Abstract
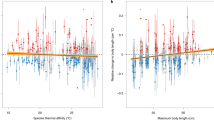
Climate change can affect organisms both directly, by affecting their physiology, growth, and behaviour1, and indirectly, for example through effects on ecosystem structure and function1,2. For ectotherms, or ‘cold-blooded’ animals, warming will directly affect their metabolism, with growth rates in temperate species predicted to increase initially as temperatures rise, but then decline as individuals struggle to maintain cardiac function and respiration in the face of increased metabolic demands3,4. We provide evidence consistent with this prediction for a marine fish (Cheilodactylus spectabilis) in the Tasman Sea; one of the most rapidly warming regions of the Southern Hemisphere ocean5. We estimated changes in the species’ growth rate over a 90-year period using otoliths—bony structures that fish use for orientation and detection of movement—and compared these changes to temperature trends across the species’ distribution. Increasing temperatures coincide with increased growth for populations in the middle of the species range, but with reduced growth for those at the warm northern edge of the species’ distribution, indicating that temperatures may have already reached levels associated with increased metabolic costs. If warming continues, the direct metabolic effects of increasing temperatures on this species may lead to declining productivity and range contraction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pörtner, H. O. & Peck, M. A. Climate change effects on fishes and fisheries: Towards a cause-and-effect understanding. J. Fish. Biol. 77, 1745–1779 (2010).
Brander, K. Impacts of climate change on fisheries. ICES J. Mar. Sci. 79, 389–402 (2010).
Pörtner, H-O. & Knust, R. Climate change affects marine fishes through the oxygen limitation of thermal tolerance. Science 315, 95–97 (2007).
Wang, T. & Overgaard, J. The heartbreak of adapting to global warming. Science 315, 49–50 (2007).
Cai, W., Shi, G., Cowan, T., Bi, D. & Ribbe, J. The response of the Southern Annular Mode, the East Australian Current, and the southern mid-latitude ocean circulation to global warming. Geophys. Res. Lett. 32, L23706 (2005).
Fry, F. E. I. in Fish Physiology Vol. VI Environmental Relations and Behavior (eds Hoar, W. S. & Randall, D. J.) 1–99 (Academic, 1971).
Takasuka, A. & Aoki, I. Environmental determinants of growth rates for larval Japanese anchovy Engraulis japonicus in different waters. Fish. Oceanogr. 15, 139–149 (2006).
Pörtner, H-O. & Farrell, A. P. Physiology and climate change. Science 322, 690–692 (2008).
Katersky, R. S. & Carter, C. G. High growth efficiency occurs over a wide temperature range for juvenile barramundi Lates calcarifer fed a balanced diet. Aquaculture 272, 444–450 (2007).
Ridgway, K. R. Long-term trend and decadal variability of the southward penetration of the East Australian Current. Geophys. Res. Lett. 34, L13613 (2007).
Pecl, G. et al. The East Coast Tasmanian Rock Lobster Fishery—Vulnerability to Climate Change Impacts and Adaptation Response Options (Australian Government Department of Climate Change, 2009).
Hill, K. L., Rintoul, S. R., Coleman, R. & Ridgway, K. R. Wind forced low frequency variability of the East Australia Current. Geophys. Res. Lett. 35, L08602 (2008).
Campana, S. E. & Neilson, J. D. Microstructure of fish otoliths. Can. J. Fish. Aquat. Sci. 42, 1014–1032 (1985).
Neilson, J. D. & Geen, G. H. Effects of feeding regimes and diel temperature cycles on otolith increment formation in juvenile chinook salmon, Oncorhynchus tshawytscha. Fish. Bull. 83, 91–101 (1985).
Chambers, R. C. & Miller, T. J. in Recent Developments in Otolith Research (eds Secor, S. H., Dean, J. M. & Campana, S. E.) 155–175 (Univ. South Carolina Press, 1995).
Thresher, R. E., Koslow, J. A., Morison, A. K. & Smith, D. C. Depth-mediated reversal of the effects of climate change on long-term growth rates of exploited marine fish. Proc. Natl Acad. Sci. USA 104, 7461–7465 (2007).
Ewing, G. P., Lyle, J. M., Murphy, R. J., Kalish, J. M. & Ziegler, P. E. Validation of age and growth in a long-lived temperate reef fish using otolith structure, oxytetracycline and bomb radiocarbon methods. Mar. Freshwat. Res. 58, 944–955 (2007).
Armsworthy, S. L. & Campana, S. E. Age determination, bomb-radiocarbon validation and growth of Atlantic halibut (Hippoglossus hippoglossus) from the Northwest Atlantic. Environ. Biol. Fish. 89, 279–295 (2010).
Ziegler, P. E., Lyle, J. M., Haddon, M. & Ewing, G. P. Rapid changes in life-history characteristics of a long-lived temperate reef fish. Mar. Freshwat. Res. 58, 1096–1107 (2007).
Grewe, P. M., Smolenski, A. J. & Ward, R. D. Mitochondrial DNA variation in jackass morwong, Nemadactylus macropterus (Teleostei: Cheilodactylidae) from Australian and New Zealand waters. Can. J. Fish. Aquat. Sci. 51, 1101–1109 (1994).
Murphy, R. J. & Lyle, J. M. Impact of Gillnet Fishing on Inshore Temperate Reef Fishes, with Particular Reference to Banded Morwong. FRDC Project No. 95/145 (Marine Research Laboratories—Tasmanian Aquaculture and Fisheries Institute, University of Tasmania, 1999).
McCormick, M. I. & Choat, J. H. Estimating total abundance of a large temperate-reef fish using visual strip-transects. Mar. Biol. 96, 469–478 (1987).
Rayner, N. A. et al. Improved analyses of changes and uncertainties in sea surface temperature measured in situ since the mid-nineteenth century: The HadSST2 Dataset. J. Clim. 19, 446–469 (2006).
Rijnsdorp, A. D., Peck, M. A., Engelhard, G. H., Möllmann, C. & Pinnegar, J. K. Resolving the effect of climate change on fish populations. ICES J. Mar. Sci. 66, 1570–1583 (2009).
Heinimaa, S. & Heinimaa, P. Effect of the female size on egg quality and fecundity of the wild Atlantic salmon in the sub-arctic River Teno. Bor. Environ. Res. 9, 55–62 (2004).
Mittelbach, G. G. Foraging efficiency and body size: A study of optimal diet and habitat use by bluegills. Ecology 62, 1370–1386 (1981).
Beamish, R. J., Mahnken, C. & Neville, C. M. Evidence that reduced early marine growth is associated with lower marine survival of coho salmon. Trans. Am. Fisheries Soc. 133, 26–33 (2004).
Pörtner, H-O. et al. Cod and climate in a latitudinal cline: Physiological analyses of climate effects in marine fishes. Clim. Res. 37, 253–270 (2008).
Neuheimer, A. B. & Taggart, C. T. Can changes in length-at-age and maturation timing in Scotian Shelf haddock (Melanogrammus aeglefinus) be explained by fishing? Can. J. Fish. Aquat. Sci. 67, 854–865 (2010).
Clark, T. D. & Seymour, R. S. Cardiorespiratory physiology and swimming energetics of a high-energy-demand teleost, the yellowtail kingfish (Seriola lalandi). J. Exp. Biol. 209, 3940–3951 (2006).
Acknowledgements
We thank K. Clements and all who participated in the otolith collection supporting this study. We thank H. Choat, M. McCormick, and T. Stokie for sharing otolith samples. We thank J. McAllister, G. Ewing, S. Robertson, A. Hobday, P. Last and T. Mosgaard for discussions and guidance. We thank D. Webber, P. Frappell and J. Hagg for assistance with the activity experiments. This work was supported by the CSIRO Climate Adaptation Flagship Program, an Endeavour Awards Fellowship to A.B.N. and a Winnifred Violet Scott Trust Grant to J.M.S.
Author information
Authors and Affiliations
Contributions
All authors contributed to the design of the study. A.B.N. prepared (where necessary), aged and measured all otoliths, and analysed resulting growth increment and temperature data. J.M.S. designed, implemented and analysed the activity experiments. A.B.N. prepared the manuscript. R.E.T., J.M.L. and J.M.S. edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 730 kb)
Rights and permissions
About this article
Cite this article
Neuheimer, A., Thresher, R., Lyle, J. et al. Tolerance limit for fish growth exceeded by warming waters. Nature Clim Change 1, 110–113 (2011). https://doi.org/10.1038/nclimate1084
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nclimate1084
This article is cited by
-
Cascading effects of climate change on recreational marine flats fishes and fisheries
Environmental Biology of Fishes (2023)
-
Arctic charr phenotypic responses to abrupt generational scale temperature change: an insight into how cold-water fish could respond to extreme climatic events
Environmental Biology of Fishes (2023)
-
Arabian/Persian Gulf artisanal fisheries: magnitude, threats, and opportunities
Reviews in Fish Biology and Fisheries (2023)
-
Energy use, growth and survival of coral reef snapper larvae reared at elevated temperatures
Coral Reefs (2023)
-
Varying growth rates of a marine eel, the whitespotted conger (Conger myriaster), are explained by the interaction between seasonal temperature and prey availability
Marine Biology (2022)