Abstract
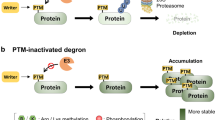
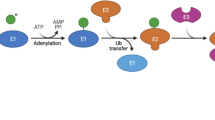
Non-enzymatic post-translational modifications of proteins can occur when a nucleophilic or redox-sensitive amino acid side chain encounters a reactive metabolite. In many cases, the biological function of these modifications is limited by their irreversibility, and consequently these non-enzymatic modifications are often considered as indicators of stress and disease. Certain non-enzymatic post-translational modifications, however, can be reversed, which provides an additional layer of regulation and renders these modifications suitable for controlling a diverse set of cellular processes ranging from signaling to metabolism. Here we summarize recent examples of irreversible and reversible non-enzymatic modifications, with an emphasis on the latter category. We use two examples, lysine glutarylation and pyrophosphorylation, to highlight principles of the regulation of reversible non-enzymatic post-translational modifications in more detail. Overall, a picture emerges that goes well beyond nonspecific chemical reactions and cellular damage, and instead portrays multifaceted functions of non-enzymatic post-translational modifications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Walsh, C.T., Garneau-Tsodikova, S. & Gatto, G.J. Jr. Protein posttranslational modifications: the chemistry of proteome diversifications. Angew. Chem. Int. Ed. Engl. 44, 7342–7372 (2005).
UniProt Consortium. Controlled vocabulary of posttranslational modifications (PTM). UniProt http://www.uniprot.org/docs/ptmlist (2017).
Barski, A. et al. High-resolution profiling of histone methylations in the human genome. Cell 129, 823–837 (2007).
Meierhofer, D., Wang, X., Huang, L. & Kaiser, P. Quantitative analysis of global ubiquitination in HeLa cells by mass spectrometry. J. Proteome Res. 7, 4566–4576 (2008).
Keller, M.A., Piedrafita, G. & Ralser, M. The widespread role of non-enzymatic reactions in cellular metabolism. Curr. Opin. Biotechnol. 34, 153–161 (2015).
Ubersax, J.A. & Ferrell, J.E. Jr. Mechanisms of specificity in protein phosphorylation. Nat. Rev. Mol. Cell Biol. 8, 530–541 (2007).
Manning, G., Whyte, D.B., Martinez, R., Hunter, T. & Sudarsanam, S. The protein kinase complement of the human genome. Science 298, 1912–1934 (2002).
Bretón-Romero, R. & Lamas, S. Hydrogen peroxide signaling in vascular endothelial cells. Redox Biol. 2, 529–534 (2014).
Claiborne, A. et al. Protein-sulfenic acids: diverse roles for an unlikely player in enzyme catalysis and redox regulation. Biochemistry 38, 15407–15416 (1999).
Ahmed, N. Advanced glycation endproducts—role in pathology of diabetic complications. Diabetes Res. Clin. Pract. 67, 3–21 (2005).
Dickinson, D.A. & Forman, H.J.A.Y. Glutathione in defense and signaling: lessons from a small thiol. Ann. NY Acad. Sci. 973, 488–504 (2002).
Forman, H.J., Ursini, F. & Maiorino, M. An overview of mechanisms of redox signaling. J. Mol. Cell. Cardiol. 73, 2–9 (2014).
Jaisson, S. & Gillery, P. Evaluation of nonenzymatic posttranslational modification-derived products as biomarkers of molecular aging of proteins. Clin. Chem. 56, 1401–1412 (2010).
Ray, P.D., Huang, B.-W. & Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell. Signal. 24, 981–990 (2012).
Stadtman, E.R. Protein oxidation and aging. Free Radic. Res. 40, 1250–1258 (2006).
Singh, R., Barden, A., Mori, T. & Beilin, L. Advanced glycation end-products: a review. Diabetologia 44, 129–146 (2001).
Verbrugge, F.H., Tang, W.H.W. & Hazen, S.L. Protein carbamylation and cardiovascular disease. Kidney Int. 88, 474–478 (2015).
Dalle-Donne, I., Giustarini, D., Colombo, R., Rossi, R. & Milzani, A. Protein carbonylation in human diseases. Trends Mol. Med. 9, 169–176 (2003).
Frizzell, N., Lima, M. & Baynes, J.W. Succination of proteins in diabetes. Free Radic. Res. 45, 101–109 (2011).
Kalim, S., Karumanchi, S.A., Thadhani, R.I. & Berg, A.H. Protein carbamylation in kidney disease: pathogenesis and clinical implications. Am. J. Kidney Dis. 64, 793–803 (2014).
Baynes, J.W. & Thorpe, S.R. Role of oxidative stress in diabetic complications: a new perspective on an old paradigm. Diabetes 48, 1–9 (1999).
Ciolino, H.P. & Levine, R.L. Modification of proteins in endothelial cell death during oxidative stress. Free Radic. Biol. Med. 22, 1277–1282 (1997).
Dickinson, B.C. & Chang, C.J. Chemistry and biology of reactive oxygen species in signaling or stress responses. Nat. Chem. Biol. 7, 504–511 (2011).
Madian, A.G. & Regnier, F.E. Proteomic identification of carbonylated proteins and their oxidation sites. J. Proteome Res. 9, 3766–3780 (2010).
Mirzaei, H. & Regnier, F. Creation of allotypic active sites during oxidative stress. J. Proteome Res. 5, 2159–2168 (2006).
Thomson, E. et al. Identifying peroxidases and their oxidants in the early pathology of cystic fibrosis. Free Radic. Biol. Med. 49, 1354–1360 (2010).
Hartley, D.P., Kroll, D.J. & Petersen, D.R. Prooxidant-initiated lipid peroxidation in isolated rat hepatocytes: detection of 4-hydroxynonenal- and malondialdehyde-protein adducts. Chem. Res. Toxicol. 10, 895–905 (1997).
Rahman, I. et al. 4-Hydroxy-2-nonenal, a specific lipid peroxidation product, is elevated in lungs of patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 166, 490–495 (2002).
Aksenov, M.Y., Aksenova, M.V., Butterfield, D.A., Geddes, J.W. & Markesbery, W.R. Protein oxidation in the brain in Alzheimer's disease. Neuroscience 103, 373–383 (2001).
Fu, M.-X. et al. The advanced glycation end product, Ne-(carboxymethyl)lysine, is a product of both lipid peroxidation and glycoxidation reactions. J. Biol. Chem. 271, 9982–9986 (1996).
Lo, T.W., Westwood, M.E., McLellan, A.C., Selwood, T. & Thornalley, P.J. Binding and modification of proteins by methylglyoxal under physiological conditions. A kinetic and mechanistic study with N α-acetylarginine, N α-acetylcysteine, and N α-acetyllysine, and bovine serum albumin. J. Biol. Chem. 269, 32299–32305 (1994).
Manini, P., La Pietra, P., Panzella, L., Napolitano, A. & d'Ischia, M. Glyoxal formation by Fenton-induced degradation of carbohydrates and related compounds. Carbohydr. Res. 341, 1828–1833 (2006).
Glomb, M.A. & Monnier, V.M. Mechanism of protein modification by glyoxal and glycolaldehyde, reactive intermediates of the Maillard reaction. J. Biol. Chem. 270, 10017–10026 (1995).
Thornalley, P.J. et al. Quantitative screening of advanced glycation endproducts in cellular and extracellular proteins by tandem mass spectrometry. Biochem. J. 375, 581–592 (2003).
Dyer, D.G. et al. Accumulation of Maillard reaction products in skin collagen in diabetes and aging. J. Clin. Invest. 91, 2463–2469 (1993).
Alderson, N.L. et al. S-(2-Succinyl)cysteine: a novel chemical modification of tissue proteins by a Krebs cycle intermediate. Arch. Biochem. Biophys. 450, 1–8 (2006).
Blatnik, M., Thorpe, S.R. & Baynes, J.W. Succination of proteins by fumarate: mechanism of inactivation of glyceraldehyde-3-phosphate dehydrogenase in diabetes. Ann. NY Acad. Sci. 1126, 272–275 (2008).
Adam, J., Yang, M., Soga, T. & Pollard, P.J. Rare insights into cancer biology. Oncogene 33, 2547–2556 (2014).
Bardella, C. et al. Aberrant succination of proteins in fumarate hydratase-deficient mice and HLRCC patients is a robust biomarker of mutation status. J. Pathol. 225, 4–11 (2011).
Adam, J. et al. Renal cyst formation in Fh1-deficient mice is independent of the Hif/Phd pathway: roles for fumarate in KEAP1 succination and Nrf2 signaling. Cancer Cell 20, 524–537 (2011).
Ohta, T. et al. Loss of Keap1 function activates Nrf2 and provides advantages for lung cancer cell growth. Cancer Res. 68, 1303–1309 (2008).
Kraus, L.M. & Kraus, A.P. Jr. Carbamoylation of amino acids and proteins in uremia. Kidney Int. Suppl. 78, S102–S107 (2001).
Wang, Z. et al. Protein carbamylation links inflammation, smoking, uremia and atherogenesis. Nat. Med. 13, 1176–1184 (2007).
Dalle-Donne, I. et al. Protein carbonylation, cellular dysfunction, and disease progression. J. Cell. Mol. Med. 10, 389–406 (2006).
Koeck, T. et al. Rapid and selective oxygen-regulated protein tyrosine denitration and nitration in mitochondria. J. Biol. Chem. 279, 27257–27262 (2004).
Aulak, K.S., Koeck, T., Crabb, J.W. & Stuehr, D.J. Dynamics of protein nitration in cells and mitochondria. Am. J. Physiol. Heart Circ. Physiol. 286, H30–H38 (2004).
Sengupta, S. & Bhattacharjee, A. Dynamics of protein tyrosine nitration and denitration. RE:view 1, 1–13 (2016).
Deeb, R.S. et al. Characterization of a cellular denitrase activity that reverses nitration of cyclooxygenase. Am. J. Physiol. Heart Circ. Physiol. 305, H687–H698 (2013).
Kim, H.-Y. & Gladyshev, V.N. Methionine sulfoxide reduction in mammals: characterization of methionine-R-sulfoxide reductases. Mol. Biol. Cell 15, 1055–1064 (2004).
Drazic, A. & Winter, J. The physiological role of reversible methionine oxidation. Biochim. Biophys. Acta 1844, 1367–1382 (2014).
Kaya, A., Lee, B.C. & Gladyshev, V.N. Regulation of protein function by reversible methionine oxidation and the role of selenoprotein MsrB1. Antioxid. Redox Signal. 23, 814–822 (2015).
Drazic, A. et al. Methionine oxidation activates a transcription factor in response to oxidative stress. Proc. Natl. Acad. Sci. USA 110, 9493–9498 (2013).
Paulsen, C.E. & Carroll, K.S. Cysteine-mediated redox signaling: chemistry, biology, and tools for discovery. Chem. Rev. 113, 4633–4679 (2013).
Chung, H.S., Wang, S.-B., Venkatraman, V., Murray, C.I. & Van Eyk, J.E. Cysteine oxidative posttranslational modifications: emerging regulation in the cardiovascular system. Circ. Res. 112, 382–392 (2013).
Buckley, D.A., Cheng, A., Kiely, P.A., Tremblay, M.L. & O'Connor, R. Regulation of insulin-like growth factor type I (IGF-I) receptor kinase activity by protein tyrosine phosphatase 1B (PTP-1B) and enhanced IGF-I-mediated suppression of apoptosis and motility in PTP-1B-deficient fibroblasts. Mol. Cell. Biol. 22, 1998–2010 (2002).
Mahadev, K., Zilbering, A., Zhu, L. & Goldstein, B.J. Insulin-stimulated hydrogen peroxide reversibly inhibits protein-tyrosine phosphatase 1b in vivo and enhances the early insulin action cascade. J. Biol. Chem. 276, 21938–21942 (2001).
Lee, S.-R., Kwon, K.-S., Kim, S.-R. & Rhee, S.G. Reversible inactivation of protein-tyrosine phosphatase 1B in A431 cells stimulated with epidermal growth factor. J. Biol. Chem. 273, 15366–15372 (1998).
Salmeen, A. et al. Redox regulation of protein tyrosine phosphatase 1B involves a sulphenyl-amide intermediate. Nature 423, 769–773 (2003).
van Montfort, R.L.M., Congreve, M., Tisi, D., Carr, R. & Jhoti, H. Oxidation state of the active-site cysteine in protein tyrosine phosphatase 1B. Nature 423, 773–777 (2003).
Denu, J.M. & Tanner, K.G. Specific and reversible inactivation of protein tyrosine phosphatases by hydrogen peroxide: evidence for a sulfenic acid intermediate and implications for redox regulation. Biochemistry 37, 5633–5642 (1998).
Moellering, R.E. & Cravatt, B.F. Functional lysine modification by an intrinsically reactive primary glycolytic metabolite. Science 341, 549–553 (2013).
Lin, H., Su, X. & He, B. Protein lysine acylation and cysteine succination by intermediates of energy metabolism. ACS Chem. Biol. 7, 947–960 (2012).
Zhang, Z. et al. Identification of lysine succinylation as a new post-translational modification. Nat. Chem. Biol. 7, 58–63 (2011).
Baeza, J., Smallegan, M.J. & Denu, J.M. Site-specific reactivity of nonenzymatic lysine acetylation. ACS Chem. Biol. 10, 122–128 (2015).
Kulkarni, R.A. et al. Discovering targets of non-enzymatic acylation by thioester reactivity profiling. Cell Chem. Biol. 24, 231–242 (2017).
Wagner, G.R. & Hirschey, M.D. Nonenzymatic protein acylation as a carbon stress regulated by sirtuin deacylases. Mol. Cell 54, 5–16 (2014).
Tan, M. et al. Lysine glutarylation is a protein posttranslational modification regulated by SIRT5. Cell Metab. 19, 605–617 (2014).
Du, J. et al. Sirt5 is a NAD-dependent protein lysine demalonylase and desuccinylase. Science 334, 806–809 (2011).
Wagner, G.R. & Payne, R.M. Widespread and enzyme-independent Ne-acetylation and Ne-succinylation of proteins in the chemical conditions of the mitochondrial matrix. J. Biol. Chem. 288, 29036–29045 (2013).
Wagner, G.R. et al. A class of reactive acyl-CoA species reveals the non-enzymatic origins of protein acylation. Cell Metab. 25, 823–837.e8 (2017).
McCleary, W.R., Stock, J.B. & Ninfa, A.J. Is acetyl phosphate a global signal in Escherichia coli? J. Bacteriol. 175, 2793–2798 (1993).
Klein, A.H., Shulla, A., Reimann, S.A., Keating, D.H. & Wolfe, A.J. The intracellular concentration of acetyl phosphate in Escherichia coli is sufficient for direct phosphorylation of two-component response regulators. J. Bacteriol. 189, 5574–5581 (2007).
Azevedo, C., Livermore, T. & Saiardi, A. Protein polyphosphorylation of lysine residues by inorganic polyphosphate. Mol. Cell 58, 71–82 (2015).
Chakraborty, A. et al. Inositol pyrophosphates inhibit Akt signaling, thereby regulating insulin sensitivity and weight gain. Cell 143, 897–910 (2010).
Saiardi, A., Resnick, A.C., Snowman, A.M., Wendland, B. & Snyder, S.H. Inositol pyrophosphates regulate cell death and telomere length through phosphoinositide 3-kinase-related protein kinases. Proc. Natl. Acad. Sci. USA 102, 1911–1914 (2005).
Azevedo, C., Burton, A., Ruiz-Mateos, E., Marsh, M. & Saiardi, A. Inositol pyrophosphate mediated pyrophosphorylation of AP3B1 regulates HIV-1 Gag release. Proc. Natl. Acad. Sci. USA 106, 21161–21166 (2009).
Chanduri, M. et al. Inositol hexakisphosphate kinase 1 (IP6K1) activity is required for cytoplasmic dynein-driven transport. Biochem. J. 473, 3031–3047 (2016).
Penkert, M. et al. Unambiguous identification of serine and threonine pyrophosphorylation using neutral-loss-triggered electron-transfer/higher-energy collision dissociation. Anal. Chem. 89, 3672–3680 (2017).
Saiardi, A., Bhandari, R., Resnick, A.C., Snowman, A.M. & Snyder, S.H. Phosphorylation of proteins by inositol pyrophosphates. Science 306, 2101–2105 (2004).
Bhandari, R. et al. Protein pyrophosphorylation by inositol pyrophosphates is a posttranslational event. Proc. Natl. Acad. Sci. USA 104, 15305–15310 (2007).
Wu, M. et al. Elucidating diphosphoinositol polyphosphate function with nonhydrolyzable analogues. Angew. Chem. Int. Ed. Engl. 53, 7192–7197 (2014).
Hager, A. et al. Cellular cations control conformational switching of inositol pyrophosphate analogues. Chemistry 22, 12406–12414 (2016).
Yates, L.M. & Fiedler, D. Establishing the stability and reversibility of protein pyrophosphorylation with synthetic peptides. ChemBioChem 16, 415–423 (2015).
Beltrao, P., Bork, P., Krogan, N.J. & van Noort, V. Evolution and functional cross-talk of protein post-translational modifications. Mol. Syst. Biol. 9, 714 (2013).
Yoshida, T. et al. Nitric oxide activates TRP channels by cysteine S-nitrosylation. Nat. Chem. Biol. 2, 596–607 (2006).
Mustafa, A.K. et al. H2S signals through protein S-sulfhydration. Sci. Signal. 2, ra72 (2009).
Kathayat, R.S. et al. Active and dynamic mitochondrial S-depalmitoylation revealed by targeted fluorescent probes. Nat. Commun. 9, 334 (2018).
Acknowledgements
R.H. gratefully acknowledges funding from the Leibniz-Gemeinschaft (SAW-2017-FMP-1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Harmel, R., Fiedler, D. Features and regulation of non-enzymatic post-translational modifications. Nat Chem Biol 14, 244–252 (2018). https://doi.org/10.1038/nchembio.2575
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2575
This article is cited by
-
Acylspermidines are conserved mitochondrial sirtuin-dependent metabolites
Nature Chemical Biology (2024)
-
GnT-V-mediated aberrant N-glycosylation of TIMP-1 promotes diabetic retinopathy progression
Molecular Biology Reports (2024)
-
Functions, Mechanisms, and therapeutic applications of the inositol pyrophosphates 5PP-InsP5 and InsP8 in mammalian cells
Journal of Cardiovascular Translational Research (2024)
-
Roles of protein post-translational modifications in glucose and lipid metabolism: mechanisms and perspectives
Molecular Medicine (2023)
-
Association between diabetes and cancer. Current mechanistic insights into the association and future challenges
Molecular and Cellular Biochemistry (2023)