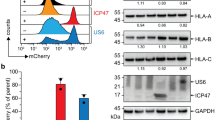
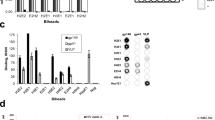
Abstract
How ABC transporters work is a key issue because of their important roles in multidrug resistance of pathogenic bacteria, reduced efficacy of antitumor drugs, cholesterol metabolism, cell homeostasis and immune response. In the past few years, significant progress has been made in crystallization and structure determination of (mostly) bacterial ABC transporters, as well as in functional studies on ABC systems involved in human pathology. In this review, we use the transporter associated with antigen processing (TAP) to illustrate what is known regarding the mechanism of substrate transport. We also discuss the chemical basis of substrate recognition by TAP and the allosteric cross-talk between the binding of substrate, the release of chemical energy by ATP hydrolysis and cross-membrane translocation. Finally, we detail the role of TAP in a large macromolecular assembly, which optimally loads MHC class I molecules, and the interference with this machinery by TAP-targeted viral factors. Because of structural and probable mechanistic similarities, the understanding of the detailed structure and mechanism of TAP will be applicable to all ABC systems, including those of medical relevance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
17 September 2010
In the version of this article initially published, four of the ATP molecules in Figure 4a were incorrectly labeled as ADP. The errors have been corrected in the HTML and PDF versions of the article.
References
Higgins, C.F. ABC transporters: from microorganisms to man. Annu. Rev. Cell Biol. 8, 67–113 (1992).
Schmitt, L. & Tampé, R. Structure and mechanism of ABC transporters. Curr. Opin. Struct. Biol. 12, 754–760 (2002).
Rees, D.C., Johnson, E. & Lewinson, O. ABC transporters: the power to change. Nat. Rev. Mol. Cell Biol. 10, 218–227 (2009).
Hollenstein, K., Dawson, R.J. & Locher, K.P. Structure and mechanism of ABC transporter proteins. Curr. Opin. Struct. Biol. 17, 412–418 (2007).
Hollenstein, K., Frei, D.C. & Locher, K.P. Structure of an ABC transporter in complex with its binding protein. Nature 446, 213–216 (2007).
Oldham, M.L., Khare, D., Quiocho, F.A., Davidson, A.L. & Chen, J. Crystal structure of a catalytic intermediate of the maltose transporter. Nature 450, 515–521 (2007).
Kadaba, N.S., Kaiser, J.T., Johnson, E., Lee, A. & Rees, D.C. The high-affinity E. coli methionine ABC transporter: structure and allosteric regulation. Science 321, 250–253 (2008).
Locher, K.P., Lee, A.T. & Rees, D.C. The E. coli BtuCD structure: a framework for ABC transporter architecture and mechanism. Science 296, 1091–1098 (2002).
Pinkett, H.W., Lee, A.T., Lum, P., Locher, K.P. & Rees, D.C. An inward-facing conformation of a putative metal-chelate-type ABC transporter. Science 315, 373–377 (2007).
Dawson, R.J. & Locher, K.P. Structure of a bacterial multidrug ABC transporter. Nature 443, 180–185 (2006).
Ward, A., Reyes, C.L., Yu, J., Roth, C.B. & Chang, G. Flexibility in the ABC transporter MsbA: alternating access with a twist. Proc. Natl. Acad. Sci. USA 104, 19005–19010 (2007).
Aller, S.G. et al. Structure of P-glycoprotein reveals a molecular basis for poly-specific drug binding. Science 323, 1718–1722 (2009).
Lankat-Buttgereit, B. & Tampé, R. The transporter associated with antigen processing: function and implications in human diseases. Physiol. Rev. 82, 187–204 (2002).
Wearsch, P.A. & Cresswell, P. The quality control of MHC class I peptide loading. Curr. Opin. Cell Biol. 20, 624–631 (2008).
Koch, J. & Tampé, R. The macromolecular peptide-loading complex in MHC class I-dependent antigen presentation. Cell. Mol. Life Sci. 63, 653–662 (2006).
Koch, J., Guntrum, R., Heintke, S., Kyritsis, C. & Tampé, R. Functional dissection of the transmembrane domains of the transporter associated with antigen processing (TAP). J. Biol. Chem. 279, 10142–10147 (2004).
Schrodt, S., Koch, J. & Tampé, R. Membrane topology of the transporter associated with antigen processing (TAP1) within an assembled functional peptide-loading complex. J. Biol. Chem. 281, 6455–6462 (2006).
Procko, E., Raghuraman, G., Wiley, D.C., Raghavan, M. & Gaudet, R. Identification of domain boundaries within the N-termini of TAP1 and TAP2 and their importance in tapasin binding and tapasin-mediated increase in peptide loading of MHC class I. Immunol. Cell Biol. 83, 475–482 (2005).
Leonhardt, R.M., Keusekotten, K., Bekpen, C. & Knittler, M.R. Critical role for the tapasin-docking site of TAP2 in the functional integrity of the MHC class I-peptide-loading complex. J. Immunol. 175, 5104–5114 (2005).
Ortmann, B. et al. A critical role for tapasin in the assembly and function of multimeric MHC class I-TAP complexes. Science 277, 1306–1309 (1997).
Oancea, G. et al. Structural arrangement of the transmission interface in the antigen ABC transport complex TAP. Proc. Natl. Acad. Sci. USA 106, 5551–5556 (2009).
Herget, M. et al. Purification and reconstitution of the antigen transport complex TAP: a prerequisite for determination of peptide stoichiometry and ATP hydrolysis. J. Biol. Chem. 284, 33740–33749 (2009).
Momburg, F. et al. Selectivity of MHC-encoded peptide transporters from human, mouse and rat. Nature 367, 648–651 (1994).
van Endert, P.M. et al. A sequential model for peptide binding and transport by the transporters associated with antigen processing. Immunity 1, 491–500 (1994).
Blanchard, N. & Shastri, N. Coping with loss of perfection in the MHC class I peptide repertoire. Curr. Opin. Immunol. 20, 82–88 (2008).
Heemels, M.T. & Ploegh, H.L. Substrate specificity of allelic variants of the TAP peptide transporter. Immunity 1, 775–784 (1994).
van Endert, P.M. et al. The peptide-binding motif for the human transporter associated with antigen processing. J. Exp. Med. 182, 1883–1895 (1995).
Uebel, S. et al. Recognition principle of the TAP transporter disclosed by combinatorial peptide libraries. Proc. Natl. Acad. Sci. USA 94, 8976–8981 (1997).
York, I.A., Goldberg, A.L., Mo, X.Y. & Rock, K.L. Proteolysis and class I major histocompatibility complex antigen presentation. Immunol. Rev. 172, 49–66 (1999).
Uebel, S. & Tampé, R. Specificity of the proteasome and the TAP transporter. Curr. Opin. Immunol. 11, 203–208 (1999).
Higgins, C.F. ABC transporters: physiology, structure and mechanism–an overview. Res. Microbiol. 152, 205–210 (2001).
Szakács, G., Paterson, J.K., Ludwig, J.A., Booth-Genthe, C. & Gottesman, M.M. Targeting multidrug resistance in cancer. Nat. Rev. Drug Discov. 5, 219–234 (2006).
Nijenhuis, M. & Hämmerling, G.J. Multiple regions of the transporter associated with antigen processing (TAP) contribute to its peptide binding site. J. Immunol. 157, 5467–5477 (1996).
Herget, M. et al. Mechanism of substrate sensing and signal transmission within an ABC transporter: use of a Trojan horse strategy. J. Biol. Chem. 282, 3871–3880 (2007).
Crowley, E. & Callaghan, R. Multidrug efflux pumps: drug binding—gates or cavity? FEBS J. 277, 530–539 (2010).
Gatlik-Landwojtowicz, E., Aanismaa, P. & Seelig, A. Quantification and characterization of P-glycoprotein-substrate interactions. Biochemistry 45, 3020–3032 (2006).
Hopfner, K.P. & Tainer, J.A. Rad50/SMC proteins and ABC transporters: unifying concepts from high-resolution structures. Curr. Opin. Struct. Biol. 13, 249–255 (2003).
Smith, P.C. et al. ATP binding to the motor domain from an ABC transporter drives formation of a nucleotide sandwich dimer. Mol. Cell 10, 139–149 (2002).
Jones, P.M. & George, A.M. Subunit interactions in ABC transporters: towards a functional architecture. FEMS Microbiol. Lett. 179, 187–202 (1999).
Chen, M., Abele, R. & Tampé, R. Peptides induce ATP hydrolysis at both subunits of the transporter associated with antigen processing. J. Biol. Chem. 278, 29686–29692 (2003).
Lapinski, P.E., Raghuraman, G. & Raghavan, M. Nucleotide interactions with membrane-bound transporter associated with antigen processing proteins. J. Biol. Chem. 278, 8229–8237 (2003).
Procko, E., Ferrin-O'Connell, I., Ng, S.L. & Gaudet, R. Distinct structural and functional properties of the ATPase sites in an asymmetric ABC transporter. Mol. Cell 24, 51–62 (2006).
Janas, E. et al. The ATP hydrolysis cycle of the nucleotide-binding domain of the mitochondrial ATP-binding cassette transporter Mdl1p. J. Biol. Chem. 278, 26862–26869 (2003).
van der Does, C. & Tampé, R. How do ABC transporters drive transport? Biol. Chem. 385, 927–933 (2004).
Abele, R. & Tampé, R. The ABCs of immunology: structure and function of TAP, the transporter associated with antigen processing. Physiology (Bethesda) 19, 216–224 (2004).
Higgins, C.F. & Linton, K.J. The ATP switch model for ABC transporters. Nat. Struct. Mol. Biol. 11, 918–926 (2004).
Senior, A.E. & Gadsby, D.C. ATP hydrolysis cycles and mechanism in P-glycoprotein and CFTR. Semin. Cancer Biol. 8, 143–150 (1997).
Jones, P.M., O'Mara, M.L. & George, A.M. ABC transporters: a riddle wrapped in a mystery inside an enigma. Trends Biochem. Sci. 34, 520–531 (2009).
Dalmas, O. et al. The Q-loop disengages from the first intracellular loop during the catalytic cycle of the multidrug ABC transporter BmrA. J. Biol. Chem. 280, 36857–36864 (2005).
Zolnerciks, J.K., Wooding, C. & Linton, K.J. Evidence for a Sav1866-like architecture for the human multidrug transporter P-glycoprotein. FASEB J. 21, 3937–3948 (2007).
Pagant, S., Brovman, E.Y., Halliday, J.J. & Miller, E.A. Mapping of interdomain interfaces required for the functional architecture of Yor1p, a eukaryotic ATP-binding cassette (ABC) transporter. J. Biol. Chem. 283, 26444–26451 (2008).
Serohijos, A.W. et al. Phenylalanine-508 mediates a cytoplasmic-membrane domain contact in the CFTR 3D structure crucial to assembly and channel function. Proc. Natl. Acad. Sci. USA 105, 3256–3261 (2008).
Uebel, S. et al. Requirements for peptide binding to the human transporter associated with antigen processing revealed by peptide scans and complex peptide libraries. J. Biol. Chem. 270, 18512–18516 (1995).
Cresswell, P., Bangia, N., Dick, T. & Diedrich, G. The nature of the MHC class I peptide loading complex. Immunol. Rev. 172, 21–28 (1999).
Peaper, D.R. & Cresswell, P. Regulation of MHC class I assembly and peptide binding. Annu. Rev. Cell Dev. Biol. 24, 343–368 (2008).
Rufer, E., Leonhardt, R.M. & Knittler, M.R. Molecular architecture of the TAP-associated MHC class I peptide-loading complex. J. Immunol. 179, 5717–5727 (2007).
Elliott, T. & Williams, A. The optimization of peptide cargo bound to MHC class I molecules by the peptide-loading complex. Immunol. Rev. 207, 89–99 (2005).
Praveen, P.V., Yaneva, R., Kalbacher, H. & Springer, S. Tapasin edits peptides on MHC class I molecules by accelerating peptide exchange. Eur. J. Immunol. 40, 214–224 (2010).
Wearsch, P.A. & Cresswell, P. Selective loading of high-affinity peptides onto major histocompatibility complex class I molecules by the tapasin-ERp57 heterodimer. Nat. Immunol. 8, 873–881 (2007).
Garbi, N., Hammerling, G. & Tanaka, S. Interaction of ERp57 and tapasin in the generation of MHC class I-peptide complexes. Curr. Opin. Immunol. 19, 99–105 (2007).
Dong, G., Wearsch, P.A., Peaper, D.R., Cresswell, P. & Reinisch, K.M. Insights into MHC class I peptide loading from the structure of the tapasin-ERp57 thiol oxidoreductase heterodimer. Immunity 30, 21–32 (2009).
Peaper, D.R. & Cresswell, P. The redox activity of ERp57 is not essential for its functions in MHC class I peptide loading. Proc. Natl. Acad. Sci. USA 105, 10477–10482 (2008).
Purcell, A.W. & Elliott, T. Molecular machinations of the MHC-I peptide loading complex. Curr. Opin. Immunol. 20, 75–81 (2008).
Koopmann, J.O. et al. Export of antigenic peptides from the endoplasmic reticulum intersects with retrograde protein translocation through the Sec61p channel. Immunity 13, 117–127 (2000).
Procko, E., O'Mara, M.L., Bennett, W.F., Tieleman, D.P. & Gaudet, R. The mechanism of ABC transporters: general lessons from structural and functional studies of an antigenic peptide transporter. FASEB J. 23, 1287–1302 (2009).
Demirel, O., Bangert, I., Tampé, R. & Abele, R. Tuning the cellular trafficking of the lysosomal peptide transporter TAPL by its N-terminal domain. Traffic 11, 383–393 (2010).
Ahn, K. et al. Molecular mechanism and species specificity of TAP inhibition by herpes simplex virus ICP47. EMBO J. 15, 3247–3255 (1996).
Früh, K. et al. A viral inhibitor of peptide transporters for antigen presentation. Nature 375, 415–418 (1995).
Tomazin, R. et al. Stable binding of the herpes simplex virus ICP47 protein to the peptide binding site of TAP. EMBO J. 15, 3256–3266 (1996).
Hill, A. et al. Herpes simplex virus turns off the TAP to evade host immunity. Nature 375, 411–415 (1995).
Neumann, L., Kraas, W., Uebel, S., Jung, G. & Tampé, R. The active domain of the herpes simplex virus protein ICP47: a potent inhibitor of the transporter associated with antigen processing. J. Mol. Biol. 272, 484–492 (1997).
Galocha, B. et al. The active site of ICP47, a herpes simplex virus-encoded inhibitor of the major histocompatibility complex (MHC)-encoded peptide transporter associated with antigen processing (TAP), maps to the NH2-terminal 35 residues. J. Exp. Med. 185, 1565–1572 (1997).
Beinert, D., Neumann, L., Uebel, S. & Tampé, R. Structure of the viral TAP-inhibitor ICP47 induced by membrane association. Biochemistry 36, 4694–4700 (1997).
Aisenbrey, C. et al. Structure and dynamics of membrane-associated ICP47, a viral inhibitor of the MHC I antigen-processing machinery. J. Biol. Chem. 281, 30365–30372 (2006).
Ahn, K. et al. The ER-luminal domain of the HCMV glycoprotein US6 inhibits peptide translocation by TAP. Immunity 6, 613–621 (1997).
Hengel, H. et al. A viral ER-resident glycoprotein inactivates the MHC-encoded peptide transporter. Immunity 6, 623–632 (1997).
Lehner, P.J., Karttunen, J.T., Wilkinson, G.W. & Cresswell, P. The human cytomegalovirus US6 glycoprotein inhibits transporter associated with antigen processing-dependent peptide translocation. Proc. Natl. Acad. Sci. USA 94, 6904–6909 (1997).
Hewitt, E.W., Gupta, S.S. & Lehner, P.J. The human cytomegalovirus gene product US6 inhibits ATP binding by TAP. EMBO J. 20, 387–396 (2001).
Kyritsis, C. et al. Molecular mechanism and structural aspects of transporter associated with antigen processing inhibition by the cytomegalovirus protein US6. J. Biol. Chem. 276, 48031–48039 (2001).
Ackerman, A.L., Kyritsis, C., Tampé, R. & Cresswell, P. Early phagosomes in dendritic cells form a cellular compartment sufficient for cross presentation of exogenous antigens. Proc. Natl. Acad. Sci. USA 100, 12889–12894 (2003).
Ackerman, A.L., Giodini, A. & Cresswell, P. A role for the endoplasmic reticulum protein retrotranslocation machinery during crosspresentation by dendritic cells. Immunity 25, 607–617 (2006).
Burgdorf, S., Scholz, C., Kautz, A., Tampé, R. & Kurts, C. Spatial and mechanistic separation of cross-presentation and endogenous antigen presentation. Nat. Immunol. 9, 558–566 (2008).
Koppers-Lalic, D. et al. Varicelloviruses avoid T cell recognition by UL49.5-mediated inactivation of the transporter associated with antigen processing. Proc. Natl. Acad. Sci. USA 102, 5144–5149 (2005).
Koppers-Lalic, D. et al. Varicellovirus UL 49.5 proteins differentially affect the function of the transporter associated with antigen processing, TAP. PLoS Pathog. 4, e1000080 (2008).
Loch, S. et al. Signaling of a varicelloviral factor across the endoplasmic reticulum membrane induces destruction of the peptide-loading complex and immune evasion. J. Biol. Chem. 283, 13428–13436 (2008).
Horst, D. et al. Specific targeting of the EBV lytic phase protein BNLF2a to the transporter associated with antigen processing results in impairment of HLA class I-restricted antigen presentation. J. Immunol. 182, 2313–2324 (2009).
Croft, N.P. et al. Stage-specific inhibition of MHC class I presentation by the Epstein-Barr virus BNLF2a protein during virus lytic cycle. PLoS Pathog. 5, e1000490 (2009).
Alzhanova, D. et al. Cowpox virus inhibits the transporter associated with antigen processing to evade T cell recognition. Cell Host Microbe 6, 433–445 (2009).
Byun, M. et al. Two mechanistically distinct immune evasion proteins of cowpox virus combine to avoid antiviral CD8 T cells. Cell Host Microbe 6, 422–432 (2009).
DeLano, W.L. The PyMOL Graphics System (DeLano Scientific, San Carlos, California, USA, 2002).
Pettersen, E.F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Petrey, D. et al. Using multiple structure alignments, fast model building, and energetic analysis in fold recognition and homology modeling. Proteins 53, 430–435 (2003).
Chen, M., Abele, R. & Tampé, R. Functional non-equivalence of ATP-binding cassette signature motifs in the transporter associated with antigen processing (TAP). J. Biol. Chem. 279, 46073–46081 (2004).
Zaitseva, J., Jenewein, S., Jumpertz, T., Holland, I.B. & Schmitt, L. H662 is the linchpin of ATP hydrolysis in the nucleotide-binding domain of the ABC transporter HlyB. EMBO J. 24, 1901–1910 (2005).
Armandola, E.A. et al. A point mutation in the human transporter associated with antigen processing (TAP2) alters the peptide transport specificity. Eur. J. Immunol. 26, 1748–1755 (1996).
Momburg, F., Armandola, E.A., Post, M. & Hammerling, G.J. Residues in TAP2 peptide transporters controlling substrate specificity. J. Immunol. 156, 1756–1763 (1996).
Baldauf, C., Schrodt, S., Herget, M., Koch, J. & Tampé, R. Single residue within the antigen translocation complex TAP controls the epitope repertoire by stabilizing a receptive conformation. Proc. Natl. Acad. Sci. USA 107, 9135–9140 (2010).
Acknowledgements
The work in R.T.'s lab is supported by the German Research Foundation (SFB 807 – Transport and Communication Across Biological Membranes, TA157/7 and AB149/1) as well as by the European Drug Initiative on Channels and Transporters project, funded by the European Commission Seventh Framework Program.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Parcej, D., Tampé, R. ABC proteins in antigen translocation and viral inhibition. Nat Chem Biol 6, 572–580 (2010). https://doi.org/10.1038/nchembio.410
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.410
This article is cited by
-
Antigen presentation in cancer — mechanisms and clinical implications for immunotherapy
Nature Reviews Clinical Oncology (2023)
-
A guide to antigen processing and presentation
Nature Reviews Immunology (2022)
-
Different structures of berberine and five other protoberberine alkaloids that affect P-glycoprotein-mediated efflux capacity
Acta Pharmacologica Sinica (2019)
-
Flexible-to-rigid transition is central for substrate transport in the ABC transporter BmrA from Bacillus subtilis
Communications Biology (2019)
-
Single-molecule probing of the conformational homogeneity of the ABC transporter BtuCD
Nature Chemical Biology (2018)