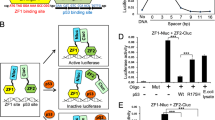
Abstract
p53-dependent apoptosis contributes to the side effects of cancer treatment, and genetic or pharmacological inhibition of p53 function can increase normal tissue resistance to genotoxic stress1,2,3,4,5,6. It has recently been shown that p53 can induce apoptosis through a mechanism that does not depend on transactivation but instead involves translocation of p53 to mitochondria7,8,9,10,11,12,13. To determine the impact of this p53 activity on normal tissue radiosensitivity, we isolated a small molecule named pifithrin-μ (PFTμ, 1) that inhibits p53 binding to mitochondria by reducing its affinity to antiapoptotic proteins Bcl-xL and Bcl-2 but has no effect on p53-dependent transactivation. PFTμ has a high specificity for p53 and does not protect cells from apoptosis induced by overexpression of proapoptotic protein Bax or by treatment with dexamethasone (2). PFTμ rescues primary mouse thymocytes from p53-mediated apoptosis caused by radiation and protects mice from doses of radiation that cause lethal hematopoietic syndrome. These results indicate that selective inhibition of the mitochondrial branch of the p53 pathway is sufficient for radioprotection in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Komarova, E.A. & Gudkov, A.V. Could p53 be a target for therapeutic suppression? Semin. Cancer Biol. 8, 389–400 (1998).
Komarov, P.G. et al. A chemical inhibitor of p53 that protects mice from the side effects of cancer therapy. Science 285, 1733–1737 (1999).
Botchkarev, V.A. et al. p53 is essential for chemotherapy-induced hair loss. Cancer Res. 60, 5002–5006 (2000).
Komarova, E.A. & Gudkov, A.V. Chemoprotection from p53-dependent apoptosis: potential clinical applications of the p53 inhibitors. Biochem. Pharmacol. 62, 657–667 (2001).
Gudkov, A.V. & Komarova, E.A. The role of p53 in determining sensitivity to radiotherapy. Nat. Rev. Cancer 3, 117–129 (2003).
Gudkov, A.V. & Komarova, E.A. Prospective therapeutic applications of p53 inhibitors. Biochem. Biophys. Res. Commun. 331, 726–736 (2005).
Marchenko, N.D., Zaika, A. & Moll, U.M. Death signal-induced localization of p53 protein to mitochondria. A potential role in apoptotic signaling. J. Biol. Chem. 275, 16202–16212 (2000).
Mihara, M. et al. p53 has a direct apoptogenic role at the mitochondria. Mol. Cell 11, 577–590 (2003).
Sansome, C., Zaika, A., Marchenko, N.D. & Moll, U.M. Hypoxia death stimulus induces translocation of p53 protein to mitochondria. Detection by immunofluorescence on whole cells. FEBS Lett. 488, 110–115 (2001).
Dumont, P., Leu, J.I., Della Pietra, A.C. III, George, D.L. & Murphy, M. The codon 72 polymorphic variants of p53 have markedly different apoptotic potential. Nat. Genet. 33, 357–365 (2003).
Chipuk, J.E. et al. Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 303, 1010–1014 (2004).
Erster, S., Mihara, M., Kim, R.H., Petrenko, O. & Moll, U.M. In vivo mitochondrial p53 translocation triggers a rapid first wave of cell death in response to DNA damage that can precede p53 target gene activation. Mol. Cell. Biol. 24, 6728–6741 (2004).
Erster, S. & Moll, U.M. Stress-induced p53 runs a transcription-independent death program. Biochem. Biophys. Res. Commun. 331, 843–850 (2005).
Soussi, T. & Beroud, C. Assessing TP53 status in human tumours to evaluate clinical outcome. Nat. Rev. Cancer 1, 233–240 (2001).
Vousden, K.H. p53: death star. Cell 103, 691–694 (2000).
May, P. & May, E. Twenty years of p53 research: structural and functional aspects of the p53 protein. Oncogene 18, 7621–7636 (1999).
Willis, A.C. & Chen, X. The promise and obstacle of p53 as a cancer therapeutic agent. Curr. Mol. Med. 2, 329–345 (2002).
Fang, B. & Roth, J.A. Tumor-suppressing gene therapy. Cancer Biol. Ther. 2, S115–S121 (2003).
Cui, Y.F. et al. Apoptosis in bone marrow cells of mice with different p53 genotypes after gamma-rays irradiation in vitro. J. Environ. Pathol. Toxicol. Oncol. 14, 159–163 (1995).
Song, S. & Lambert, P.F. Different responses of epidermal and hair follicular cells to radiation correlate with distinct patterns of p53 and p21 induction. Am. J. Pathol. 155, 1121–1127 (1999).
Komarova, E.A. et al. Transgenic mice with p53-responsive lacZ: p53 activity varies dramatically during normal development and determines radiation and drug sensitivity in vivo. EMBO J. 16, 1391–1400 (1997).
Chow, B.M., Li, Y.Q. & Wong, C.S. Radiation-induced apoptosis in the adult central nervous system is p53 dependent. Cell Death Differ. 7, 712–720 (2000).
Komarova, E.A. et al. Dual effect of p53 on radiation sensitivity in vivo: p53 promotes hematopoietic injury, but protects from gastro-intestinal syndrome in mice. Oncogene 23, 3265–3271 (2004).
Jeffers, J.R. et al. Puma is an essential mediator of p53-dependent and -independent apoptotic pathways. Cancer Cell 4, 321–328 (2003).
Lowe, S., Schmitt, E., Smith, S., Osborne, B. & Jacks, T. p53 is required for radiation-induced apoptosis in mouse thymocytes. in Nature 362, 847–849 (1993).
Merritt, A.J., Allen, T.D., Potten, C.S. & Hickman, J.A. Apoptosis in small intestinal epithelial from p53-null mice: evidence for a delayed, p53-independent G2/M-associated cell death after gamma irradiation. Oncogene 14, 2759–2766 (1997).
Komarova, E.A. et al. p53 is a suppressor of inflammatory response in mice. FASEB J. 19, 1030–1032 (2005).
Pfeifer, A., Ikawa, M., Dayn, Y. & Verma, I.M. Transgenesis by lentiviral vectors: lack of gene silencing in mammalian embryonic stem cells and preimplantation embryos. Proc. Natl. Acad. Sci. USA 99, 2140–2145 (2002).
Williams, G.T. & Morimoto, R.I. Maximal stress-induced transcription from the human HSP70 promoter requires interactions with the basal promoter elements independent of rotational alignment. Mol. Cell. Biol. 10, 3125–3136 (1990).
Bogenhagen, D. & Clayton, D.A. The number of mitochondrial deoxyribonucleic acid genomes in mouse L and human HeLa cells. Quantitative isolation of mitochondrial deoxyribonucleic acid. J. Biol. Chem. 249, 7991–7995 (1974).
Acknowledgements
We thank P. Chumakov, A. Almasan and M. Oren for providing recombinant constructs. This work was supported by grant CA75179 to A.V.G., CA103283 grant to Quark Biotech, Inc. and contract HHSN261200422015C to Cleveland BioLabs, Inc. from the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
E.S. generated data, performed data analyses and prepared the manuscript; S.S. generated data and performed data analyses; P.G.K. generated data and performed data analyses; O.B.C. managed part of the work; I.P., I.S., D.A.B. and L.G.B. generated data; R.M.M. performed data analysis; R.S. organized the research and performed data analysis; E.A.K. generated data, performed data analyses, prepared the manuscript and organized the research; and A.V.G. performed data analyses, prepared the manuscript and managed the project overall.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Effect of PFTμ (10 μM) on staurosporine-induced cell death in H1299 and HeLa cells. (PDF 91 kb)
Supplementary Fig. 2
Effect of PFTμ (0.1–10 μM) on Fas-, TNF- and staurosporine-induced apoptosis in human lung fibroblasts WI38. (PDF 91 kb)
Supplementary Fig. 3
Effect of PFTμ on TNF-induced activation of NFκB. (PDF 90 kb)
Supplementary Fig. 4
PFTμ (10 μM) does not affect the p53-mediated transactivation of promoters of Bax, Hdm2 and Cyclin G genes. (PDF 117 kb)
Supplementary Fig. 5
Lack of effect of PFTμ (10 μM) on viability of p53-null mouse thymocytes treated with 10 Gy of gamma radiation (estimated by Trypan blue uptake assay 24 h after irradiation). (PDF 90 kb)
Supplementary Fig. 6
A single intraperitoneal injection of 40 mg kg−1 of body weight of PFTμ did not change survival or dynamic of death of mice treated with 11 Gy of gamma radiation. (PDF 90 kb)
Rights and permissions
About this article
Cite this article
Strom, E., Sathe, S., Komarov, P. et al. Small-molecule inhibitor of p53 binding to mitochondria protects mice from gamma radiation. Nat Chem Biol 2, 474–479 (2006). https://doi.org/10.1038/nchembio809
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio809
This article is cited by
-
p53 Inhibition in Pancreatic Progenitors Enhances the Differentiation of Human Pluripotent Stem Cells into Pancreatic β-Cells
Stem Cell Reviews and Reports (2023)
-
CD44a functions as a regulator of p53 signaling, apoptosis and autophagy in the antibacterial immune response
Communications Biology (2022)
-
The regulatory roles of p53 in cardiovascular health and disease
Cellular and Molecular Life Sciences (2021)
-
The Localization of p53 in the Crayfish Mechanoreceptor Neurons and Its Role in Axotomy-Induced Death of Satellite Glial Cells Remote from the Axon Transection Site
Journal of Molecular Neuroscience (2020)
-
Involvement of p53 in the Responses of Cardiac Muscle Cells to Heat Shock Exposure and Heat Acclimation
Journal of Cardiovascular Translational Research (2020)