fig. 2b)3,19. Second, both the BcII and VIM-2 structures reveal that in the MBL-active sites the bicyclic phenyl-boronate ring of 2 is very similarly positioned to the cephalosporin dihydrothiazine ring (or analogous penicillin/carbapenem derived rings)19, with both being positioned to make hydrophobic interactions with the conserved Trp87 and Phe61 residues. Notably, the ‘endocyclic’ boronate ester oxygen of 2 coordinates to Zn2, mimicking the coordination of the cephalosporin-derived dihydrothiazine ring nitrogen in the anionic intermediate. Third, the binding mode of the side chain of 2 is analogous to that of the 7β side chain of cephalosporins (Fig. 2b,d), in that the carbonyl oxygen of the 7β-acetamido group of the side chain of 2 and the cephalosporin intermediate are both positioned to hydrogen bond with the main chain NH group of residue-119 (Ala (BcII), Asp (VIM-2) or Glu (NDM-1)). Fourth, as observed in a complex of NDM-1 with a cephalosporin-derived intermediate, the C-6 carboxylate arising from β-lactam hydrolysis is positioned to coordinate Zn1 and hydrogen bond via one of its oxygen atoms to the Asn233 side chain. Fifth, binding of the two ‘exocyclic’ boron-bound oxygens/hydroxides mimics the binding modes proposed for the two oxygens in the oxyanion intermediate. The pro-(S) boron-bound exocyclic oxygen coordinates with Zn1 and is positioned to hydrogen bond with Asn233 and the NH of the acetamido side chain. In MBL catalysis, the highly conserved Asn233 side chain is proposed to stabilize the oxyanionic intermediate via hydrogen bonding with the lactam carbonyl-derived oxygen. 20 (The structures suggest that hydrogen bonding to the substrate side chain is also involved in stabilizing the oxyanion). The pro-(R) boron-bound exocyclic oxygen bridges Zn1 and Asp120, mimicking the proposed position of the hydroxide-derived oxygen in the oxyanion intermediate. Overall, the complexes of 2 with VIM-2 and BcII reveal that binding of the cyclic boronates closely mimics that proposed for the high-energy tetrahedral intermediate in MBL catalysis21 (Fig. 2a,e), both in terms of structure and bonding.
Structural analysis of cyclic boronate binding to OXA-10
We then investigated the binding mode of the cyclic boronates to SBLs, by determining a high-resolution crystal structure of the Class D β-lactamase OXA-10 in complex with 1 (1.5 Å resolution; Supplementary Fig. 3,7). OXA-10 requires a carbamylated active site lysine (KCX70) for catalysis22 (Fig. 3a), which was observed in the OXA-10:1 complex structure (Fig. 3b, Supplementary Fig. 3). Inspection of maps (Supplementary Fig. 3) reveals continuous electron density connecting the inhibitor boron atom with the terminal hydroxyl group of the nucleophilic serine (Ser67) and provides clear evidence for a tetrahedral boron atom, contrasting with the planar geometry of the carbonyl carbon atoms of β-lactam acyl-enzyme complexes (Supplementary Fig.8). The structure of the complex with 1 thus represents a covalent species that better resembles the ‘first’ tetrahedral intermediate, which is involved in acyl-enzyme formation, rather than the acyl-enzyme itself (Fig. 3a). Hence, analogous to our structural observations with the VIM-2 and BcII MBLs (above), the OXA-10:1 complex structure reveals that 1 mimics the mechanistically important tetrahedral oxyanion that is transiently present on the hydrolytic pathway. Furthermore, comparison of the OXA-10:1 complex with that of a penicillin (benzylpenicillin) bound to a ‘deacylation-deficient’ OXA-10 (K70C variant)23 reveals strikingly related overall binding modes. In particular, the carboxylate of 1 and the C-3 carboxylate of penicillin (Fig. 3b,c) are both positioned to interact with Arg250, while the acetamido group of 1 and the benzylpenicillin C6 amide group are analogously positioned to hydrogen bond with the main chain carbonyl of Phe208. Finally, the endocyclic boronate ester oxygen of 1 and the β-lactam-derived nitrogen (from the penicillin thiazolidine ring) are both positioned to hydrogen bond with Ser115.
Structural analysis of cyclic boronate binding to PBP 5
We then carried out crystallographic studies to investigate the binding mode of the cyclic boronates to PBPs (Fig. 4a), by solving a crystal structure of PBP 5 in complex with 2 (2.5 Å resolution; Supplementary Fig. 3,9). Comparison of the PBP 5:2 complex structure with reported PBP5 structures reveals that the fold of Domain 1 (aa 3–262) of PBP 5, which contains the transpeptidase/penicillin-binding active site, is very similar in both structures (Supplementary Fig. 10); However, clear differences are observed in the position of Domain 2 (aa 263–356).
To date, all reported PBP 5 structures with ligands (substrate-like peptide boronic acids or acyl-enzyme complexes with penicillins, cephalosporins or carbapenems) manifest weak or absent density for the C6 or C7 side chains of the penicillins or cephalosporins, respectively, or the C3′ cephalosporin side chain24,25,26. Importantly, for the PBP 5:2 complex, inspection of the experimental map revealed clear electron density for the whole inhibitor molecule (Supplementary Fig. 3).
Comparison of the PBP 5:2 complex with a PBP 5:cloxacillin acyl-enzyme structure25 reveals similar binding modes for the two compounds in the active site (Fig. 4b,c), including with respect to (i) interactions with the conserved active site motifs (SxxK and SxN), the conformations of which overlap almost exactly in the two structures; (ii) conservation of the predicted active site hydrogen-bonding network; and (iii) the same bidendate interaction of Arg248 with the carboxylate of 2 or the C-3 penicillin carboxylates25.
Three other features of the PBP 5:2 complex structure are of interest. First, the conserved Ω-like loop that is conserved in PBPs and Class A SBLs adopts a ‘closed’ position (that is, closer to the active site) when complexed with 2, compared with previously published apo- or acyl-enzyme structures (Supplementary Fig. 11). Interestingly, in the structure of PBP 5 complexed with substrate-like peptide boronic acid, the Ω-like loop was refined in dual conformations, one similar to that observed in the PBP5:2 structure and one similar to the apo- and the acyl-enzyme structures (Supplementary Fig. 11). Second, compared with the previous acyl-enzyme structures, in the structure with 2, His216 moves >3.5 Å to interact with the carboxylate of 2, which is analogous to binding of the penicillin C-3 carboxylate, implying an important role for this residue in substrate binding. Third, the ‘bridging’ water molecule that is proposed to polarize and orient the hydrolytic water molecule for hydrolysis in the acyl-enzyme complex was absent in the PBP 5:2 structure (Supplementary Fig. 12).
Discussion
The combined results reveal that cyclic boronates act as dual action inhibitors of SBLs and MBLs by mimicking a common high-energy tetrahedral intermediate; they also reveal that cyclic boronates can potently inhibit some PBPs via the same mechanism. Cyclic boronates thus represent a promising line of investigation not only for the protection of BLAs from both MBLs and/or SBLs, but also for direct inhibition of PBPs. The structural results presented here will aid in the development of optimized cyclic boronates, possibly with activity against the essential PBPs.
The demonstrated utility of a single compound (class) to simultaneously inhibit both nucleophilic serine- and metallo-hydrolases is also of interest in other drug discovery fields, including cancer, where it may be desirable to inhibit different classes of functionally related proteases27. Notably, the cyclic boronates, but not acyclic boronic acids, were potent MBL inhibitors, suggesting that cyclic, or otherwise conformationally constrained transition state analogues, either employing boronates or related functional groups that mimic intermediates (for example, fluoromethyl ketones) may represent a generally productive route towards simultaneous inhibition of mechanistically different types of hydrolases by the same compound.
Methods
Synthesis
the procedures for the synthesis of cyclic boronates are described in Supplementary Methods. For NMR and MS analysis of the compounds in this article, see Supplementary Figs 13–59.
Protein production and purification
Recombinant forms of NDM-1, NDM-1 M67C, VIM-2, SPM-1, IMP-1, BcII, CphA, Tem-1, OXA-10, PBP 3 and PBP 5 were produced in E. coli as described9,10,23,28,29,30. Purified BcII, VIM-2, OXA-10 and PBP 5 proteins were separately dialysed into fresh crystallization buffer (50 mM HEPES pH 7.5, 150 mM NaCl and 1 μg ZnCl2 for BcII and VIM-2 or 50 mM MES pH 6.0, 100 mM NaCl for OXA-10 or 50 mM TRIS pH 7.5 and 500 mM NaCl for PBP 5).
Crystallography
Crystals of BcII:2, VIM-2:2, OXA-10:1 and PBP5:2 were grown using the conditions stated in Supplementary Table 2. Crystals were cryoprotected using well solution diluted to 25% glycerol and harvested using nylon loops then flash-cooled and stored in liquid nitrogen. Diffraction data for VIM-2:2 and OXA-10:1 crystals were collected at the Diamond Light Source synchrotron beamline I04, data for BcII:2 were collected using a Rigaku FRE+ Superbright diffractometer and data for PBP5:2 were collected at the Diamond Light Source synchrotron beamline I02. All data sets were collected at 100 K. Data for BcII, VIM-2 and PBP 5 were indexed, integrated and scaled using HKL-2000 and for OXA-10 with Mosflm and Scala, respectively31. All structures were solved by molecular replacement using Phaser32 using the previosly published VIM-2 (PDB ID: 4BZ3)17, OXA-10 (PDB ID: 1K6R)22, BcII (PDB ID: 4C09)17 or PBP 5 (PDB ID: 1NZO) as search models for their respective data set. The structures were then iteratively fit and refined using COOT33 and PHENIX31 until Rwork and Rfree no longer converged. Data collection and refinement statistics are given in Supplementary Table 3.
Kinetic analyses
Kinetic and inhibition analyses, and 19F NMR studies10 against bacterial β-lactamases were performed as described9. The FC-5 cephalosporin9 was used as substrate for BcII, VIM-2, IMP-1, NDM-1, SPM-1, TEM-1 and OXA-10, meropenem28 for CphA and nitrocefin34 for PBP 3 and PBP 5.
In brief IC50 values were determined by preincubation of the appropriate amount of enzyme with the cyclic boronates in the assay buffers for 10 min at room temperature prior to the initiation of the assay by the addition of the appropriate substrate. The enzyme concentrations used are the following: 1 nM BcII, 100 nM VIM-2, 10 nM IMP-1, 10 nM NDM-1, 1 nM SPM-1, 1 nM TEM-1, 1 nM OXA-10, 2.5 nM CphA, 4 μM PBP 3 and 5 μM PBP 5; note that the final concentrations are stated. The assay buffers used are the following: 50 mM HEPES, pH 7.2 supplemented with 1 μg BSA, 1 μg ZnSO4 and 0.01% Triton for BcII, VIM-2, IMP-1, NDM-1 and SPM-1, 15 mM MES, pH 6.5 for CphA, 50 mM HEPES, pH 7.2 supplemented with 200 mM NaCl and 0.01% Triton 100 for TEM-1, 100 mM sodium phosphate, pH 7.2 supplemented with 50 mM NaHCO3 and 0.01% Triton 100 for OXA-10 and 50 mM HEPES, pH 7.2 supplemented with 200 mM NaCl and 0.01% Triton 100 for PBP 3 and PBP 5. The amount of substrates used is the following: 5 μM for BcII, VIM-2, IMP-1, NDM-1 and SPM-1, 10 μM for TEM-1 and OXA-10, 125 μM for CphA and 25 μM for PBP 3 and PBP 5; note that the final concentrations are stated.
The experiments were performed using a PHERAstar FS microplate reader (BMG Labtech) at room temperature (24–25 °C). All enzymes and inhibitors were prepared in the specific assay buffer. The kinetic values in this study are the means from at least three independent measurements; at least nine different concentrations of the inhibitor were used to determine the kinetic parameters (IC50). The IC50 values were determined using the software package Graph Prism 5.
Cytotoxicity assay
HEK293 cells were seeded (2,000 per well) in normal DMEM in 96-well plates.34 After 24 h, wells were dosed with titrated concentration of compounds in 1% dimethyl sulphoxide (DMSO)—final volume of media about 100 μl. Following a further 24 h incubation period, 20 μl tetrazolium (MTS, CellTiter96Aqueous One Solution (Promega)) was added to the media in each well. The plate was incubated at 37 °C, 5% CO2 for 4 h in the dark before absorbance was read at 490 nm to determine cell proliferation values.
Microbiological susceptibility testing
Bacterial strains: E. coli ATCC 25922 (ref. 35) and K. pneumoniae NCTC 5055 (ref. 36) were used as reference strains. K. pneumoniae Ecl8 (ref. 16) and its ramR knock-out37 were used as previously described38,39. K. pneumoniae strain IR8 (ref. 40), and E. coli strains IR10 and IR60 (ref. 17) were generous gifts of Prof. T.R. Walsh. Plasmid pSU18:NDM-1 contains the complete blaNDM-1 open reading frame, together with its promoter, cloned into plasmid pSU18 (ref. 41). The recombinant plasmid was made by PCR amplifying the blaNDM-1 gene and surrounding sequences from E. coli IR10 with primers NDM1F 5′- ACACCATTAGAGAAATTTGC -3′ and NDM1R 5′- GGCGATGACAGCATCATC -3′, TA cloning the amplicon into pCR2.1 TOPO (Invitrogen) and subcloning into pSU18 using BamHI and XbaI.
MIC values were determined by broth microdilution, in triplicate, in cation-adjusted Mueller Hinton broth (Sigma) according to the Clinical Laboratory Standards Institute (CLSI) guidelines42. Experiments were carried out in microtitre plates (Corning) containing the medium plus meropenem (Sequoia Research Products, Pangbourne, UK) and/or inhibitor 2 as appropriate. Chloramphenicol (20 μg ml−1) was included in assays of strains (K. pneumoniae Ecl8 and derivative) that carried pSU18 plasmids or derivatives (pSU18:NDM-1) to ensure maintenance of plasmids throughout. Inhibitor 2 was dissolved in DMSO to 100 mg ml−1 and diluted in broth. The final concentration of DMSO in the assays was kept constant at 0.01% (experiments using 10 μg ml−1 inhibitor) or 0.03% (experiments using 25 μg ml−1 inhibitor). Plates were incubated overnight at 37 °C for 18–24 h and absorbance at 600 nm read using a Polarstar Omega (BMG LabTech) plate reader.
Data availability
Data supporting the findings of this study are available within the article (and its Supplementary Information files) and from the corresponding author upon reasonable request. Coordinates and structure factors have been deposited in the Protein Data Bank under accession codes 5FQB (BcII-2), 5FQC (VIM-2:2), 5FQ9 (OXA-10:1) and 5J8X (PBP 5:2).
Additional information
Accession codes: Coordinates and structure factors have been deposited in the Protein Data Bank under accession codes 5FQB (BcII:2), 5FQC (VIM-2:2), 5FQ9 (OXA-10:1) and 5J8X (PBP 5:2).
How to cite this article: Brem, J. et al. Structural basis of metallo-β-lactamase, serine-β-lactamase and penicillin-binding protein inhibition by cyclic boronates. Nat. Commun. 7:12406 doi: 10.1038/ncomms12406 (2016).
References
Bush, K. A resurgence of β-lactamase inhibitor combinations effective against multidrug-resistant Gram-negative pathogens. Int. J. Antimicrob. Agents 46, 483–493 (2015).
Drawz, S. M. & Bonomo, R. A. Three decades of β-lactamase inhibitors. Clin. Microbiol. Rev. 23, 160–201 (2010).
Meini, M. R., Llarrull, L. I. & Vila, A. J. Overcoming differences: the catalytic mechanism of metallo-β-lactamases. FEBS Lett. 589, 3419–3432 (2015).
Frère, J. M. & Page, M. G. Penicillin-binding proteins: evergreen drug targets. Curr. Opin. Pharmacol. 18, 112–119 (2014).
Karsisiotis, A. I., Damblon, C. F. & Roberts, G. C. A variety of roles for versatile zinc in metallo-β-lactamases. Metallomics 6, 1181–1197 (2014).
Page, M. I. & Badarau, A. The mechanisms of catalysis by metallo β-lactamases. Bioinorg. Chem. Appl. 576297 (2008).
Faridoon, U. I. & Islam, N. An update on the status of potent inhibitors of metallo-β-lactamases. Sci. Pharm. 81, 309–327 (2013).
Burns, C. J. et al. Beta-lactamase inhibitors. WO 2010/130708 A1 (2010).
van Berkel, S. S. et al. Assay platform for clinically relevant metallo-β-lactamases. J. Med. Chem. 56, 6945–6953 (2013).
Rydzik, A. M. et al. Monitoring conformational changes in the NDM-1 metallo-β-lactamase by 19F NMR spectroscopy. Angew. Chem. Int. Ed. 53, 3129–3133 (2014).
Brem, J. et al. Studying the active-site loop movement of the São Paolo metallo-β-lactamase-1. Chem. Sci. 6, 956–963 (2015).
Galleni, M. et al. Standard numbering scheme for class B β-lactamases. Antimicrob. Agents Chemother. 45, 660–663 (2001).
Bush, K. & Jacoby, G. A. Updated functional classification of β-lactamases. Antimicrob. Agents Chemother. 54, 969–976 (2010).
Egan, A. J., Biboy, J., van't Veer, I., Breukink, E. & Vollmer, W. Activities and regulation of peptidoglycan synthases. Philos. Trans. R. Soc. Lond. B Biol. Sci. 370,, (2015).
Denome, S. A., Elf, P. K., Henderson, T. A., Nelson, D. E. & Young, K. D. Escherichia coli mutants lacking all possible combinations of eight penicillin binding proteins: viability, characteristics, and implications for peptidoglycan synthesis. J. Bacteriol. 181, 3981–3993 (1999).
Fookes, M., Yu, J., De Majumdar, S., Thomson, N. & Schneiders, T. Genome sequence of Klebsiella pneumoniae Ecl8, a reference strain for targeted genetic manipulation. Genome Announc. 1, e00027–12 (2013).
Brem, J. et al. Structural basis of metallo-β-lactamase inhibition by captopril stereoisomers. Antimicrob. Agents Chemother. 60, 142–150 (2015).
Wang, Z., Fast, W. & Benkovic, S. J. Direct observation of an enzyme-bound intermediate in the catalytic cycle of the metallo-β-lactamase from Bacteroides fragilis. J. Am. Chem. Soc. 120, 10788–10789 (1998).
Feng, H. et al. Structural and mechanistic insights into NDM-1 catalyzed hydrolysis of cephalosporins. J. Am. Chem. Soc. 136, 14694–14697 (2014).
Brown, N. G., Horton, L. B., Huang, W., Vongpunsawad, S. & Palzkill, T. Analysis of the functional contributions of Asn233 in metallo-β-lactamase IMP-1. Antimicrob. Agents Chemother. 55, 5696–5702 (2011).
Aitha, M., Al-Adbul-Wahid, S., Tierney, D. L. & Crowder, M. W. Probing substrate binding to the metal binding sites in metallo-β-lactamase L1 during catalysis. MedChemComm 7, 194–201 (2016).
Vercheval, L. et al. Three factors that modulate the activity of class D β-lactamases and interfere with the post-translational carboxylation of Lys70. Biochem. J. 432, 495–504 (2010).
Baurin, S. et al. Critical role of tryptophan 154 for the activity and stability of class D β-lactamases. Biochemistry 48, 11252–11263 (2009).
Sauvage, E. et al. Crystal structures of complexes of bacterial DD-peptidases with peptidoglycan-mimetic ligands: the substrate specificity puzzle. J. Mol. Biol. 381, 383–393 (2008).
Nicola, G., Tomberg, J., Pratt, R. F., Nicholas, R. A. & Davies, C. Crystal structures of covalent complexes of β-Lactam antibiotics with Escherichia coli penicillin-binding protein 5: toward an understanding of antibiotic specificity. Biochemistry 49, 8094–8104 (2010).
Nicola, G. et al. Crystal structure of Escherichia coli penicillin-binding protein 5 bound to a tripeptide boronic acid inhibitor: a role for Ser-110 in deacylation. Biochemistry 44, 8207–8217 (2005).
Johnson, S. L. & Pellecchia, M. Structure- and fragment-based approaches to protease inhibition. Curr. Top. Med. Chem. 6, 317–329 (2006).
Bebrone, C. et al. Dramatic broadening of the substrate profile of the Aeromonas hydrophila CphA metallo-β-lactamase by site-directed mutagenesis. J. Biol. Chem. 280, 28195–28202 (2005).
Inglis, S. R., Strieker, M., Rydzik, A. M., Dessen, A. & Schofield, C. J. A boronic-acid-based probe for fluorescence polarization assays with penicillin binding proteins and β-lactamases. Anal. Biochem. 420, 41–47 (2012).
van Berkel, S. S. et al. Binding of (5S)-penicilloic acid to penicillin binding protein 3. ACS Chem. Biol. 8, 2112–2116 (2013).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Brem, J. et al. Rhodanine hydrolysis leads to potent thioenolate mediated metallo-β-lactamase inhibition. Nat. Chem. 6, 1084–1090 (2014).
Minogue, T. D. et al. Complete genome assembly of Escherichia coli ATCC 25922, a serotype O6 reference strain. Genome Announc. 2, e00969–14 (2014).
Snijders, E. P. & Goslings, W. R. O. Untersuchungen über das Scleroma respiratorium (Sklerom). I. Mitteilung: das Sklerom in Niederländisch-Indien, nebst einigen Bemerkungen über die Epidemiologie dieser Krankheit. Zbl. Bakt., I. Abt. Orig. 133, 343–348 (1934).
Rosenblum, R., Khan, E., Gonzalez, G., Hasan, R. & Schneiders, T. Genetic regulation of the ramA locus and its expression in clinical isolates of Klebsiella pneumoniae. Int. J. Antimicrob. Agents 38, 39–45 (2011).
De Majumdar, S., Yu, J., Spencer, J., Tikhonova, I. G. & Schneiders, T. Molecular basis of non-mutational derepression of ramA in Klebsiella pneumoniae. J. Antimicrob. Chemother. 69, 2681–2689 (2014).
Jimenez-Castellanos, J. C. et al. Comparative effects of overproducing the AraC-type transcriptional regulators MarA, SoxS, RarA and RamA on antimicrobial drug susceptibility in Klebsiella pneumoniae. J. Antimicrob. Chemother. 71, 1820–1825 (2016).
Yong, D. et al. Characterization of a new metallo-β-lactamase gene, bla(NDM-1), and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob. Agents Chemother. 53, 5046–5054 (2009).
Bartolome, B., Jubete, Y., Martinez, E. & de la Cruz, F. Construction and properties of a family of pACYC184-derived cloning vectors compatible with pBR322 and its derivatives. Gene 102, 75–78 (1991).
Wayne, P. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard, 10th edn. CLSI M07-A10 (American Society for Microbiology (2015).
Acknowledgements
We thank the Medical Research Council (MRC)/Canadian Grant G1100135 (R.C., J.S., C.W.G.F., J.B. and C.J.S.), MRC Grants MR/L007665/1, MC-PC-14103 and MR/N002679/1 (J.B. and C.J.S.) for funding. J.-C.J.-C. was funded by a postgraduate scholarship from CONACyT, Mexico.
Author information
Authors and Affiliations
Contributions
J.B, R.C., C.W.G.F. and C.J.S. conceived the research. R.C. with the help of C.W.G.F. synthesized and characterized compounds used for the study. J.B. carried out the kinetic studies, purified the enzymes used in biochemical studies and crystallized the inhibitors with VIM-2, OXA-10 and PBP 5. S.C. crystallized the inhibitor with BcII. J.B., S.C., M.A.M. and I.J.C. collected X-ray data and solved the crystal structures. J.-C.J.-C., M.B.A. and J.S. designed and performed the MIC experiments. J.B. and C.J.S. wrote the first draft of the manuscript. All authors discussed the results and contributed to writing the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-59, Supplementary Tables 1-3, Supplementary Methods and Supplementary References. (PDF 2874 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Brem, J., Cain, R., Cahill, S. et al. Structural basis of metallo-β-lactamase, serine-β-lactamase and penicillin-binding protein inhibition by cyclic boronates. Nat Commun 7, 12406 (2016). https://doi.org/10.1038/ncomms12406
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms12406
This article is cited by
-
Analysis of Antibacterial Action of Mammalian Host-Defense Cathelicidins and Induction of Resistance to Them in MβL-Producing Pseudomonas aeruginosa
Bulletin of Experimental Biology and Medicine (2022)
-
Imitation of β-lactam binding enables broad-spectrum metallo-β-lactamase inhibitors
Nature Chemistry (2022)
-
Integration of antibody-antigen and receptor-ligand reactions to establish a gold strip biosensor for detection of 33 β-lactam antibiotics
Science China Materials (2021)
-
Structure and mechanism of potent bifunctional β-lactam- and homoserine lactone-degrading enzymes from marine microorganisms
Scientific Reports (2020)
-
Virtual screening identifies broad-spectrum β-lactamase inhibitors with activity on clinically relevant serine- and metallo-carbapenemases
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.