Abstract
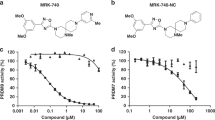
Protein lysine methyltransferases G9a and GLP modulate the transcriptional repression of a variety of genes via dimethylation of Lys9 on histone H3 (H3K9me2) as well as dimethylation of non-histone targets. Here we report the discovery of UNC0638, an inhibitor of G9a and GLP with excellent potency and selectivity over a wide range of epigenetic and non-epigenetic targets. UNC0638 treatment of a variety of cell lines resulted in lower global H3K9me2 levels, equivalent to levels observed for small hairpin RNA knockdown of G9a and GLP with the functional potency of UNC0638 being well separated from its toxicity. UNC0638 markedly reduced the clonogenicity of MCF7 cells, reduced the abundance of H3K9me2 marks at promoters of known G9a-regulated endogenous genes and disproportionately affected several genomic loci encoding microRNAs. In mouse embryonic stem cells, UNC0638 reactivated G9a-silenced genes and a retroviral reporter gene in a concentration-dependent manner without promoting differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Kouzarides, T. Chromatin modifications and their function. Cell 128, 693–705 (2007).
Martin, C. & Zhang, Y. The diverse functions of histone lysine methylation. Nat. Rev. Mol. Cell Biol. 6, 838–849 (2005).
Jenuwein, T. & Allis, C.D. Translating the histone code. Science 293, 1074–1080 (2001).
Bernstein, B.E., Meissner, A. & Lander, E.S. The mammalian epigenome. Cell 128, 669–681 (2007).
Gelato, K.A. & Fischle, W. Role of histone modifications in defining chromatin structure and function. Biol. Chem. 389, 353–363 (2008).
Strahl, B.D. & Allis, C.D. The language of covalent histone modifications. Nature 403, 41–45 (2000).
Huang, J. et al. Repression of p53 activity by Smyd2-mediated methylation. Nature 444, 629–632 (2006).
Huang, J. et al. G9A and GLP methylate lysine 373 in the tumor suppressor p53. J. Biol. Chem. 285, 9636–9641 (2010).
Rathert, P. et al. Protein lysine methyltransferase G9a acts on non-histone targets. Nat. Chem. Biol. 4, 344–346 (2008).
Huang, J. et al. p53 is regulated by the lysine demethylase LSD1. Nature 449, 105–108 (2007).
Copeland, R.A., Solomon, M.E. & Richon, V.M. Protein methyltransferases as a target class for drug discovery. Nat. Rev. Drug Discov. 8, 724–732 (2009).
Rea, S. et al. Regulation of chromatin structure by site-specific histone H3 methyltransferases. Nature 406, 593–599 (2000).
Cole, P.A. Chemical probes for histone-modifying enzymes. Nat. Chem. Biol. 4, 590–597 (2008).
Esteller, M. Epigenetics in cancer. N. Engl. J. Med. 358, 1148–1159 (2008).
Tachibana, M. et al. G9a histone methyltransferase plays a dominant role in euchromatic histone H3 lysine 9 methylation and is essential for early embryogenesis. Genes Dev. 16, 1779–1791 (2002).
Kondo, Y. et al. Downregulation of histone H3 lysine 9 methyltransferase G9a induces centrosome disruption and chromosome instability in cancer cells. PLoS ONE 3, e2037 (2008).
Kondo, Y. et al. Alterations of DNA methylation and histone modifications contribute to gene silencing in hepatocellular carcinomas. Hepatol. Res. 37, 974–983 (2007).
Watanabe, H. et al. Deregulation of histone lysine methyltransferases contributes to oncogenic transformation of human bronchoepithelial cells. Cancer Cell Int. 8, 15 (2008).
Goyama, S. et al. EVI-1 interacts with histone methyltransferases SUV39H1 and G9a for transcriptional repression and bone marrow immortalization. Leukemia 24, 81–88 (2010).
Tachibana, M. et al. Histone methyltransferases G9a and GLP form heteromeric complexes and are both crucial for methylation of euchromatin at H3–K9. Genes Dev. 19, 815–826 (2005).
Maze, I. et al. Essential role of the histone methyltransferase G9a in cocaine-induced plasticity. Science 327, 213–216 (2010).
Schaefer, A. et al. Control of cognition and adaptive behavior by the GLP/G9a epigenetic suppressor complex. Neuron 64, 678–691 (2009).
Imai, K., Togami, H. & Okamoto, T. Involvement of histone H3 Lysine 9 (H3K9) methyl transferase G9a in the maintenance of HIV-1 latency and its reactivation by BIX01294. J. Biol. Chem. 285, 16538–16545 (2010).
Link, P.A. et al. Distinct roles for histone methyltransferases G9a and GLP in cancer germ-line antigen gene regulation in human cancer cells and murine embryonic stem cells. Mol. Cancer Res. 7, 851–862 (2009).
Tachibana, M., Matsumura, Y., Fukuda, M., Kimura, H. & Shinkai, Y. G9a/GLP complexes independently mediate H3K9 and DNA methylation to silence transcription. EMBO J. 27, 2681–2690 (2008).
Dong, K.B. et al. DNA methylation in ES cells requires the lysine methyltransferase G9a but not its catalytic activity. EMBO J. 27, 2691–2701 (2008).
Shi, Y. et al. Induction of pluripotent stem cells from mouse embryonic fibroblasts by Oct4 and Klf4 with small-molecule compounds. Cell Stem Cell 3, 568–574 (2008).
Shi, Y. et al. A combined chemical and genetic approach for the generation of induced pluripotent stem cells. Cell Stem Cell 2, 525–528 (2008).
Kubicek, S. et al. Reversal of H3K9me2 by a small-molecule inhibitor for the G9a histone methyltransferase. Mol. Cell 25, 473–481 (2007).
Frye, S.V. The art of the chemical probe. Nat. Chem. Biol. 6, 159–161 (2010).
Liu, F. et al. Discovery of a 2,4-diamino-7-aminoalkoxyquinazoline as a potent and selective inhibitor of histone lysine methyltransferase G9a. J. Med. Chem. 52, 7950–7953 (2009).
Liu, F. et al. Protein lysine methyltransferase G9a inhibitors: design, synthesis, and structure activity relationships of 2,4-diamino-7-aminoalkoxy-quinazolines. J. Med. Chem. 53, 5844–5857 (2010).
Chang, Y. et al. Adding a lysine mimic in the design of potent inhibitors of histone lysine methyltransferases. J. Mol. Biol. 400, 1–7 (2010).
Chang, Y. et al. Structural basis for G9a-like protein lysine methyltransferase inhibition by BIX-01294. Nat. Struct. Mol. Biol. 16, 312–317 (2009).
Collazo, E., Couture, J.F., Bulfer, S. & Trievel, R.C. A coupled fluorescent assay for histone methyltransferases. Anal. Biochem. 342, 86–92 (2005).
Wigle, T.J. et al. Accessing protein methyltransferase and demethylase enzymology using microfluidic capillary electrophoresis. Chem. Biol. 17, 695–704 (2010).
Morrison, J.F. Kinetics of the reversible inhibition of enzyme-catalysed reactions by tight-binding inhibitors. Biochim. Biophys. Acta 185, 269–286 (1969).
Zee, B.M. et al. In vivo residue-specific histone methylation dynamics. J. Biol. Chem. 285, 3341–3350 (2010).
Plazas-Mayorca, M.D. et al. Quantitative proteomics reveals direct and indirect alterations in the histone code following methyltransferase knockdown. Mol. Biosyst. 6, 1719–1729 (2010).
Barski, A. et al. High-resolution profiling of histone methylations in the human genome. Cell 129, 823–837 (2007).
Rice, J.C. et al. Histone methyltransferases direct different degrees of methylation to define distinct chromatin domains. Mol. Cell 12, 1591–1598 (2003).
Vakoc, C.R., Mandat, S.A., Olenchock, B.A. & Blobel, G.A. Histone H3 lysine 9 methylation and HP1gamma are associated with transcription elongation through mammalian chromatin. Mol. Cell 19, 381–391 (2005).
Wolf, D. & Goff, S.P. TRIM28 mediates primer binding site-targeted silencing of murine leukemia virus in embryonic cells. Cell 131, 46–57 (2007).
Yokochi, T. et al. G9a selectively represses a class of late-replicating genes at the nuclear periphery. Proc. Natl. Acad. Sci. USA 106, 19363–19368 (2009).
Hosey, A.M., Chaturvedi, C.P. & Brand, M. Crosstalk between histone modifications maintains the developmental pattern of gene expression on a tissue-specific locus. Epigenetics 5, 273–281 (2010).
Epsztejn-Litman, S. et al. De novo DNA methylation promoted by G9a prevents reprogramming of embryonically silenced genes. Nat. Struct. Mol. Biol. 15, 1176–1183 (2008).
Estève, P.O. et al. Direct interaction between DNMT1 and G9a coordinates DNA and histone methylation during replication. Genes Dev. 20, 3089–3103 (2006).
Chen, L. et al. MDM2 recruitment of lysine methyltransferases regulates p53 transcriptional output. EMBO J. 29, 2538–2552 (2010).
Fritsch, L. et al. A subset of the histone H3 lysine 9 methyltransferases Suv39h1, G9a, GLP, and SETDB1 participate in a multimeric complex. Mol. Cell 37, 46–56 (2010).
Stadtfeld, M., Maherali, N., Breault, D.T. & Hochedlinger, K. Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell 2, 230–240 (2008).
Acknowledgements
We thank A. Tumber for JMJD2E assay support; J. Moffat (University of Toronto) for the gift of shRNAs; R. Bristow (University Health Network) for RV221 and PC3 cells; T. Hajian and F. Syeda for protein purification; G. Senisterra for contributing to DSF and DSLS data analysis; M. Herold for graphical design and illustration; I. Korboukh, M. Herold and J. Yost for critical reading of the manuscript; and R. Trump and C. Yates for helpful discussion. The research described here was supported by the National Institute of General Medical Sciences, US National Institutes of Health (NIH; grant RC1GM090732), the Carolina Partnership and University Cancer Research Fund from the University of North Carolina at Chapel Hill, the US National Science Foundation (NSF), the Ontario Research Fund, the Ontario Ministry of Health and Long-term Care and the Structural Genomics Consortium. The Structural Genomics Consortium is a registered charity (number 1097737) that receives funds from the Canadian Institutes for Health Research (CIHR), the Canada Foundation for Innovation, Genome Canada through the Ontario Genomics Institute, GlaxoSmithKline, Karolinska Institutet, the Knut and Alice Wallenberg Foundation, the Ontario Innovation Trust, the Ontario Ministry for Research and Innovation, Merck & Co. Inc., the Novartis Research Foundation, the Swedish Agency for Innovation Systems, the Swedish Foundation for Strategic Research and the Wellcome Trust. C.H.A. holds a Canada Research Chair in Structural Genomics. V.L. is supported by a CIHR fellowship. A.P. is supported by grants from the CIHR (199170 and 186007) and from the NIH (MH074127, MH088413, DP3DK085698 and HG004535). A.P. is Tapscott Chair in Schizophrenia Studies and a Senior Fellow of the Ontario Mental Health Foundation. J.E. is supported by CIHR grant IG1-102956. B.A.G. is supported by grants from the NSF (Early Faculty CAREER award and CBET-0941143), the American Society for Mass Spectrometry and the NIH Office of the Director (DP2OD007447). P.A.D. is supported by NIH postdoctoral fellowship F32 NRSA.
Author information
Authors and Affiliations
Contributions
M.V., A.A.-H., A.S. and I.C. performed SAHH-coupled, fluorescence-polarization, DSF, DSLS and DNMT1 assays; D.B.-L. developed and performed in-cell western, MTT, ChIP, gene expression, clonogenicity, western blotting and immunofluorescence studies; F.L. developed the synthetic route to UNC0638 and UNC0737 and synthesized the compounds; S.R.-G. and J.E. developed and performed mES cell studies; V.L., S.-C.W. and A.P. performed H3K9me2 genomic localization and DNA methylation analysis; T.J.W. and W.P.J. performed mechanism-of-action studies; P.A.D. and B.A.G. performed MS-based proteomics studies; M.V., G.A.W., A.D., W.T., D.B.K. and C.H.A. solved and analyzed the X-ray crystal structure of the G9a-UNC0638-SAH complex; T.J.W. and A.T. performed SPR studies; X.C. and S.G.P. performed UNC0638 stability studies; S.G.P. performed RT-qPCR studies; T.J.M., X.-p.H. and B.L.R. performed GPCR selectivity studies; C.D.S. and W.P.J. performed kinase selectivity studies; J.L.N. purified proteins; J.J. designed UNC0638 and UNC0737; C.H.A., J.J., S.V.F., M.V., D.B.-L., P.J.B., J.E., S.R.-G., A.P., V.L., B.A.G., T.J.W. and A.E. designed studies and discussed results; J.J., C.H.A., D.B.-L., S.R.-G., M.V., V.L., S.V.F., J.E., A.P., B.A.G., P.J.B. and T.J.W. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Methods and Supplementary Results (PDF 2603 kb)
Rights and permissions
About this article
Cite this article
Vedadi, M., Barsyte-Lovejoy, D., Liu, F. et al. A chemical probe selectively inhibits G9a and GLP methyltransferase activity in cells. Nat Chem Biol 7, 566–574 (2011). https://doi.org/10.1038/nchembio.599
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.599
This article is cited by
-
Systematic literature review reveals suboptimal use of chemical probes in cell-based biomedical research
Nature Communications (2023)
-
G9a/GLP inhibition during ex vivo lymphocyte expansion increases in vivo cytotoxicity of engineered T cells against hepatocellular carcinoma
Nature Communications (2023)
-
A specific G9a inhibitor unveils BGLT3 lncRNA as a universal mediator of chemically induced fetal globin gene expression
Nature Communications (2023)
-
Reduced and highly diverse peripheral HIV-1 reservoir in virally suppressed patients infected with non-B HIV-1 strains in Uganda
Retrovirology (2022)
-
Repression of the PRELP gene is relieved by histone deacetylase inhibitors through acetylation of histone H2B lysine 5 in bladder cancer
Clinical Epigenetics (2022)