Abstract
Membrane fission is an essential cellular process by which continuous membranes split into separate parts. We have previously identified CtBP1-S/BARS (BARS) as a key component of a protein complex that is required for fission of several endomembranes, including basolateral post-Golgi transport carriers. Assembly of this complex occurs at the Golgi apparatus, where BARS binds to the phosphoinositide kinase PI4KIIIβ through a 14-3-3γ dimer, as well as to ARF and the PKD and PAK kinases. We now report that, when incorporated into this complex, BARS binds to and activates a trans-Golgi lysophosphatidic acid (LPA) acyltransferase type δ (LPAATδ) that converts LPA into phosphatidic acid (PA); and that this reaction is essential for fission of the carriers. LPA and PA have unique biophysical properties, and their interconversion might facilitate the fission process either directly or indirectly (via recruitment of proteins that bind to PA, including BARS itself).
Similar content being viewed by others
Introduction
Membrane fission consists of a series of molecular rearrangements by which a tubular or neck-like bilayer joining two membranous compartments undergoes constriction and splits in two parts without leakage of contents. Fission is required for fundamental cellular processes such as the formation of transport vesicles during membrane traffic, organelle partitioning, cell division and in general for the maintenance of the compartmental organization of endomembranes. The mechanisms of fission have been studied intensely during the last decade, and multiple pathways leading to fission have been documented or proposed1,2,3,4,5. The best characterized fission processes are based on constriction and destabilization of membranes by the mechano-enzyme dynamin6,7,8,9,10, shallow membrane insertion of amphipathic protein domains2,11,12 and phase separation of lipid domains3,13. Nevertheless, key aspects of the lipid rearrangements leading to membrane fission remain elusive, and further analysis is required.
We have identified the protein CtBP1-S/BARS (henceforth, BARS) as a key player in the fission of post-Golgi tubular/pleiomorphic carriers5,14,15,16, macropinosomes17,18, COPI-dependent transport vesicles19,20,21 and in the Golgi ribbon partitioning during mitosis22,23. BARS (brefeldin A ADP-ribosylation substrate) is a member of the C-terminal-binding protein (CtBP) family, which evolutionarily derives from an ancestral dehydrogenase by gene duplication and functional differentiation into proteins involved in transcription, membrane transport, microtubule organization and synaptic transmission16. BARS itself is a dual-function protein that controls fission in the cytoplasm and gene transcription in the nucleus16,24. Structurally, BARS closely resembles the D-hydroxyacid dehydrogenases25 and features a ‘classical’ NAD(H)-binding Rossman fold26, which regulates the interconversion of BARS between a monomeric and a dimeric conformation depending on binding to NAD(H) and/or other ligands to the Rossman domain16,17,26,27,28. This conversion is critical for function because BARS can drive fission as a monomer, while it is fission-incompetent as a dimer17,19,26,28.
The mechanism of action of BARS in fission has been studied mostly in the context of the process of basolateral post-Golgi carrier formation14,15,16. Here BARS assembles into a complex that includes ARF, frequenin (also known as NCS-1), the phosphoinositide kinase PI4KIIIβ, 14-3-3γ and the kinases PKD and PAK, and functions to couple the budding of carriers with fission15,16. To induce fission, BARS must bind to 14-3-3γ through a phosphorylated serine (Ser147) in its dimerization surface15,16 (see also below). This binding locks BARS in its monomeric fission-competent conformation. However, how the 14-3-3γ-bound BARS leads to the lipid rearrangements leading to fission remains unclear.
We have previously proposed that BARS-dependent fission involves a lysophosphatidic acid (LPA) acyltransferase (LPAAT) activity, based on the following observations: an LPAAT activity is present in liver Golgi membranes which, on addition of suitable substrates, generates phosphatidic acid (PA)5; PA production induces, or correlates with, the fission of Golgi membranes5; the addition of BARS to these Golgi membranes stimulates both PA production and membrane fission5; treatments that inhibit the formation of monomeric fission-competent BARS inhibit both BARS-stimulated LPAAT activity and membrane fission5,19,28. We have also shown that recombinant BARS is associated with a slow LPAAT activity5 (see below) which is inhibited by anti-BARS antibodies, and proposed that this activity is ascribable to BARS itself5. This activity, however, was later shown not to be intrinsic to BARS29. The simplest interpretation of these collective findings is that BARS binds to and stimulates an endogenous LPAAT, and that this reaction is involved in membrane fission.
Here we have examined this hypothesis. There are 11 known lysophospholipid acyltransferases (LPLATs), four of which have been cloned and shown to transfer fatty acids from acyl-CoA to the sn-2 position of LPA to form PA (LPAATα, β, γ and δ), while others have mixed specificities for LPA and glycerol phosphate30,31. We find that (i) BARS interacts with LPAAT type δ (LPAATδ, also known as AGPAT4); (ii) this LPAAT localizes to the trans-Golgi and to post-Golgi carrier precursors at the trans-Golgi Network (TGN); (iii) the catalytic activity of LPAATδ is essential for Golgi carrier fission; (iv) BARS potently stimulates LPAATδ, and this stimulation is essential for carrier fission; (v) BARS needs to be incorporated into the PI4KIIIβ–14-3-3γ dimer–BARS complex15 to stimulate LPAATδ and induce fission. BARS thus appears to function as an adaptor/regulator protein that binds to and stimulates LPAATδ, to induce LPA–PA conversion and carrier fission. LPA and PA have unique biophysical properties that can markedly affect the organization of lipid bilayers32. Their interconversion might facilitate the fission process either directly or through the action of PA-binding proteins, including BARS itself21.
Results
LPAATδ localizes at the Golgi and binds directly to BARS
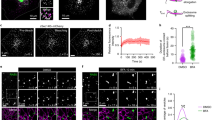
To examine whether BARS interacts with an LPAAT, we first sought to identify the LPAATs that localize to the Golgi, as most of the BARS-dependent fission reactions occur in this organelle15,16. We Flag-tagged and expressed the available mammalian LPAATs and inspected their localization by immunofluorescence microscopy. LPAATγ, LPAATδ and LPAATη localized to both the Golgi and the endoplasmic reticulum (ER) (Fig. 1a), while LPAATβ and LPAATɛ localized to the ER and mitochondria, respectively (Supplementary Fig. 1; see also refs 30, 31). We thus examined whether BARS interacts with the Golgi LPAATs by co-expressing BARS with each of these transferases and testing for co-immunoprecipitation of the two proteins. BARS co-precipitated with LPAATγ and LPAATδ (and vice versa), but not with LPAATη (Fig. 1b,c). LPAATγ has been shown to reside at the cis-Golgi and regulate Golgi structure and retrograde transport to the ER33,34, while LPAATδ has no known Golgi-related function to date. We asked whether the LPAATδ location might be compatible with a role in post-Golgi traffic by immuno-electron microscopy of the Flag-tagged protein (Fig. 1d). Tagged LPAATδ localized preferentially in the trans-Golgi and in the TGN (Fig. 1d), and to a lesser extent in the ER in the cell periphery. We also examined the distribution of the endogenous protein using specific antibodies and immunofluorescence microscopy (Fig. 2). The endogenous LPAATδ showed a localization that was very similar but not identical to that of the tagged protein, in that it was visualized mostly in the elongating tubules emanating from the TGN (Fig. 2), and gave a weaker signal in the cell periphery compared with the tagged protein. Of note, while this manuscript was being revised, it was reported that LPAATδ localizes to the mitochondrial outer membrane in murine cells35. This differs from our findings that LPAATδ localizes in the Golgi complex. However, cases of dual localization of the same transmembrane protein to the secretory pathway and to outer mitochondrial membrane have been reported several times36,37,38 and the mechanism of this dual localization is partially understood39. We thus considered that LPAATδ might localize both to the Golgi and the mitochondria, but that only the Golgi LPAATδ can be stained using the low titre antibody conditions optimized for specificity in our study, while LPAATδ in mitochondria might require a higher antibody titre for detection. We find that this is indeed the case, in all of the cell types tested (Supplementary Fig. 2a,b). Moreover, both the Golgi and mitochondrial signals were abolished by LPAATδ depletion, that is, are specific (Supplementary Fig. 2c). We conclude that LPAATδ can localize both in the secretory pathway and to a lower extent in mitochondria in most or all cells, though we do not exclude that the LPAATδ distribution might be partially cell type-dependent.
(a) Representative confocal microscopy images of COS7 cells transfected with Flag-tagged LPAATγ, LPAATδ and LPAATη, and fixed and processed for immunofluorescence with a monoclonal anti-Flag antibody (green) and with a polyclonal anti-TGN46 antibody (red; as indicated). Dotted lines indicate cell borders. (b) BARS immunoprecipitation (IP:BARS) of lysate from HeLa cells co-expressing BARS and LPAATγ–Flag, LPAATδ–Flag or LPAATη–Flag. Representative western blotting (antibodies as indicated) of total lysate (input) and immunoprecipitated proteins with preimmune-IgG (Preim-IgG) or anti-BARS-IgG (as indicated). IgGH, IgG heavy chain. (c) Immunoprecipitation with an anti-Flag antibody (IP:Flag) of lysate from HeLa cells co-transfected with BARS and LPAATγ–Flag, LPAATδ–Flag or the empty vector. Representative western blotting of total lysate (input) and Flag-immunoprecipitated proteins with an anti-Flag monoclonal antibody or the anti-BARS polyclonal antibody (as indicated). (d) Representative electron microscopy image of HeLa cells transfected with Flag-tagged LPAATδ for 24 h, and fixed and processed for cryo-immuno-electron microscopy with a monoclonal anti-Golgin-97 antibody (15-nm gold particles) and with a polyclonal anti-LPAATδ antibody (10-nm gold particles). Molecular weight standards (kDa) in b and c, are indicated on the left of each panel. Data are representative of three independent experiments. Scale bars, 10 μm (a); 200 nm (d).
(a) Representative confocal microscopy image of HeLa cells at steady-state fixed and labelled with a polyclonal anti-LPAATδ antibody (endogenous LPAATδ; red), with a monoclonal anti-GM130 antibody (green) and with an anti-TGN46 antibody (blue). Inset, bottom: Magnification of Golgi area. (b) Line scan across the Golgi area (red line across the magnified image in a) indicates the colocalization of LPAATδ with TGN46. (c) Representative confocal microscopy images of COS7 cells at steady-state (top) or VSV-infected and subjected to the VSVG TGN-exit assay (bottom). Cells were fixed and labelled with a polyclonal anti-LPAATδ antibody (endogenous LPAATδ; green) and with an anti-TGN46 antibody (red; top) or a monoclonal anti-VSVG antibody (red; bottom). Inset, right: magnification of tubular carrier precursors. Scale bars, 10 μm (a,c).
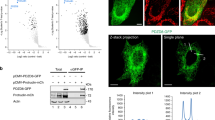
We then asked whether BARS binds directly to LPAATδ by performing pull-down experiments with the two purified recombinant proteins. After coincubation, recombinant BARS and LPAATδ (prepared as described in Methods) co-precipitated efficiently (Fig. 3a). We also tested for binding between BARS and the 152–251aa portion of LPAATδ, a cytosolic segment exposed at the protein surface (see models in ref. 30). BARS co-precipitated also with this fragment (Fig. 3a). These data indicate direct binding between the two proteins.
(a) Representative GST pull-down of equimolar amounts of GST, GST-LPAATδ (aa 152–251) and GST-LPAATδ (full length) purified recombinant proteins, for His-BARS, with unbound and eluted proteins analysed by western blotting (top, anti-BARS monoclonal antibody). GST fusion proteins were revealed by Ponceau staining (bottom). (b) Representative histidine pull-down for His or His-BARS beads of lysates from COS7 cells transfected with LPAATδ–Flag. Beads were treated with buffer alone (−) or with HPLC-purified BAC (BAC+), and then incubated with the lysates. The eluted proteins were analysed by western blotting using a monoclonal anti-Flag antibody (top), with the pulled down His-BARS revealed by Ponceau staining (bottom). Molecular weight standards (kDa) are indicated on the left of each panel. Data are representative of three independent experiments.
Finally, we examined whether LPAATδ binds selectively with the monomeric fission-competent form of BARS15,16. As noted, BARS shifts between monomeric and dimeric conformations depending on ligand binding to its Rossman fold. Dimerization is promoted by the binding either to NAD(H)19,26,27,28 or to the Brefeldin A–ADP-ribosylated conjugate (BAC), an ADP-ribosylated metabolite of brefeldin A, which locks BARS in the dimeric fission-inactive conformation28. Either BARS alone or BAC-bound BARS were added to lysates from cells overexpressing LPAATδ. BARS and LPAATδ interacted efficiently as judged from co-precipitation (see above), as expected, and this interaction was markedly reduced by the binding of BARS with BAC (Fig. 3b) (NAD(H) had similar effects, not shown), indicating that LPAATδ preferentially binds monomeric BARS.
We conclude that the LPAATδ is located in the elongating tubules emanating from the TGN (Fig. 2) and binds directly and selectively the monomeric fission-active form of BARS (Fig. 3).
Bacterial LPAAT competes with LPAATδ for BARS binding
As noted, recombinant BARS purified from Escherichia coli is associated with low levels of LPAAT activity. Since this activity belongs to the bacterial enzyme5,29 (see also Supplementary Fig. 3), we carried out pull-down experiments to examine whether BARS and E. coli LPAAT might bind to each other and do so in a specific manner. His-tagged E. coli LPAAT (which can be prepared in soluble form)40 showed strong binding with BARS (Supplementary Fig. 4a), and this binding was abolished by pre-treatment of BARS with BAC or NAD(H) (Supplementary Fig. 4b,c). This indicates that, like the mammalian enzyme (Fig. 3b), the E. coli LPAAT binds selectively to monomeric BARS (Supplementary Fig. 4b,c). We also asked whether E. coli LPAAT competes with mammalian LPAATδ for binding to BARS. This was the case (though the effect was partial; Supplementary Fig. 4d), suggesting that the mammalian and bacterial LPAATs bind to the same BARS domain, and that the BARS-binding surfaces of the mammalian LPAATδ and E. coli LPAAT are conserved. Considering the evolutionary distance between the two LPAATs, this is somewhat surprising. It is conceivable that the evolutionary ancestors of LPAATδ and BARS (probably an LPAAT and a D-hydroxyacid dehydrogenase, respectively)40, which are fundamental metabolic enzymes, were interactors in an ancient metabolic multi-enzyme complex41, and that this interaction was maintained through evolution in different functional contexts.
Regardless, these data explain our previous observations that recombinant BARS from E. coli is associated with an LPAAT activity5, which, as we now know, belongs to the E. coli enzyme. They also provides a potentially useful tool (the soluble bacterial enzyme) for in vitro reconstitution of BARS-dependent fission.
LPAATδ is required for post-Golgi carrier fission
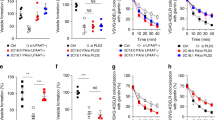
The TGN localization and the interaction of LPAATδ with BARS persuaded us to investigate the role of this LPAAT isoform in the BARS-dependent formation of post-Golgi carriers14. LPAATδ was silenced with specific small-interfering RNAs (siRNAs), and the formation of carriers, as well as the rate of transport to the plasma membrane was monitored using the temperature-sensitive vesicular stomatitis virus G protein (ts045-VSVG; henceforth, VSVG)42 as a traffic marker. The exit of VSVG out of the Golgi can be synchronized by accumulating VSVG in the TGN at 20 °C and then shifting the temperature to 32 °C14. The formation of VSVG-containing carriers from the TGN was visualized by immunofluorescence microscopy and quantified by inhibiting the fusion of these carriers with the plasma membrane with tannic acid14,15,43, which results in accumulating carriers close to the cell surface. The depletion of LPAATδ markedly reduced the formation of VSVG-containing carriers (Fig. 4a), whereas the depletion of LPAATγ, which is located at the cis-Golgi and is involved in Golgi-to-ER traffic33,34, had no effect (Fig. 4b). LPAATδ depletion had no effect on cell viability, growth and morphology for the duration of the experiment and longer.
(a,b) Representative images of COS7 cells treated with non-targeting and LPAATδ siRNAs in a or LPAATγ siRNAs in b before VSV infection and the TGN-exit assay with 0.5% tannic acid. The cells were fixed following a 20 °C block (0 min) or 30 min after the shift to 32 °C, and stained for VSVG-positive post-Golgi carriers. Quantification is on the right (see Methods). (c–e) Representative images of COS7 cells treated with non-targeting and LPAATδ siRNAs before co-transfection for the last 16 h with VSVG-ts045-KDELR-myc in c or with the endocytosis-defective LDLR–GFP receptor in d or with a plasmid encoding p75–GFP in e. (c) The distribution of the chimeric KDELR was examined following the shift to the non-permissive temperature for 30 min. Quantification of ER distribution of the chimeric KDELR (right). (d) Following the 2 h at 20 °C transport block (0 min) and 60 min after the shift to 32 °C (with cycloheximide), the cells were fixed and labelled with TGN46 (Golgi marker; red). Insets: enlarged view of merged signals for the Golgi area. (e) Following the 3 h at 20 °C block (0 min) and 60 min after the shift to 32 °C (with cycloheximide), the cells were fixed and stained for GM130 (Golgi marker; red). (d,e) Quantification of LDLRY18A–GFP in d and p75–GFP in e in the Golgi area (right). (f) Representative microscopy of HeLa cells stably transfected with hGH-FM–GFP and treated with non-targeting and LPAATδ siRNAs before subjection to a secretion assay. Release of hGH-FM–GFP from ER was performed by the addition of DD-solubilizer at 37 °C for the indicated times. Quantification of hGH-FM–GFP in the Golgi area (right). (a–e), Dotted lines indicate cell borders. (a–f), The efficiency of interference was monitored by western blotting of the cell lysates using polyclonal anti-LPAATδ in a,c–f, or polyclonal anti-LPAATγ in b antibodies. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is shown for the internal protein levels and molecular weight standards (kDa) are indicated on the left of each panel in a–f. Data are means±s.d. of three independent experiments. ***P<0.005 (Student’s t-tests). Scale bars, 10 μm.
To determine whether the reduction in VSVG-containing carriers was due to inhibition of carrier budding or of fission, we monitored carrier formation in living cells expressing VSVG–green fluorescent protein (VSVG–GFP). While control cells exhibited dynamic tubules that formed and underwent multiple fission events to generate a large number of free-moving carriers, which then fused with the plasma membrane, LPAATδ-depleted cells showed several long (>10 μm) tubular extensions that contained VSVG–GFP. These tubules represent carrier precursors that elongate out of the Golgi but do not detach to form mature transport intermediates14,15 (Supplementary Movies 1 and 2). When they (rarely) did detach, however, they could be seen to move towards, and fuse with, the plasma membrane (Supplementary Movie 2). This phenotype was similar to that induced by expressing BARS dominant-negative mutants or by depleting BARS14,15 or by depleting 14-3-3γ, a key component of the PI4KIIIβ–14-3-3γ dimer–BARS complex required for fission, or by expressing dominant-negative mutants of PKD16. Very similar effects were induced by microinjection of an affinity-purified antibody against LPAATδ (Supplementary Movie 3; see also Supplementary Fig. 5) and by the general LPAAT inhibitor CI-976 (ref. 44; Supplementary Movie 4).
These findings indicate an essential role for LPAATδ in the fission of tubular carriers exiting the Golgi complex.
We also determined the role of LPAATδ in other traffic steps. We first examined retrograde traffic from the Golgi to the ER (which is known to require LPAATγ)33,34, by monitoring the well-characterized retrograde transport marker VSVG-KDELR (a fusion of VSVG with the KDEL receptor)45. The transport of VSVG-KDELR was not affected by LPAATδ depletion (Fig. 4c). Second, since BARS controls the fission of basolateral but not apical carriers14, we examined the role of LPAATδ in Golgi export of the apical cargo p75 (ref. 46) in comparison with export of the LDL receptor (a basolateral cargo, like VSVG)47. Depletion of LPAATδ inhibited export of the LDL receptor (Fig. 4d), but not that of p75 (Fig. 4e). Finally, we probed a soluble basolateral cargo, the stably expressed constitutively secreted GFP-tagged variant of the human growth hormone (hGH)48. Depletion of LPAATδ strongly inhibited export of hGH-FM–GFP from the Golgi to the plasma membrane (Fig. 4f). Therefore, like BARS, LPAATδ appears to be selectively required for the fission of basolateral carriers.
The LPAATδ activity is needed for post-Golgi carrier fission
To examine whether the enzymatic activity of LPAATδ49 is required for fission, we first set-up an assay to determine such activity. We prepared and incubated post-nuclear supernatants with the acyl donor [1-14C]-oleoyl-CoA and the acyl acceptor oleoyl-LPA, with [1-14C]-PA measured as the reaction product (see Methods and Fig. 5a). These are standard conditions used for LPAAT assays49,50, as attempts to purify the LPAAT enzymes result in activity loss49,50. Extracts from control cells showed an efficient LPAAT activity, which was suppressed by the general LPAAT inhibitor CI-976 (Fig. 5b)34,51. A difficulty with these extracts is that they contain multiple LPAATs. We therefore designed conditions to selectively determine the LPAATδ activity (Fig. 5a), based on suppressing or overexpressing this enzyme. Extracts from LPAATδ-depleted cells (Fig. 5a) or treatment of control extracts with a specific affinity-purified antibody against LPAATδ (Fig. 5b) (see Methods for antibody characterization) showed a reproducibly lower (≈25%) activity than in controls (Fig. 5a,b), confirming that LPAATδ is responsible for a fraction of the total LPAAT activity, and in line with the presence of other LPAATs such as the abundant glycerolipid synthetic enzymes LPAATα and LPAATβ30,52. Extracts from LPAATδ-overexpressing cells showed a ≈40% increase in LPAAT activity over controls (Fig. 5a–c), and this increase was completely inhibited by antibodies against LPAATδ (Fig. 5b), down to the levels found in the absence of LPAATδ (Fig. 5a).
(a) Quantification of phosphatidic acid (PA) production in the LPAAT assay for post-nuclear supernatants from HeLa cells transfected for 48 h with an empty Flag-vector (Ctr) or with LPAATδ–Flag (LPAATδ), or for 72 h with non-targeting or LPAATδ siRNAs. The curly brackets indicate the LPAATδ-dependent and independent activities, as defined in Methods. (b) Quantification as in a, with the post-nuclear fractions also incubated with 50 μM CI-976, or polyclonal anti-LPAATδ antibody (anti-LPAATδ IgG), or anti-preimmune-IgG (anti-Preim-IgG; as control) for 30 min at 25 °C before LPAAT assay. (c) Quantification as in a, also in parallel with the LPAATδH96V–Flag (LPAATδH96V) catalytically inactive mutant. (a–c) The dashed line indicates the level of endogenous LPAAT activity not associated with LPAATδ (see text for details). Bottom: representative western blotting with an anti-Flag antibody, for the transfection efficiencies of these proteins used for the LPAAT assays. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is shown for the internal protein levels. Molecular weight standards (kDa) in a–c, are indicated on the left of each panel. (d) Representative images of COS7 cells transfected with non-targeting or LPAATδ siRNA (duplex #3; LPAATδ siRNA #3), and with LPAATδwt–Flag or the LPAATδH96V–Flag catalytically inactive mutant, and subjected to VSV infection and the TGN-exit assay with 0.5% tannic acid. The cells were fixed 30 min after the shift to the permissive temperature (32 °C) and processed for immunofluorescence with monoclonal anti-Flag and polyclonal anti-VSVG (p5D4) antibodies, to monitor formation of VSVG-containing carriers. Dotted lines show cell borders. Asterisks represent LPAATδwt–Flag and LPAATδH96V–Flag transfected cells (see inserts for staining with anti-Flag antibody; bottom images). Scale bars, 10 μm. Quantification of VSVG-positive carriers (right). Data are means±s.d. of three independent experiments. **P<0.01, ***P<0.005 versus control (Student’s t-tests).
LPAATδ silencing or overexpression did not affect the cellular levels of other LPAATs (Supplementary Fig. 6). Similar data were obtained using [1-14C]-palmitoyl-CoA as acyl donor and arachidonoyl-LPA as acyl acceptor (extracts from LPAATδ-overexpressing cells showed a ≈45% increase in LPAAT activity over controls). We thus define the LPAATδ-dependent activity (or LPAATδ activity) as the activity value of LPAATδ-overexpressing extracts (as measured using concentrations of substrates below the Km values, see below) minus the activity value of LPAATδ-depleted (or antibody-treated) extracts (Fig. 5a,b; see dashed line and Methods). The maximum reaction rate Vmax and Michaelis–Menten constant Km of this LPAATδ activity were 38±3 nmol min−1 per mg2 protein and 58±18 μM, respectively, for oleoyl-CoA, and 38±1 nmol min−1 per mg2 protein and 29±1 μM, respectively, for oleoyl-LPA (data are means±s.d. of three independent experiments). These rates are comparable to those reported for LPAATγ53. Importantly, they are potentially sufficient, depending on substrate availability, to change the PA concentrations in the TGN rapidly and substantially.
Finally, we asked whether the LPAATδ catalytic activity is required for post-Golgi carrier fission. We generated a single-point mutant (LPAATδH96V) in the conserved acyltransferase catalytic site of LPAATδ (NHX4D)30,54. Overexpressed LPAATδH96V was devoid of LPAAT activity (Fig. 5c), confirming that LPAATδ is a canonical LPAAT family member30,54. We then depleted cells of LPAATδ, with the consequent inhibition of the post-Golgi transport of VSVG (see Fig. 4a and Supplementary Movie 2) and expressed either a siRNA-resistant variant of LPAATδ or of the catalytically dead LPAATδH96V mutant. Only the wild-type LPAATδ rescued carrier formation, while LPAATδH96V was completely inactive (Fig. 5d). These data indicate that the catalytic activity of LPAATδ, and hence most likely the formation of PA from LPA, is necessary for post-Golgi carrier formation.
Activation of LPAATδ by BARS is required for carrier fission
Since BARS and LPAATδ interact directly (Fig. 3a) and are required for post-Golgi carrier fission (see refs 14, 15, and Supplementary Movies 2 and 3, respectively), we asked whether BARS regulates the enzymatic activity of LPAATδ, and whether this regulation is required for carrier fission. We first silenced BARS and measured the LPAATδ activity in cell extracts. BARS depletion abolished the LPAATδ activity (Fig. 6a). We then re-expressed BARS in BARS-silenced cells, using a siRNA-resistant replacement BARS construct (Supplementary Fig. 7a). This nearly completely restored the LPAATδ activity (Fig. 6a). As a specificity control, the above BARS manipulations did not affect the cellular levels of LPAATδ (Supplementary Fig. 7a) or of other LPAATs (Supplementary Fig. 7b). These results indicate that LPAATδ requires BARS to express its activity.
Quantification of phosphatidic acid (PA) production in the LPAAT assay for post-nuclear supernatants from HeLa cells transfected with: (a) empty Flag-vector (Ctr) or LPAATδ–Flag (LPAATδ) and with BARS siRNAs for 48 h, and with the last 12 h with siRNA-resistant replacement BARS–YFP-encoding vector (BARS re-expression); (b) LPAATδ–Flag (LPAATδ) and/or with BARS siRNAs and/or LPAATδ siRNAs. Post-nuclear fractions were incubated with immunopurified BARS (Purified BARS) for 30 min at 25 °C before LPAAT assay (as indicated). (c) Quantification of PA production in the LPAAT assay for post-nuclear supernatants from HeLa cells transfected with empty Flag-vector (Ctr) or LPAATδ–Flag (LPAATδ) for 48 h or with LPAATδ siRNAs for 72 h. The anti-BARS polyclonal antibody (Anti-BARS IgG) or anti-preimmune-IgG (Anti-Preim IgG, as control) was incubated with the indicated post-nuclear fraction for 30 min at 25 °C before LPAAT assay. (d) Quantification of PA production in the LPAAT assay for post-nuclear supernatants from HeLa cells transfected with empty Flag-vector (Ctr) or LPAATδ–Flag (LPAATδ) and with BARS siRNAs for 48 h. Post-nuclear fractions were incubated with HPLC-purified BAC (BAC+) or with buffer alone (Buffer+) for 30 min at 25 °C before LPAAT assay (as indicated). (e,f) Quantification of PA production in the LPAAT assay for post-nuclear supernatants from HeLa cells transfected with: (e) empty Flag-vector (Ctr) or LPAATδ–Flag (LPAATδ) for 48 h and the last 12 h with BARSS147A–YFP, BARSD355A–YFP or BARSwt–YFP (as indicated); (f) empty Flag-vector (Ctr) or LPAATδ–Flag (LPAATδ) for 48 h and the last 12 h with BARSwt–YFP, BARSD355A–YFP, BARSS147A–YFP or BARSS147D–YFP (as indicated). Cells were infected with VSV, subjected to TGN-exit assay and post-nuclear fractionations were prepared 10 min after the shift to 32 °C temperature-release block. The dashed line indicates the level of endogenous LPAAT activity not associated with LPAATδ (see text for details). Data are means±s.d. of three independent experiments. *P<0.05, **P<0.01, ***P<0.005 versus control (Student’s t-test). See also Supplementary Fig. 7 for LPAATδ and BARS (wild type and mutants) expression levels in post-nuclear samples.
We then sought to manipulate the BARS levels acutely in in vitro lysates to exclude transcriptional or compensatory effects that might arise in siRNA-depletion experiments16,24. We prepared extracts from LPAATδ-expressing and BARS-depleted cells, where LPAATδ is inactive (Fig. 6a), and added immunopurified BARS to the assay mixture to a final BARS concentration of 5 μg ml−1 (comparable to the levels of endogenous BARS)14. Under these conditions, BARS completely restored the LPAATδ-dependent activity (Fig. 6b) (of note, the LPAAT activity associated with immunopurified BARS was quantitatively negligible)5,29. We also added BARS to control LPAATδ-expressing extracts. This treatment only slightly stimulated the LPAATδ-dependent activity (Fig. 6b), suggesting that endogenous BARS is sufficient to activate LPAATδ, at least in extracts from quiescent cells (that is, in cells not subjected to a traffic pulse; see below). As a further control, we used extracts from cells depleted of both BARS and LPAATδ (Fig. 6b). Here addition of immunopurified BARS had no effect on the LPAAT activity, suggesting that other LPAAT isoforms are not detectably stimulated by BARS, at least under our experimental conditions.
Further along this line, we sought to inhibit BARS by adding to the LPAAT assay mixture a characterized neutralizing affinity-purified anti-BARS antibody which, when microinjected into cells, inhibits carrier fission14,15,55. This antibody inhibited the LPAATδ-dependent activity, while preimmune-IgG addition had no effect (Fig. 6c). Moreover, a pre-treatment of the assay mixture with BAC, which locks BARS in its dimeric fission-incompetent conformation and inhibits the BARS-LPAATδ binding (see above and Fig. 3b)28, reduced the LPAATδ activity (Fig. 6d), supporting the role of BARS in LPAATδ activation and indicating that monomeric BARS is required for LPAATδ to express its enzymatic activity.
We further examined the relationship between LPAATδ activity, BARS and carrier fission by expressing suitable BARS mutants. Previously, we have characterized two single-point mutants, BARSD355A and BARSS147A that have dominant-negative effects on carrier fission in living cells14,15. We tested these mutants in the LPAATδ activity assay by co-expressing each of them with LPAATδ. Both nearly completely inhibited the LPAATδ-dependent activity (Fig. 6e), again without affecting LPAATδ expression levels (Supplementary Fig. 7c). As a control, we tested the effects of overexpressing wild-type BARS (of note, wild-type BARS and the dominant-negative mutants showed comparable expression levels in these experiments; see Supplementary Fig. 7c). Overexpressed BARS did not have significant effects on the LPAATδ activity in extracts from ‘quiescent’ cells (Fig. 6e), again suggesting that basal BARS levels are sufficient to support LPAATδ activity under these conditions (see above). We noted, however, that BARS is recruited to the Golgi during a traffic pulse, which suggests that active traffic increases the requirement for BARS15, possibly to stimulate LPAATδ. Indeed, in extracts prepared during a traffic pulse, the overexpression of BARS stimulated the LPAATδ activity over control levels (Fig. 6f). Moreover, in these extracts, the expression of the fission-active BARSS147D mutant that mimics the activatory phosphorylation of BARS on Ser14715,17,18 stimulated the LPAATδ activity to an even greater extent (Fig. 6f). These collective data are consistent with the idea that LPAATδ is activated during a traffic pulse and that this activation requires higher (local) levels of BARS than those present in quiescent cells. Testing this possibility further, we examined whether the interaction between BARS and LPAATδ might be enhanced during a pulse. Co-precipitation experiments indicate that BARS co-precipitates more efficiently with LPAATδ under pulse conditions (Supplementary Fig. 8).
We also tested the role of the PI4KIIIβ–14-3-3γ dimer–BARS complex in LPAATδ activation. As noted, within this complex, 14-3-3γ binds to phosphorylated Ser147 in the BARS dimerization interface and is necessary for Golgi carrier fission15,16. The LPAATδ activity of cell extracts was markedly suppressed by 14-3-3γ depletion (Fig. 7a), while depletion of other 14-3-3 isoforms had no effect (Fig. 7b and see also Supplementary Fig. 9). Moreover, addition to cell extracts of a characterized affinity-purified anti-14-3-3γ antibody15 also suppressed the LPAATδ activity (Fig. 7c). These data indicate that 14-3-3γ is required for LPAATδ activity, presumably because it stabilizes BARS in its monomeric fission-competent conformation.
(a–c) Quantification of phosphatidic acid (PA) production in the LPAAT assay for post-nuclear supernatants from HeLa cells transfected with the empty Flag-vector (Ctr) or LPAATδ–Flag (LPAATδ) plus: (a) transfection with non-targeting siRNAs or 14-3-3γ siRNAs for 48 h (as indicated); (b) transfection with 14-3-3γ, ζ, ɛ, τ and β siRNAs for 48 h (as indicated); (c) treatment of the post-nuclear supernatant with an anti-14-3-3γ polyclonal antibody (Anti-14-3-3γ IgG) or anti-preimmune-IgG (Anti-Preim IgG, as control) for 30 min at 25 °C before the LPAAT assay. (a–c) The dashed line indicates the level of endogenous LPAAT activity not associated with LPAATδ (see text for details). Bottom: representative western blotting with an anti-Flag antibody in a–c, and with an anti-14-3-3γ monoclonal antibody in a to monitor the transfection of LPAATδ and the depletion of 14-3-3γ in the lysate used for LPAAT assay. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is shown for the internal protein levels and molecular weight standards (kDa) are indicated on the left of each panel. Data are means±s.d. of three independent experiments. *P<0.05; **P<0.01; ***P<0.005 versus control (Student’s t-tests). See also Supplementary Fig. 9 for 14-3-3s expression levels.
Finally, we repeated a few of the above experiments using a Golgi-membrane-enriched fraction (Supplementary Fig. 10a). Here the LPAATδ overexpression increased LPAAT activity by ≈60% over controls (Supplementary Fig. 10b), while LPAATδ-depletion or treatment of the membranes with the anti-LPAATδ antibody (see also Fig. 5a,b) reduced the activity by ≈35% compared with controls (Supplementary Fig. 10b,c), in line with the notion that Golgi membranes are enriched in LPAATδ over total cell extracts (compare with Fig. 5a). Also, as seen with total extracts (Fig. 6), the LPAATδ activity in Golgi membranes was nearly abolished by BARS depletion or inhibition by the anti-BARS antibody (Supplementary Fig. 10d).
In summary, a number of experimental results based on BARS silencing or overexpression, or on the use of BARS activatory or dominant-negative mutants and of anti-BARS antibodies and inhibitors, or on manipulations of the BARS-containing complex, converge towards the conclusion that the BARS-induced LPAATδ activation controls the fission of Golgi carriers. The stimulation of LPAATδ by BARS is potent, and appears to occur rapidly, most likely via the direct physical interaction between BARS and LPAATδ during assembly of the BARS protein complex that is required for carrier formation.
The BARS–LPAATδ interaction occurs at the Golgi complex
The effects of BARS and LPAATδ on the fission of carriers emanating from the Golgi suggest that this BARS–LPAATδ interaction occurs at this organelle. To verify this notion, we first re-examined the Golgi localization of BARS and of the other components of the complex, 14-3-3γ and PI4KIIIβ, focusing, in particular, on the carrier precursors elongating out of the Golgi, where fission takes place. Similar to LPAATδ (Fig. 2), these proteins were all seen to localize at the TGN (Fig. 8a) and on the VSVG-containing tubular carriers that form during synchronized exit from the TGN (Fig. 8b). Next, we directly examined whether the interaction between BARS and LPAATδ occurs at the Golgi using an approach based on Förster resonance energy transfer (FRET) and fluorescence lifetime imaging microscopy (FLIM). This technique reveals the co-presence of suitable donor and acceptor fluorophores within the same complex (that is, at a distance of ≤8 nm). We expressed E0GFP–BARS as the FRET donor, and tdTomato-LPAATδ as the acceptor, and the donor lifetime was measured to assess FRET under steady state and traffic pulse conditions. A comparison of the donor decay lifetime in control cells (cells not expressing the acceptor) and in cells co-expressing donor and acceptor showed in the latter case a marked reduction of donor lifetime at the Golgi (Fig. 8c,d), indicating that FRET, and hence interaction, between the fluorescent partners takes place (Fig. 8e). Moreover, the FRET signal at the Golgi markedly increased during a VSVG traffic pulse (Fig. 8c–e), consistent with the enhanced BARS–LPAATδ interaction observed during a pulse (Supplementary Fig. 8). These results indicate that LPAATδ is in complex with BARS in vivo at the Golgi and that it colocalizes at the TGN with BARS, 14-3-3γ and PI4KIIIβ, in agreement with the BARS–LPAATδ co-precipitation and interaction data (Figs 1 and 3 and Supplementary Fig. 8).
(a,b) Representative images of COS7 cells at steady state in a, or VSV-infected and subjected to the VSVG TGN-exit assay in b. The cells were fixed and labelled with polyclonal anti-BARS, anti-14-3-3γ or anti-PI4KIIIβ (a,b; green) antibodies, and with an anti-TGN46 antibody (a; red) or a monoclonal anti-VSVG antibody (b; red). Insets, right: magnification of the tubular carrier precursors in the Golgi area. Dotted lines indicate cell borders. Scale bars, 10 μm. (c,d) Pixel-by-pixel FLIM analysis and photon distributions of COS7 cells transfected with E0GFP–BARS alone or in combination with tdTomato-LPAATδ and fixed at the steady state or during the traffic pulse (as indicated). (c) The shortening of E0GFP–BARS donor lifetime in the presence of the tdTomato-LPAATδ acceptor due to FRET is lower in the steady-state fixed cell (where a basal interaction between the two proteins occurs) and is higher, as indicated by the blue pixels, in the traffic pulse cell (fixed after 10 min of the 32 °C temperature-release block, see Methods) where the interaction between the two proteins is quantitatively increased (see also Supplementary Fig. 8). (d) Representative photon-counting events during the time of the donor E0GFP–BARS fluorescence emission, measured in pixels containing >300 photons in the Golgi area, when overexpressed alone (blue) or with tdTomato-LPAATδ under steady-state (red) or during traffic pulse (green) conditions. The average fluorescence lifetime of E0GFP–BARS were 2.08±0.07 ns (E0GFP–BARS alone), and 1.62±0.22 ns and 1.28±0.27 ns (with tdTomato-LPAATδ) under steady-state or during traffic pulse conditions, respectively. (e) Quantification of FLIM-FRET efficiency in the Golgi area under steady-state or during a traffic pulse conditions (as indicated). Data are mean±s.d. (n=10 cells per condition). ***P<0.005 (Student’s t-tests) versus steady state.
Discussion
The main finding of this study is that BARS induces fission of post-Golgi basolateral carriers by interacting directly with, and activating, the enzyme LPAATδ, a member of the acyltransferase family, which localizes at the TGN and converts LPA into PA. This indicates that the LPA–PA conversion plays a role in BARS-dependent fission. Notably, the metabolism of PA has been implicated in various aspects of membrane dynamics by other groups, albeit generally based on indirect evidence18,21,34,56,57,58.
Based on current and previous results, we propose the following working model for basolateral carrier formation. ARF initiates the process by recruiting and activating PI4KIIIβ at the Golgi. This produces a local increase in phosphatidylinositol 4-phosphate (PtdIns4P)59, which supports the budding of tubular carrier precursors, most likely through recruitment of PtdIns4P-binding proteins16. Carrier precursor budding is assisted by phospholipase A2 (PLA2) via production of positively curved lysolipids, including LPA, which facilitate the bending of membranes into tubules2,33,34,60,61. Concomitantly, BARS assembles with 14-3-3γ and PI4KIIIβ into a complex where 14-3-3γ binds to the BARS dimerization surface, and thus keeps BARS in a monomeric fission-competent conformation15,16. BARS then activates LPAATδ at the TGN and on the elongating tubular carrier precursors, where the PLA2-generated LPA is converted into PA by LPAATδ, leading to carrier fission, in a process that might be aided by the property of BARS itself and ARF to insert into lipid membranes21,62.
LPA and PA have biophysical properties that may be relevant for fission. PA has a highly charged headgroup close to the glycerol backbone, a tendency to form intramolecular and intermolecular hydrogen bonds and segregate into microdomains. Because PA can act as a binding site for proteins bearing amphipathic/hydrophobic surfaces56, which can lead to fission through the shallow hydrophobic insertion mechanism11, its above properties might cause PA-binding proteins to insert into membranes at the optimal depth and extent for causing fission. Also of note is that BARS itself21 has been reported to bind PA. One can thus hypothesize that a runaway process might take place at the tubular precursor surface by which BARS-bound LPAATδ converts LPA into PA, causing more BARS molecules to be recruited to the membrane and bind to LPAATδ, and hence more LPA to be converted into PA (similar to the positive-feedback Sec7–Arf1 loop whereby Sec7 activates Arf1 to recruit more Arf1 to activate more Sec7 and so on)63, until the local concentrations of these molecules become high enough to induce fission. In addition, under physiological conditions LPA and PA have strongly positive and negative spontaneous curvatures, respectively32; thus the conversion from LPA to PA may generate negatively curved PA microdomains within overall positively curved membrane areas, which might lead to membrane destabilization. And, finally, the formation of PA might lead to other potential fission-related lipid-based mechanisms, such as the enzymatic conversion of PA into diacylglycerol57,64,65.
The precise role of the lipid and protein players considered in this study must now be defined. Most of the key components involved in BARS-dependent fission are available in pure form, and the possibility to reconstitute this fission pathway in artificial membranes using known components appears to be within reach.
Methods
Plasmids and chemicals and recombinant proteins
Human LPAAT cDNAs were from ImaGenes GmbH (for subcloning and mutations, see Supplementary Table 1); tdTomato-C1 cloning vector was from Addgene; BARS-pCDNA3, BARS–yellow fluorescent protein (BARS–YFP), BARSS147A–YFP, BARSS147D–YFP and BARSD355A–YFP were prepared as previously described14,15,17; E0GFP was provided by L. Marchetti (Scuola Normale Superiore, Pisa, Italy); LDLRY18A–GFP was provided by R. Polishchuk (TIGEM, Naples, Italy); the HeLa hGH–GFP–FM-expressing cell line was provided by A. Peden (the University of Sheffield, Sheffield, UK). CI-976 was from Tocris Bioscience, tannic acid and BFA from Fluka, protease inhibitors as Complete Mini EDTA-free tablets from Roche, cycloheximide, Protein A Sepharose, anti-FLAG M2 affinity gel antibody beads and 3 × FLAG peptide from Sigma-Aldrich, AP21998 (D/D solubilizer) from Clontech, oleoyl-LPA from Avanti Polar Lipids, [14C]oleoyl-coenzyme A (specific activity, 60 mCi mmol−1) and dioleoyl [14C]-PA (specific activity, 140 mCi mmol−1) from PerkinElmer, and TRICH-labelled dextran, FITC-labelled dextran and MitoTracker Orange CM-H2TMRos from Molecular Probes. NAD+, BAC and HeLa (CD38+) cells were described previously28. Ni-NTA agarose and glutathione Sepharose beads were from Amersham, and Protein A Gold was from Cell Microscopy Center (the University Medical Center Utrecht, the Netherlands). Recombinant purified GST and GST-BARS proteins were produced from E. coli XL1Blue transformed cells with pGEX4T1 or pGEX4T1-BARS cells, respectively, grown at A600=0.4 before induction with 0.1 mM isopropyl-β-D-1-thiogalactopyranoside for 2 h at 37 °C (described in detail in ref. 55). Full-length and partial GST-LPAATδ (aa 152–251) purified by in vitro wheat germ expression system were from Antibodies-Online.com (catalogue number: ABIN1344584). His-EcLPAAT (His-plsC) purifed in vitro wheat germ expression system was from Cusabio (catalogue number: CSB-EP34083ENV). LPAATδ siRNAs (M-008620-00), LPAATγ siRNAs (M-009283-01) and BARS siRNAs (M-008609-02) were from Dharmacon.
Antibodies
All of the individually sourced antibodies for western blotting (WB), immunofluorescence and the LPAAT assay were obtained and used as detailed in ref. 15, unless otherwise stated. The rabbit polyclonal anti-LPAATδ (ab188002, 1:1,000) and anti-giantin (ab24586, 1:5,000) antibodies (for WB use) were from Abcam; the mouse polyclonal anti-LPAATδ antibody (for microinjection and LPAAT assay use) was from Abnova (H00056895-B01P); the rabbit polyclonal anti-LPAATδ antibody (1:50 for IF use) was from Sigma-Aldrich (SAB4502436), the anti-GM130 monoclonal antibody (1:200 for IF use) was from BD Transduction Laboratories (610823) and the rabbit polyclonal anti-Calnexin antibody (1:500 for WB use) was from Santa Cruz (AF18 sc23954). The anti-LPAATδ polyclonal antibodies from Abcam and Abnova recognize specifically LPAATδ and not other LPAATs by WB analysis (our unpublished data). Commercially available antibodies used: anti-TGN46 1:400 for IF use (Biorad, AHP500GT); anti-Giantin 1:500 for IF use (Alexis Corporation, 804-600-C100); anti-Flag 1:5,000 for WB and 1:500 for IF use (Sigma, F1804); anti-GAPDH 1:50,000 for WB use (ABD Serotec, MCA4740); anti-penta-His 1:1,000 for WB use (Life Technologies, P21315); anti-LPAATγ 1:1,000 for WB use (Abcam, ab93181). Source images from relevant WB are available in the Supplementary Figs 11–13.
Immunoprecipitations and pull-down assays
HeLa cells in 10-cm Petri dishes were transiently transfected with 7 μg of each DNA (BARS-pCDNA3 and LPAATs–Flag) using 42 μl TransIT-LT1 per dish. Twenty-four hours after transfection, the cells were washed three times with phosphate-buffered saline (PBS) and lysed using 1 ml lysis buffer/dish (25 mM Tris, pH 7.4, 150 mM NaCl, 5 mM EDTA, 5 mM MgCl2, 10 mM NaF, 40 mM β-glycerophosphate, 1 mM Na3VO4, 1 mM dithiothreitol) supplemented with 1% Triton X-100 and protease inhibitor mixture (30 min, 4 °C, shaking). The lysates were centrifuged (13,000g, 10 min, 4 °C), with the supernatants assayed for protein concentration (Bradford assay) and used fresh.
For BARS immunoprecipitation, 500 μg lysate protein from these HeLa cells was brought to 0.2% (v/v) Triton X-100 (final concentration), and incubated with 3 μg anti-BARS polyclonal antibody (overnight, 4 °C, shaking)55. Then 50 μl protein A Sepharose beads were added for a further 1 h of incubation (4 °C, shaking). For LPAAT immunoprecipitation, 1.2 mg lysate protein from the HeLa cells was brought to 0.2% (v/v) Triton X-100 (final concentration), and incubated with 40 μl anti-FLAG M2 affinity-gel-purified antibody (2 h, 4 °C, shaking). For BARS immunoprecipitation in the presence of EcLPAAT, 0.8 mg lysate protein from the COS7 cells was brought to 0.2% (v/v) Triton X-100 (final concentration) and incubated with 160 μg purified EcLPAAT (2 h, 4 °C, shaking) and then incubated with 3 μg anti-BARS polyclonal antibody (overnight, 4 °C, shaking). The immune complexes were collected by centrifugation (500g, 5 min, 4 °C). After three washes with lysis buffer with 0.2% Triton X-100, and twice with lysis buffer without Triton X-100, the bound protein was eluted from the protein A Sepharose beads or from anti-FLAG M2 affinity-gel-purified antibody by boiling (10 min) in 100 μl Laemmli buffer. Thirty micrograms of input and 70% of the eluted proteins were separated by 10% SDS–PAGE, and subjected to WB via transfer to nitrocellulose membranes (Millipore).
For histidine pull-down from LPAATδ–Flag expressing cell lysate, His-BARS (20 μg) was incubated for 3 h at 37 °C with buffer alone (20 mM Tris, pH 7.4, 10 mM sucrose) or with 120 μM HPLC-purified BAC, to allow binding of BAC to His-BARS. The reaction mixture was stopped on ice, and 1 mg lysate protein from LPAATδ–Flag expressing cells was incubated with each sample (2 h, 4 °C, shaking). Then, 30 μl Ni-NTA agarose beads were added, and the samples were incubated (1 h, 4 °C, shaking). The beads were then washed three times with lysis buffer at pH 8.0 supplemented with 0.2% (v/v) Triton X-100 and 20 mM imidazole, by centrifugation (700g, 5 min), and then twice with lysis buffer at pH 8.0 without Triton X-100 but supplemented with 20 mM imidazole. The bound protein was eluted from the Ni-NTA agarose beads by boiling (10 min) in 100 μl Laemmli buffer. Twenty micrograms of input and 50% of the eluted proteins were separated by 10% SDS–PAGE, and subjected to WB via transfer to nitrocellulose membranes.
For GST pull-down with LPAATδ, 3 μg of His-BARS were incubated with 1.5 μg GST as control or with 4 μg GST-LPAATδ full length or with 3 μg GST-LPAATδ (aa 152–251) in GST incubation buffer (50 mM Tris, pH 8.0, 100 mM KCl, 0.2% Triton X-100, protease inhibitors; 2 h, 4 °C, shaking). Then, 30 μl glutathione sepharose beads were added for a further incubation (1 h, 4 °C, shaking). The beads were then washed five times with GST incubation buffer, by centrifugation (500g, 5 min). The bound protein was eluted from the glutathione sepharose beads with GST elution buffer (100 mM Tris, pH 8.0, 20 mM glutathione, 5 mM dithiothreitol). The 10% and 50% of the unbound and eluted proteins, respectively, were separated by 10% SDS–PAGE, and subjected to WB via transfer to nitrocellulose membranes.
For GST pull-down with EcLPAAT, 3 μg of EcLPAAT were incubated with 3 μg GST as control or with 5 μg GST-BARS in GST incubation buffer (20 mM Tris, pH 8.0, 1 mM EDTA, 0.2% Triton X-100, 100 mM KCl) (overnight, 4 °C, shaking). Then, 30 μl glutathione sepharose beads were added for a further incubation (1 h, 4 °C, shaking). The beads were then washed five times with GST incubation buffer, by centrifugation (500g, 5 min). The bound protein was eluted from the glutathione sepharose beads with GST elution buffer (100 mM Tris, pH 8.0, 20 mM glutathione, 5 mM dithiothreitol). Fifty per cent of the eluted proteins were separated by 10% SDS–PAGE, and subjected to WB via transfer to nitrocellulose membranes. For the GST pull-down with BAC-treated BARS, 5 μg GST-BARS was initially incubated with buffer alone (20 mM Tris, pH 7.4, 10 mM sucrose) or with 120 μM HPLC-purified BAC28 (3 h, 37 °C), to allow binding of BAC to GST-BARS in the GST incubation buffer. For the GST pull-down with NAD-treated BARS, 5 μg GST-BARS was initially incubated with 100 μM NAD+ in GST incubation buffer (1 h, room temperature). Full-scan images of all WB data are reported in Supplementary Figs 11–13.
Transport protocols and light and wide-field microscopy
For the TGN-exit assay of VSVG, the cells were transfected with VSVG–GFP cDNA or infected with VSV, then incubated for 2 h at 40 °C, followed by 2 h at 20 °C (with 100 μg ml−1 cycloheximide) to accumulate VSVG in the Golgi complex. For the TGN-exit assay of p75, the cells were transfected with p75–GFP cDNA, incubated overnight in complete medium and for 2 h at 20 °C (with 100 μg ml−1 cycloheximide). To quantify VSVG-containing carriers, the TGN-exit assay was carried out in the presence of 0.5% tannic acid that inhibits fusion of the mature carriers to the plasma membrane and therefore leads to the accumulation of these carriers near the cell surface. The cells were then fixed and labelled with the P5D4 anti-VSVG antibody and the anti-TGN46 antibody. The fluorescent spots (0.5–2.0 μm) stained by both antibodies (at least 80% of VSVG-containing carriers) were then counted. The p75–GFP-containing carriers were counted as above, although without the co-staining for TGN46. All values are means±s.d. from three independent experiments. For the COPI transport assay, the cells were transfected with VSVG-ts045-KDELR-myc and then equilibrated at the permissive temperature of 32 °C for 3 h with the subsequent redistribution of the misfolded VSVG-KDELR chimera into the ER by shifting to the non-permissive temperature of 40 °C. The cells were immunostained with anti-VSVG antibody and the percentage of the cells with the ER staining was quantifed19. For the transport of the endocytosis-defective LDL–GFP receptor (LDLRY18A), the cells were transfected with LDLRY18A–GFP constructs and then equilibrated 2 h at 20 °C to accumulate the protein in the TGN with the subsequent shifting to 37 °C to allow for synchronized release of the proteins from the TGN66. For the hGH–GFP–FM transport, the HeLa cells stably transfected with hGH-FM–GFP were treated with the DD-solubilizer at 37 °C to induce the release of the protein from the ER to the PM48. The CI-976 treatment was performed during the VSVG TGN-exit assay, at 50 μM for 10 min, before the 32 °C temperature-release block and during the 32 °C temperature-release block. The anti-LPAATδ antibody (1 μg ml−1) was microinjected 1 h after the beginning of the 20 °C incubation in the VSVG transport assay, and after 1 h of recovery the cells were then processed for wide-field microscopy. Wide-field microscopy was performed as described previously15, with some modifications. COS7 cells were transfected with siRNAs for LPAATδ (Smart Pool, Lipofectamine 2000), and after 48 h the cells were transfected with VSVG–cyan fluorescent protein (VSVG–CFP; overnight, 40 °C) and then incubated with 100 μg ml−1 cycloheximide (3 h, 20 °C). The cells were then shifted to 32 °C (with continued cycloheximide), and followed by fast video microscopy (see ref 15). For CI-976 treatment, COS7 cells were treated with 50 μM CI-976 for 10 min before the shift to 32 °C.
FLIM measurements
The DNA coding for E0GFP–BARS and LPAATδ-tdTomato were generated using the Restriction Free (RF) cloning procedure67 using the following two pairs of primers: 5′-cacgctggccggagagccttatgtcaggcgtccgacct-3′ and 5′-tttaaacgggccctctagagcactacaactggtcagtcgta-3′; and 5′-ctaccggactcagatctcgagatggacctcgcgggac-3′ and 5′-cccttgctcaccatggtggcctgattcagtttctggttg-3′, respectively. BARS and LPAATδ genes were amplified as follows: a single denaturation step (95 °C, 5 min), followed by 35 cycles PCR (95 °C, 1 min; 50 °C, 30 s; 72 °C, 2 min) and a final elongation step of 5 min (72 °C), in a final volume of 50 μl (including 100 ng of template DNA, 0.4 μM of each primer, 200 μM of each dNTPs, 1 × Pfu buffer and 2 U Pfu Turbo Cx hotstart DNA polymerase, Agilent). Then the amplified genes were purified using the Agilent Strataprep PCR purification kit and used as mega-primer in the second PCR for gene cloning. LPAATδ was cloned into tdTomato-N1 vector, fused to the N-terminal of tdTomato sequence, and BARS was cloned into pcDNA3.1+ vector carrying the E0GFP sequence68 fused to the C-terminal of E0GFP sequence. The RF reactions were carried out as follows: a single denaturation step (95 °C, 5 min) was performed followed by 20 cycles PCR (95 °C, 1 min; 50 °C, 30 s; 72 °C, 7 min) and a final elongation step of 10 min at 72 °C. RF reactions were performed in a final volume of 50 μl (including 50 ng of target DNA, 300 ng of PCR mega-primer products, 200 μM of each dNTPs, 1 × Pfu buffer and 5 U Pfu Turbo Cx hotstart DNA polymerase). Thirty microlitre of each PCR mix were digested using 20 U DpnI (37 °C, 2 h), and 10 μl of each aliquot were used to transform E. coli Top10 strain (Invitrogen). Colony PCR screening were used to search positive clones by using specific primers for BARS and LPAATδ. The cloned fragments were then completely sequenced and verified.
COS7 cells were transiently transfected with 0.4 μg E0GFP–BARS and 0.4 μg LPAATδ-tdTomato using TransIT-LT1 Transfection Reagent, according to manufacturer’s instructions. Sixteen hours after transfection, the cells were subjected to the VSVG TGN-exit assay. After 10 min of the 32 °C temperature-release block, the cells were fixed with 4% (w/v) PFA in 20 mM Hepes pH 7.4, for 10 min at room temperature. The samples were then treated with 100 mM glycine in 20 mM Hepes for 30 min, washed three times with 20 mM Hepes and mounted in glycerol/20 mM Hepes (1:1).
The lifetime values were extracted from the fitting of decay curves obtained from the donor emission (E0GFP–BARS) on the Golgi area, and global and pixel kinetic data analyses are computed by a commercially available software package SymPho Time, Version 5.3.2.2 (PicoQuant GmbH, Berlin, Germany). Time-resolved images were acquired by the confocal microscope Leica SP5 II equipped with the White Light Laser (both Leica Microsystems GmbH, Mannheim, Germany) and the TCSPC measurements were performed with the SMD Upgrade Kit (PicoQuant GmbH). The excitation light was pulsed at 80 MHz and the donor fluorophore was excited at 475 nm and acquired from 495 to 530 nm. The measurements were carried out in fixed cells, for ease and speed of acquisition of a statistically reliable number of samples. The donor lifetime values were extracted from the fitting of decay curves obtained from the donor emission (E0GFP–BARS) in the Golgi area, and both global and pixel kinetic data analyses were computed by the commercially available software package SymPho Time, Version 5.3.2.2. Changes in donor lifetime due to FRET were assessed by comparing the donor lifetime in cells expressing only the donor with the lifetime in cells co-transfected with donor and acceptor and fixed either under steady-state condition or during a traffic pulse.
Cryo-immunogold electron microscopy
HeLa cells were transiently transfected with 8 μg plasmid DNA encoding Flag-LPAATδ for 24 h (using TransIT-LT1) and then fixed with 2% formaldehyde and 0.2% glutaraldehyde in 100 mM Phosphate buffer pH 7.4. The cells were pelleted by centrifugation, embedded in 12% gelatin, cooled on ice and cut into 1-mm3 cubes at 4 °C. The cubes were immersed in 2.3 M sucrose at 4 °C overnight, frozen in liquid nitrogen and cut using Leica EM FC7 ultramicrotome69. Thin sections (50 nm) were picked up in a mix of 2% methylcellulose and 2.3 M sucrose (1:1) and incubated with the rabbit anti-LPAATδ (1:10) and the mouse anti-Golgin-97 (Life Technologies, catalogue number: A21270, 1:50) antibodies. Each of these incubations was followed by incubations with Protein A gold; 10 nm Gold particles for anti-LPAATδ; and 15 nm Gold particles for anti-Golgin-97. After labelling, the sections were treated with 1% glutaraldehyde and embedded in methylcellulose uranyl acetate. Electron microscopy images were acquired using FEI Tecnai-12 electron microscope.
Transfections with siRNAs
COS7 and HeLa cells were transfected with a non-targeting siRNA or with 150 nm of a Smart Pool of LPAATδ/M-009283 or LPAATγ/M-008620 siRNAs, for 72 h (except for BARS and 14-3-3s siRNAs, where 100 nm of a Smart Pool was used for 48 h, as previously described in ref. 15) using Lipofectamine 2000, according to manufacturer’s instructions. The efficiency of interference was assessed by WB. The treatment with Smart Pool siRNAs for LPAATγ (M-008620) and for LPAATδ (M-009283) specifically reduces the endogenous protein levels of LPAATγ and LPAATδ (by WB), respectively, without affecting the levels of other tested LPAATs (our unpublished data). Alternatively, COS7 cells were transfected with the siRNAs (as above) in combination for the last 16 h with VSVG–CFP, VSVG–GFP, VSVG-ts045-KDELR-myc, LDLRY18A–GFP or p75–GFP, and then subjected to the specified Golgi-transport assay14,15,66. For the rescue experiments, COS7 cells were transfected with siRNAs for LPAATδ/D-009283-03 (5′-GCACACGGUUCACGGAGAA-3′, Dharmacon) for 48 h, and transfected for a further 24 h (using TransIT-LT1) with Flag-LPAATδwt or Flag-LPAATδH96V (both encode an siRNA-resistant silent mutation), followed by infection with VSV for the TGN-exit assay.
In vitro acyltransferase assay
HeLa cells (1 × 106) in 10-cm Petri dishes were transiently transfected with 8 μg plasmid DNA encoding Flag-LPAATδwt or Flag-LPAATδH96V for 48 h (using TransIT-LT1). Alternatively, the HeLa cells were transfected with siRNAs (as above) in combination with Flag-LPAATδwt for 48 h (using Lipofectamine 2000). The cells were washed three times with PBS, harvested as 250 μl per dish in homogenization buffer (100 mM Tris, pH 7.4, 5 mM NaCl, 3 mM MgCl2) supplemented with the protease inhibitor mixture, and homogenized (6 pulses, 30% amplitude; Branson Digital Sonifier). The lysate was centrifuged at 600g for 10 min at 4 °C, and the post-nuclear supernatant fraction was used in the acyltransferase assay.
Golgi membranes from HeLa cells treated as described above were obtained as described previously70, with some modifications. HeLa cells were washed with PBS, harvested by trypsinization and pelleted (400g, 5 min, 4 °C). The cell pellets were resuspended in 1:4 vol. homogenization buffer (250 mM sucrose, 100 mM Tris, pH 7.4) supplemented with the protease inhibitor mixture. The cells were then passed six times each direction (12 passes total) through the Balch homogenizer with a 7.988 mm diameter tungsten-carbide ball bearing (clearance, 12 μm) using constant manual pressure. Following homogenization, the samples (≈ 850 μl in 0.25 M sucrose, 10 mM Tris-HCl, pH 7.4) were adjusted to 1.4 M sucrose by addition of ice-cold 2.3 M sucrose containing 10 mM Tris-HCl (pH 7.4). Then, 1 mM Na2EDTA was added from a 100 mM stock solution, and the samples were vortexed vigorously to ensure uniform mixing. The suspension (≈ 2.8 ml) was layered onto sucrose gradients (700 μl 2.0 M and 1.4 ml 1.6 M sucrose, from the bottom of the tubes for a swing-out rotor) and covered with 4.2 ml 1.2 M and 2.8 ml 0.8 M sucrose, from the suspension fraction. After centrifugation (90,000g, 2.5 h, 4 °C), the Golgi-enriched fraction was collected at the 0.8/1.2 M sucrose interface, aliquoted, frozen in liquid nitrogen and stored at −80 °C.
Two micrograms of the post-nuclear supernatant or of the purified Golgi membranes fractions were incubated with the LPAAT reaction buffer (75 mM Tris, pH 7.4, 4 mM MgCl2, 1 mM dithiothreitol, 4 mM NaF, 1 mg ml−1 bovine serum albumin fatty acid free, 50 μM oleoyl-LPA, 20 μM [14C]oleoyl-CoA) in a final volume of 100 μl, for 20 min at 25 °C. The total lipids were extracted by adding 450 μl cold CHCl3/CH3OH (2:1, v/v). After 30 min on ice, the samples were centrifuged (10,000g, 5 min). The lower, organic, phase was dried under a stream of N2, resuspended in 50 μl CHCl3, and loaded onto an oxalate‐pretreated TLC plate55. The lipids were separated by running the TLC plates with CHCl3/CH3OH/33% NH4OH/H2O (54:42:2.9:9.1; v/v/v/v). The radiolabelled spots were quantified by gas ionization counting (Beta-Imager Systems, Biospace Laboratories). Dioleoyl [14C]-PA was used as a standard.
For CI-976 treatment, the post-nuclear fraction from HeLa cells was incubated with 50 μM CI-976 for 30 min at 25 °C, followed by the addition of the LPAAT reaction buffer (as above). For anti-LPAATδ or anti-BARS antibody treatment, the post-nuclear or Golgi membrane fraction from HeLa cells was incubated with 100 ng anti-LPAATδ or anti-BARS affinity-purified polyclonal antibody for 30 min at 25 °C, followed by addition of the LPAAT reaction buffer (as above). For immunopurified BARS treatment, the post-nuclear fraction from HeLa cells was incubated with 500 ng immunoprecipitated BARS purified from rat-brain cytosol with anti-BARS-IgG crosslinked matrice, as described in ref. 15 for 30 min at 25 °C, followed by addition of the LPAAT reaction buffer (as above).
In these experiments, LPAATδ-dependent activity (or LPAATδ activity) is defined as the activity value of LPAATδ-overexpressing extracts minus the activity value of LPAATδ depleted (or antibody-treated) extracts (see also Fig. 5a). In Figs 5, 6, 7 and Supplementary Fig. 10, the LPAATδ-independent activity (that is, derived from LPAATδ-depleted or antibody-treated extracts) is indicated with a dashed line. The LPAATδ-independent activity was reproducibly ≈50% of the total activity in LPAATδ-overexpressing extracts (as evaluated in >30 independent experiments) and ≈30% of the total activity in purified Golgi membranes from LPAATδ-overexpressing cells (as evaluated in >20 independent experiments).
Size-exclusion chromatography
For the fast protein liquid chromatography, 600 μg purified recombinant GST or GST-BARS was applied to a Sephacryl S-200 High Resolution HiPrep 16/60 (Amersham Pharmacia) gel filtration column equilibrated with PBS buffer (4 °C; flow rate, 0.3 ml min−1), with 1 ml fractions collected using an AKTA Fast Protein Liquid Chromatography (FPLC) system. The eluted protein was detected by monitoring absorbance at 280 nm, and 10 μl of the collected fractions was separated on 10% SDS–PAGE gels, and analysed by silver staining. Eighty microlitres of each of these fractions was then subjected to the LPAAT assay.
Ion-exchange chromatography
For the fast protein liquid chromatography, 600 μg purified recombinant GST-BARS was applied to a MonoQ column (HR5/5 Pharmacia LKB) equilibrated with buffer A (25 mM Tris, pH 8.00, 50 mM KCl; 4 °C; flow rate, 1 ml min−1), with 0.5 ml fractions collected using an AKTA FPLC system. After washing the MonoQ column with buffer A, the protein was eluted with a 20 ml gradient of 0.05–1 M KCl, and 10 μl of the collected fractions were separated on 10% SDS–PAGE gels and analysed by silver staining. Eighty microlitres of each of these fractions was then subjected to the LPAAT assay.
Statistical analysis
Two-tailed Student’s t-tests were applied to the data. Significance is indicated as *P<0.05, **P<0.01 and ***P<0.005.
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information files or are available from the corresponding authors upon request.
Additional information
How to cite this article: Pagliuso, A. et al. Golgi membrane fission requires the CtBP1-S/BARS-induced activation of lysophosphatidic acid acyltransferase δ. Nat. Commun. 7:12148 doi: 10.1038/ncomms12148 (2016).
References
Kozlov, M. M., McMahon, H. T. & Chernomordik, L. V. Protein-driven membrane stresses in fusion and fission. Trends Biochem. Sci. 35, 699–706 (2010).
Campelo, F. & Malhotra, V. Membrane fission: the biogenesis of transport carriers. Annu. Rev. Biochem. 81, 407–427 (2012).
Johannes, L. & Mayor, S. Induced domain formation in endocytic invagination, lipid sorting, and scission. Cell 142, 507–510 (2010).
Frolov, V. A., Escalada, A., Akimov, S. A. & Shnyrova, A. V. Geometry of membrane fission. Chem. Phys. Lipids 185, 129–140 (2015).
Weigert, R. et al. CtBP/BARS induces fission of Golgi membranes by acylating lysophosphatidic acid. Nature 402, 429–433 (1999).
Roux, A. Reaching a consensus on the mechanism of dynamin? F1000Prime Rep. 6, 86 (2014).
Ferguson, S. M. & De Camilli, P. Dynamin, a membrane-remodelling GTPase. Nat. Rev. Mol. Cell Biol. 13, 75–88 (2012).
Schmid, S. L. & Frolov, V. A. Dynamin: functional design of a membrane fission catalyst. Annu. Rev. Cell Dev. Biol. 27, 79–105 (2011).
McMahon, H. T. & Boucrot, E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat. Rev. Mol. Cell Biol. 12, 517–533 (2011).
Daumke, O., Roux, A. & Haucke, V. BAR domain scaffolds in dynamin-mediated membrane fission. Cell 156, 882–892 (2014).
Boucrot, E. et al. Membrane fission is promoted by insertion of amphipathic helices and is restricted by crescent BAR domains. Cell 149, 124–136 (2012).
Lee, M. C. et al. Sar1p N-terminal helix initiates membrane curvature and completes the fission of a COPII vesicle. Cell 122, 605–617 (2005).
Lenz, M., Morlot, S. & Roux, A. Mechanical requirements for membrane fission: common facts from various examples. FEBS Lett. 583, 3839–3846 (2009).
Bonazzi, M. et al. CtBP3/BARS drives membrane fission in dynamin-independent transport pathways. Nat. Cell Biol. 7, 570–580 (2005).
Valente, C. et al. A 14-3-γ dimer-based scaffold bridges CtBP1-S/BARS to PI(4)KIIIβ to regulate post-Golgi carrier formation. Nat. Cell Biol. 14, 343–354 (2012).
Valente, C., Luini, A. & Corda, D. Components of the CtBP1/BARS-dependent fission machinery. Histochem. Cell Biol. 140, 407–421 (2013).
Liberali, P. et al. The closure of Pak1-dependent macropinosomes requires the phosphorylation of CtBP1/BARS. EMBO J. 27, 970–981 (2008).
Haga, Y., Miwa, N., Jahangeer, S., Okada, T. & Nakamura, S. CtBP1/BARS is an activator of phospholipase D1 necessary for agonist-induced macropinocytosis. EMBO J. 28, 1197–1207 (2009).
Yang, J. S. et al. A role for BARS at the fission step of COPI vesicle formation from Golgi membrane. EMBO J. 24, 4133–4143 (2005).
Yang, J. S. et al. Key components of the fission machinery are interchangeable. Nat. Cell Biol. 8, 1376–1382 (2006).
Yang, J. S. et al. A role for phosphatidic acid in COPI vesicle fission yields insights into Golgi maintenance. Nat. Cell Biol. 10, 1146–1153 (2008).
Hidalgo Carcedo, C. et al. Mitotic Golgi partitioning is driven by the membrane-fissioning protein CtBP3/BARS. Science 305, 93–96 (2004).
Colanzi, A. et al. The Golgi mitotic checkpoint is controlled by BARS-dependent fission of the Golgi ribbon into separate stacks in G2. EMBO J. 26, 2465–2476 (2007).
Corda, D., Colanzi, A. & Luini, A. The multiple activities of CtBP/BARS proteins: the Golgi view. Trends Cell Biol. 16, 167–173 (2006).
Kumar, V. et al. Transcription corepressor CtBP is an NAD(+)-regulated dehydrogenase. Mol. Cell 10, 857–869 (2002).
Nardini, M. et al. CtBP/BARS: a dual-function protein involved in transcription co-repression and Golgi membrane fission. EMBO J. 22, 3122–3130 (2003).
Nardini, M. et al. CtBP1/BARS Gly172-->Glu mutant structure: impairing NAD(H)-binding and dimerization. Biochem. Biophys. Res. Commun. 381, 70–74 (2009).
Colanzi, A. et al. Molecular mechanism and functional role of brefeldin A-mediated ADP-ribosylation of CtBP1/BARS. Proc. Natl Acad. Sci. USA 110, 9794–9799 (2013).
Gallop, J. L., Butler, P. J. & McMahon, H. T. Endophilin and CtBP/BARS are not acyl transferases in endocytosis or Golgi fission. Nature 438, 675–678 (2005).
Yamashita, A. et al. Glycerophosphate/acylglycerophosphate acyltransferases. Biology 3, 801–830 (2014).
Shindou, H., Hishikawa, D., Harayama, T., Eto, M. & Shimizu, T. Generation of membrane diversity by lysophospholipid acyltransferases. J. Biochem. 154, 21–28 (2013).
Kooijman, E. E. & Burger, K. N. Biophysics and function of phosphatidic acid: a molecular perspective. Biochim. Biophys. Acta 1791, 881–888 (2009).
Schmidt, J. A. & Brown, W. J. Lysophosphatidic acid acyltransferase 3 regulates Golgi complex structure and function. J. Cell Biol. 186, 211–218 (2009).
Yang, J. S. et al. COPI acts in both vesicular and tubular transport. Nat. Cell Biol. 13, 996–1003 (2011).
Bradley, R. M. et al. Acylglycerophosphate acyltransferase 4 (AGPAT4) is a mitochondrial lysophosphatidic acid acyltransferase that regulates brain phosphatidylcholine, phosphatidylethanolamine, and phosphatidylinositol levels. Biochim. Biophys. Acta 1851, 1566–1576 (2015).
Marchi, S., Patergnani, S. & Pinton, P. The endoplasmic reticulum-mitochondria connection: one touch, multiple functions. Biochim. Biophys. Acta 1837, 461–469 (2014).
Kuroda, R. et al. Charged amino acids at the carboxyl-terminal portions determine the intracellular locations of two isoforms of cytochrome b(5). J. Biol. Chem. 273, 31097–31102 (1998).
Tamir, S. et al. Nutrient-deprivation autophagy factor-1 (NAF-1): biochemical properties of a novel cellular target for anti-diabetic drugs. PLoS ONE 8, e61202 (2013).
Borgese, N., Colombo, S. & Pedrazzini, E. The tale of tail-anchored proteins: coming from the cytosol and looking for a membrane. J. Cell Biol. 161, 1013–1019 (2003).
Coleman, J. Characterization of the Escherichia coli gene for 1-acyl-sn-glycerol-3-phosphate acyltransferase (plsC). Mol. Gen. Genet. 232, 295–303 (1992).
Ovadì, J. & Srere, P. A. Macromolecular compartmentation and channeling. Int. Rev. Cytol. 192, 255–280 (2000).
Mironov, A. A. et al. Small cargo proteins and large aggregates can traverse the Golgi by a common mechanism without leaving the lumen of cisternae. J. Cell Biol. 155, 1225–1238 (2001).
Polishchuk, R., Di Pentima, A. & Lippincott-Schwartz, J. Delivery of raft-associated, GPI-anchored proteins to the apical surface of polarized MDCK cells by a transcytotic pathway. Nat. Cell Biol. 6, 297–307 (2004).
Drecktrah, D. et al. Inhibition of a Golgi complex lysophospholipid acyltransferase induces membrane tubule formation and retrograde trafficking. Mol. Biol. Cell 14, 3459–3469 (2003).
Cole, N. B., Ellenberg, J., Song, J., DiEuliis, D. & Lippincott-Schwartz, J. Retrograde transport of Golgi-localized proteins to the ER. J. Cell Biol. 140, 1–15 (1998).
Yeaman, C. et al. The O-glycosylated stalk domain is required for apical sorting of neurotrophin receptors in polarized MDCK cells. J. Cell Biol. 139, 929–940 (1997).
Matter, K., Hunziker, W. & Mellman, I. Basolateral sorting of LDL receptor in MDCK cells: the cytoplasmic domain contains two tyrosine-dependent targeting determinants. Cell 71, 741–753 (1992).
Cancino, J. et al. Control systems of membrane transport at the interface between the endoplasmic reticulum and the Golgi. Dev. Cell 30, 280–294 (2014).
Eto, M., Shindou, H. & Shimizu, T. A novel lysophosphatidic acid acyltransferase enzyme (LPAAT4) with a possible role for incorporating docosahexaenoic acid into brain glycerophospholipids. Biochem. Biophys. Res. Commun. 443, 718–724 (2014).
Chen, Y. Q. et al. AGPAT6 is a novel microsomal glycerol-3-phosphate acyltransferase. J. Biol. Chem. 283, 10048–10057 (2008).
Chambers, K., Judson, B. & Brown, W. J. A unique lysophospholipid acyltransferase (LPAT) antagonist, CI-976, affects secretory and endocytic membrane trafficking pathways. J. Cell Sci. 118, 3061–3071 (2005).
Leung, D. W. The structure and functions of human lysophosphatidic acid acyltransferases. Front. Biosci. 6, D944–D953 (2001).
Yuki, K., Shindou, H., Hishikawa, D. & Shimizu, T. Characterization of mouse lysophosphatidic acid acyltransferase 3: an enzyme with dual functions in the testis. J. Lipid Res. 50, 860–869 (2009).
Lewin, T. M., Wang, P. & Coleman, R. A. Analysis of amino acid motifs diagnostic for the sn-glycerol-3-phosphate acyltransferase reaction. Biochemistry 38, 5764–5771 (1999).
Valente, C., Spano, S., Luini, A. & Corda, D. Purification and functional properties of the membrane fissioning protein CtBP3/BARS. Methods Enzymol. 404, 296–316 (2005).
Stace, C. L. & Ktistakis, N. T. Phosphatidic acid- and phosphatidylserine-binding proteins. Biochim. Biophys. Acta 1761, 913–926 (2006).
Baron, C. L. & Malhotra, V. Role of diacylglycerol in PKD recruitment to the TGN and protein transport to the plasma membrane. Science 295, 325–328 (2002).
Martinez-Martinez, N., Martinez-Alonso, E., Ballesta, J. & Martinez-Menarguez, J. A. Phospholipase D2 is involved in the formation of Golgi tubules and ArfGAP1 recruitment. PLoS ONE 9, e111685 (2014).
Godi, A. et al. ARF mediates recruitment of PtdIns-4-OH kinase-beta and stimulates synthesis of PtdIns(4,5)P2 on the Golgi complex. Nat. Cell Biol. 1, 280–287 (1999).
San Pietro, E. et al. Group IV phospholipase A(2)alpha controls the formation of inter-cisternal continuities involved in intra-Golgi transport. PLoS Biol. 7, e1000194 (2009).
Ha, K. D., Clarke, B. A. & Brown, W. J. Regulation of the Golgi complex by phospholipid remodeling enzymes. Biochim. Biophys. Acta 1821, 1078–1088 (2010).
Antonny, B., Beraud-Dufour, S., Chardin, P. & Chabre, M. N-terminal hydrophobic residues of the G-protein ADP-rybosylation factor-1 insert into membrane phospholipids upon GDP exchange. Biochemistry 15, 4675–4684 (1997).
Richardson, B. C., McDonold, C. M. & Fromme, J. C. The Sec7 Arf-GEF is recruited to the trans-Golgi network by positive feedback. Dev. Cell 22, 799–810 (2012).
Fernandez-Ulibarri, I. et al. Diacylglycerol is required for the formation of COPI vesicles in the Golgi-to-ER transport pathway. Mol. Biol. Cell 18, 3250–3263 (2007).
Freyberg, Z., Siddhanta, A. & Shields, D. "Slip, sliding away": phospholipase D and the Golgi apparatus. Trends Cell Biol. 13, 540–546 (2003).
Giannotta, M. et al. The KDEL receptor couples to Galphaq/11 to activate Src kinases and regulate transport through the Golgi. EMBO J. 31, 2869–2881 (2012).
Unger, T., Jacobovitch, Y., Dantes, A., Bernheim, R. & Peleg, Y. Applications of the Restriction Free (RF) cloning procedure for molecular manipulations and protein expression. J. Struct. Biol. 172, 34–44 (2010).
Marchetti, L. et al. Homeotic proteins participate in the function of human-DNA replication origins. Nucleic Acids Res. 38, 8105–8119 (2010).
Peters, J. P., Bos, E. & Griekspoor, A. Current Protocols in Cell Biology. Ch. 4.7, (John Wiley & Sons, (2006).
Balch, W. E., Dunphy, W. G., Braell, W. A. & Rothman, J. E. Reconstitution of the transport of protein between successive compartments of the Golgi measured by the coupled incorporation of N-acetylglucosamine. Cell 39, 405–416 (1984).
Acknowledgements
We would like to thank all colleagues who kindly provided us with antibodies and reagents (as listed under ‘Reagents’); Dr D. Barneda for very preliminary experiments, Dr C.P. Berrie for editorial assistance and Dr R.E. Duncan for constructs and fruitful discussions; the BioImaging Facility at the Institute of Protein Biochemistry and the Laboratory of Ultrastructure at the Italian Institute of Technology of Naples, for support in imaging microscopy, data processing and analysis; the Italian Association for Cancer Research (to D. Corda IG10341 and 14675, to A.L. IG15767), projects PON01-00117 and PON01-00862 from MIUR, PNR-CNR Aging Program 2012–2014, NanoMax, FaReBio di Qualità and ‘Creme Campania Research in Experimental Medicine Project’ POR Campania FSE 2007/2013, POR project OcKey, for financial support. A.P. and C.V. were recipients of Italian Foundation for Cancer Research Fellowships (FIRC, Milan, Italy).
Author information
Authors and Affiliations
Contributions
A.P. and C.V. designed, carried out and analysed all of the experiments. L.L.G., A.F. and G.L. carried out immunofluorescence, immunoprecipitation and pull-down experiments. D. Circolo carried out in vitro acyltransferase assays and immunofluorescence experiments. G.T. carried out in vitro acyltransferase assays, immunofluorescence and electron microscopy experiments. V.M.M. conceived and designed FLIM experiments and provided advice on data analysis. C.V. and F.F. carried out, with V.M.M., FLIM experiments. L.M. designed the strategy for constructs subcloning, purification and testing of them for FLIM experiments. M.A.Z. with C.V and A.F. have substantially contributed to the study of protein localization and commented on the manuscript. R.S.P. carried out time-lapse microscopy. C.V., D. Corda and A.L. conceived and supervised the project, discussed and analysed the data and co-wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-13 and Supplementary Table 1 (PDF 48874 kb)
Supplementary Movie 1
Export of VSVG-GFP from the Golgi complex in COS7 cells following non-targeting siRNAs. Following 48 h of non-targeting siRNAs treatment, COS7 cells were transfected for 24 h with VSVG-GFP, subjected to the TGN-exit assay, and observed at 32°C in vivo under confocal microscopy. Several VSVG-GFP- containing carriers can be seen to be formed from the Golgi complex and to move across the cell toward the plasma membrane. (MOV 2275 kb)
Supplementary Movie 2
Export of VSVG-GFP from the Golgi complex in COS7 cells following LPAATd-targeting siRNAs. Following 48 h of LPAATd-targeting siRNAs treatment, COS7 cells were transfected for 24 h with VSVG-GFP, subjected to the TGN-exit assay, and observed at 32°C in vivo under confocal microscopy. Several post-Golgi carrier precursors can be seen to extend from the Golgi complex, but they do not undergo fission, resulting in long tubular carrier precursors. The arrowheads indicate some VSVG-GFP-containing carriers with aberrantly extended tubular shapes. The arrow indicates a carrier that after fission, moves towards, and fuses with, the plasma membrane. (MOV 1243 kb)
Supplementary Movie 3
Post-Golgi carrier formation in VSVG-GFP expressing COS7 cells following anti-LPAATd antibody injection. VSVG-GFP-expressing COS7 cells were subjected to the TGN-exit assay, and after 1 h at 20°C the cells were microinjected with an anti-LPAATd antibody and incubated for a further 1 h at 20°C. The cells were then observed at 32°C in vivo under confocal microscopy. The microinjected cell shows long tubular carrier precursors (top right: indicated by the arrowhead; see also Supplementary Fig. 5). (MOV 6091 kb)
Supplementary Movie 4
Post-Golgi carrier formation in VSVG-GFP-expressing COS7 cells following CI-976 treatment. VSVG-GFP-expressing COS7 cells were subjected to the TGN-exit assay and treated with the general LPAAT inhibitor CI-976 (50 μM, 15 min) before the 32°C temperature-block release. The cells were then observed in vivo under confocal microscopy. The CI-976 treatment dramatically reduces the fission of post-Golgi tubular carrier precursors, and increases the lengths of the fissioned postGolgi carriers. (MOV 5022 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Pagliuso, A., Valente, C., Giordano, L. et al. Golgi membrane fission requires the CtBP1-S/BARS-induced activation of lysophosphatidic acid acyltransferase δ. Nat Commun 7, 12148 (2016). https://doi.org/10.1038/ncomms12148
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms12148
This article is cited by
-
The transcriptional corepressor CtBP2 serves as a metabolite sensor orchestrating hepatic glucose and lipid homeostasis
Nature Communications (2021)
-
c-MYC Triggers Lipid Remodelling During Early Somatic Cell Reprogramming to Pluripotency
Stem Cell Reviews and Reports (2021)
-
Phosphatidylinositol 4-kinase IIIβ mediates contraction-induced GLUT4 translocation and shows its anti-diabetic action in cardiomyocytes
Cellular and Molecular Life Sciences (2021)
-
Convergent use of phosphatidic acid for hepatitis C virus and SARS-CoV-2 replication organelle formation
Nature Communications (2021)
-
Coupling fission and exit of RAB6 vesicles at Golgi hotspots through kinesin-myosin interactions
Nature Communications (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.