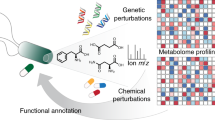
Abstract
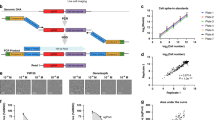
Chemical-genetic approaches offer the potential for unbiased functional annotation of chemical libraries. Mutations can alter the response of cells in the presence of a compound, revealing chemical-genetic interactions that can elucidate a compound's mode of action. We developed a highly parallel, unbiased yeast chemical-genetic screening system involving three key components. First, in a drug-sensitive genetic background, we constructed an optimized diagnostic mutant collection that is predictive for all major yeast biological processes. Second, we implemented a multiplexed (768-plex) barcode-sequencing protocol, enabling the assembly of thousands of chemical-genetic profiles. Finally, based on comparison of the chemical-genetic profiles with a compendium of genome-wide genetic interaction profiles, we predicted compound functionality. Applying this high-throughput approach, we screened seven different compound libraries and annotated their functional diversity. We further validated biological process predictions, prioritized a diverse set of compounds, and identified compounds that appear to have dual modes of action.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
07 August 2017
In the version of this article initially published online, there were several typographical errors introducing scientific inaccuracies. In Figure 5a, the compound name NPE1136 was incorrectly written NPD1136. In the Discussion, the number of strains tested, 5,000, was incorrectly given as ∼4,900. In the Online Methods, several strain descriptions in the sections "Constructing a genome-wide drug sensitive yeast deletion correction" and "Assessing compound hit rate of sensitized yeast strains" were incorrect or unclear (in particular, the MATa xxxΔ::kanMX yeast strain was indicated as MATa xxxΔ:kanMX); the unit mL was given in place of the correct μL in several places in the sections "Multi-parameter validation of cell wall targeting compounds" and "Zymolyase sensitivity assay"; references 14 and 15 were cited instead of references 8 and 17 in the section "Comparison with other chemical-genetic data sets section"; and the list of molecular descriptors calculated using PaDEL-Descriptor in the section "Computing molecular descriptors for all screened compounds" should have started with column L, not J. These errors have been corrected in all versions of the article.
04 October 2017
In the version of this article initially published, the artemisinin structure in Figure 4b was shown with incorrect stereochemistry. The error has been corrected in the HTML and PDF versions of the article.
References
Geysen, H.M., Schoenen, F., Wagner, D. & Wagner, R. Combinatorial compound libraries for drug discovery: an ongoing challenge. Nat. Rev. Drug Discov. 2, 222–230 (2003).
Roemer, T. & Boone, C. Systems-level antimicrobial drug and drug synergy discovery. Nat. Chem. Biol. 9, 222–231 (2013).
Kepp, O., Galluzzi, L., Lipinski, M., Yuan, J. & Kroemer, G. Cell death assays for drug discovery. Nat. Rev. Drug Discov. 10, 221–237 (2011).
Clemons, P.A. Complex phenotypic assays in high-throughput screening. Curr. Opin. Chem. Biol. 8, 334–338 (2004).
Sundberg, S.A. High-throughput and ultra-high-throughput screening: solution- and cell-based approaches. Curr. Opin. Biotechnol. 11, 47–53 (2000).
Giaever, G. et al. Chemogenomic profiling: identifying the functional interactions of small molecules in yeast. Proc. Natl. Acad. Sci. USA 101, 793–798 (2004).
Parsons, A.B. et al. Exploring the mode-of-action of bioactive compounds by chemical-genetic profiling in yeast. Cell 126, 611–625 (2006).
Lee, A.Y. et al. Mapping the cellular response to small molecules using chemogenomic fitness signatures. Science 344, 208–211 (2014).
Parsons, A.B. et al. Integration of chemical-genetic and genetic interaction data links bioactive compounds to cellular target pathways. Nat. Biotechnol. 22, 62–69 (2004).
Giaever, G. et al. Functional profiling of the Saccharomyces cerevisiae genome. Nature 418, 387–391 (2002).
Costanzo, M. et al. A global genetic interaction network maps a wiring diagram of cellular function. Science 353, aaf1420 (2016).
Costanzo, M. et al. The genetic landscape of a cell. Science 327, 425–431 (2010).
Smith, A.M. et al. Highly-multiplexed barcode sequencing: an efficient method for parallel analysis of pooled samples. Nucleic Acids Res. 38, e142 (2010).
Rogers, B. et al. The pleitropic drug ABC transporters from Saccharomyces cerevisiae. J. Mol. Microbiol. Biotechnol. 3, 207–214 (2001).
Andrusiak, K. Adapting S. cerevisiae Chemical Genomics for Identifying the Modes of Action of Natural Compounds (Master's thesis, University of Toronto, 2012).
Bokulich, N.A. et al. Quality-filtering vastly improves diversity estimates from Illumina amplicon sequencing. Nat. Methods 10, 57–59 (2013).
Hoepfner, D. et al. High-resolution chemical dissection of a model eukaryote reveals targets, pathways and gene functions. Microbiol. Res. 169, 107–120 (2014).
Baryshnikova, A. Systematic functional annotation and visualization of biological networks. Cell Syst. 2, 412–421 (2016).
Vancutsem, P.M. & Babish, J.G. In vitro and in vivo study of the effects of enrofloxacin on hepatic cytochrome P-450. Potential for drug interactions. Vet. Hum. Toxicol. 38, 254–259 (1996).
Desta, Z., Soukhova, N., Mahal, S.K. & Flockhart, D.A. Interaction of cisapride with the human cytochrome P450 system: metabolism and inhibition studies. Drug Metab. Dispos. 28, 789–800 (2000).
Jeong, S., Nguyen, P.D. & Desta, Z. Comprehensive in vitro analysis of voriconazole inhibition of eight cytochrome P450 (CYP) enzymes: major effect on CYPs 2B6, 2C9, 2C19, and 3A. Antimicrob. Agents Chemother. 53, 541–551 (2009).
Abdel-Rahman, S.M. et al. Potent inhibition of cytochrome P-450 2D6-mediated dextromethorphan O-demethylation by terbinafine. Drug Metab. Dispos. 27, 770–775 (1999).
Laugesen, S., Enggaard, T.P., Pedersen, R.S., Sindrup, S.H. & Brøsen, K. Paroxetine, a cytochrome P450 2D6 inhibitor, diminishes the stereoselective O-demethylation and reduces the hypoalgesic effect of tramadol. Clin. Pharmacol. Ther. 77, 312–323 (2005).
Dresser, G.K., Spence, J.D. & Bailey, D.G. Pharmacokinetic-pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition. Clin. Pharmacokinet. 38, 41–57 (2000).
Dranchak, P. et al. Profile of the GSK published protein kinase inhibitor set across ATP-dependent and-independent luciferases: implications for reporter-gene assays. PLoS One 8, e57888 (2013).
Bamford, M.J. et al. (1H-imidazo[4,5-c]pyridin-2-yl)-1,2,5-oxadiazol-3-ylamine derivatives: a novel class of potent MSK-1-inhibitors. Bioorg. Med. Chem. Lett. 15, 3402–3406 (2005).
Wang, X., Wang, R., Zhang, Y. & Zhang, H. Evolutionary survey of druggable protein targets with respect to their subcellular localizations. Genome Biol. Evol. 5, 1291–1297 (2013).
O'Neill, P.M., Barton, V.E. & Ward, S.A. The molecular mechanism of action of artemisinin--the debate continues. Molecules 15, 1705–1721 (2010).
Li, W. et al. Yeast model uncovers dual roles of mitochondria in action of artemisinin. PLoS Genet. 1, e36 (2005).
Steinbrück, L., Pereira, G. & Efferth, T. Effects of artesunate on cytokinesis and G/M cell cycle progression of tumour cells and budding yeast. Cancer Genomics Proteomics 7, 337–346 (2010).
Disbrow, G.L. et al. Dihydroartemisinin is cytotoxic to papillomavirus-expressing epithelial cells in vitro and in vivo. Cancer Res. 65, 10854–10861 (2005).
Li, Y. et al. Novel antitumor artemisinin derivatives targeting G1 phase of the cell cycle. Bioorg. Med. Chem. Lett. 11, 5–8 (2001).
Goodrich, S.K., Schlegel, C.R., Wang, G. & Belinson, J.L. Use of artemisinin and its derivatives to treat HPV-infected/transformed cells and cervical cancer: a review. Future Oncol. 10, 647–654 (2014).
Xia, W. et al. Photo-activated psoralen binds the ErbB2 catalytic kinase domain, blocking ErbB2 signaling and triggering tumor cell apoptosis. PLoS One 9, e88983 (2014).
Yu, L., Peña Castillo, L., Mnaimneh, S., Hughes, T.R. & Brown, G.W. A survey of essential gene function in the yeast cell division cycle. Mol. Biol. Cell 17, 4736–4747 (2006).
Piotrowski, J.S. et al. Plant-derived antifungal agent poacic acid targets β-1,3-glucan. Proc. Natl. Acad. Sci. USA 112, E1490–E1497 (2015).
Okada, H., Ohnuki, S., Roncero, C., Konopka, J.B. & Ohya, Y. Distinct roles of cell wall biogenesis in yeast morphogenesis as revealed by multivariate analysis of high-dimensional morphometric data. Mol. Biol. Cell 25, 222–233 (2014).
Peters, J.-U., Schnider, P., Mattei, P. & Kansy, M. Pharmacological promiscuity: dependence on compound properties and target specificity in a set of recent Roche compounds. ChemMedChem 4, 680–686 (2009).
Medina-Franco, J.L., Giulianotti, M.A., Welmaker, G.S. & Houghten, R.A. Shifting from the single to the multitarget paradigm in drug discovery. Drug Discov. Today 18, 495–501 (2013).
Grollman, A.P. & Takeshita, M. Interactions of bleomycin with DNA. Adv. Enzyme Regul. 18, 67–83 (1980).
Hay, J., Shahzeidi, S. & Laurent, G. Mechanisms of bleomycin-induced lung damage. Arch. Toxicol. 65, 81–94 (1991).
Moore, C.W., Del Valle, R., McKoy, J., Pramanik, A. & Gordon, R.E. Lesions and preferential initial localization of [S-methyl-3H]bleomycin A2 on Saccharomyces cerevisiae cell walls and membranes. Antimicrob. Agents Chemother. 36, 2497–2505 (1992).
Poddevin, B., Orlowski, S., Belehradek, J. Jr. & Mir, L.M. Very high cytotoxicity of bleomycin introduced into the cytosol of cells in culture. Biochem. Pharmacol. 42, (Suppl.), S67–S75 (1991).
Sun, I.L. & Crane, F.L. Bleomycin control of transplasma membrane redox activity and proton movement in HeLa cells. Biochem. Pharmacol. 34, 617–622 (1985).
Ekimoto, H., Takahashi, K., Matsuda, A., Takita, T. & Umezawa, H. Lipid peroxidation by bleomycin-iron complexes in vitro. J. Antibiot. (Tokyo) 38, 1077–1082 (1985).
Kanofsky, J.R. Singlet oxygen production by bleomycin. A comparison with heme-containing compounds. J. Biol. Chem. 261, 13546–13550 (1986).
Sidorova, J.M. & Breeden, L.L. Precocious G1/S transitions and genomic instability: the origin connection. Mutat. Res. 532, 5–19 (2003).
Mahé, Y., Lemoine, Y. & Kuchler, K. The ATP binding cassette transporters Pdr5 and Snq2 of Saccharomyces cerevisiae can mediate transport of steroids in vivo. J. Biol. Chem. 271, 25167–25172 (1996).
Hart, T. et al. High-resolution CRISPR screens reveal fitness genes and genotype-specific cancer liabilities. Cell 163, 1515–1526 (2015).
Myers, C.L., Barrett, D.R., Hibbs, M.A., Huttenhower, C. & Troyanskaya, O.G. Finding function: evaluation methods for functional genomic data. BMC Genomics 7, 187 (2006).
Deshpande, R. et al. Efficient strategies for screening large-scale genetic interaction networks. Preprint at http://www.biorxiv.org/content/early/2017/07/05/159632 (2017).
Kato, N., Takahashi, S., Nogawa, T., Saito, T. & Osada, H. Construction of a microbial natural product library for chemical biology studies. Curr. Opin. Chem. Biol. 16, 101–108 (2012).
O'Boyle, N.M. et al. Open Babel: An open chemical toolbox. J. Cheminform. 3, 33 (2011).
O'Boyle, N.M., Morley, C. & Hutchison, G.R. Pybel: a Python wrapper for the OpenBabel cheminformatics toolkit. Chem. Cent. J. 2, 5 (2008).
Yap, C.W. PaDEL-descriptor: an open source software to calculate molecular descriptors and fingerprints. J. Comput. Chem. 32, 1466–1474 (2011).
Steinbeck, C. et al. The Chemistry Development Kit (CDK): an open-source Java library for chemo- and bioinformatics. J. Chem. Inf. Comput. Sci. 43, 493–500 (2003).
Borgelt, C., Meinl, T. & Berthold, M. MoSS: A program for molecular substructure mining. In Proc. 1st International Workshop on Open Source Data Mining: Frequent Pattern Mining Implementations 6–15 (ACM, 2005).
Huh, W.-K. et al. Global analysis of protein localization in budding yeast. Nature 425, 686–691 (2003).
Koh, J.L.Y. et al. CYCLoPs: A comprehensive database constructed from automated analysis of protein abundance and subcellular localization patterns in Saccharomyces cerevisiae. G3 (Bethesda) 5, 1223–1232 (2015).
Ohya, Y. et al. High-dimensional and large-scale phenotyping of yeast mutants. Proc. Natl. Acad. Sci. USA 102, 19015–19020 (2005).
Acknowledgements
This work was supported by RIKEN Strategic Programs for R&D. J.S.P. and S.C.L. were funded by a RIKEN Foreign Postdoctoral Fellowship. S.W.S. is supported by an NSF Graduate Research Fellowship (00039202), an NIH Biotechnology training grant (T32GM008347), and a one-year BICB fellowship from the University of Minnesota. H.O. is a research fellow of the Japan Society for the Promotion of Science (JSPS). R.D., J.N., E.W., and C.L.M. are supported by National Institutes of Health Grants 1R01HG005084-01A1, 1R01GM104975-01, and R01HG005853 and National Science Foundation Grant DBI 0953881. C.B. and Y.O. are supported by JSPS KAKENHI Grant Numbers 15H04483. C.B. and B.A. were supported by the Canadian Institutes of Health Research, grants FDN-143264 and FDN-143265, respectively. C.L.M., C.B., M.C., J.L., and B.A. are supported by the Canadian Institute for Advanced Research Genetic Networks Program. Y.O. is supported by Ministry of Education, Culture, Sports, Science and Technology, Japan Grant for Scientific Research 24370002 and JSPS KAKENHI Grant Numbers 15H04402. M. Yoshida is supported by JSPS KAKENHI Grant Number 26221204. A.B. is supported by a Lewis Sigler fellowship at Princeton University. G.W.B. and N.P.T. are supported by Canadian Cancer Society Research Institute impact grant 702310. K.S. is supported by The Core Research for Evolutional Science and Technology (CREST) from the Japan Science and Technology Agency (JST). We thank Astellas Pharma, Inc. (Tokyo, Japan) for their kind gift of micafungin. We thank T. Saito for help with NPDepo compound access. Sequencing was provided by RIKEN Center for Life Science Technologies, Division of Genomic Technologies, Genome Network Analysis Support Facility (GeNAS) RIKEN CLST and the University of Chicago.
Author information
Authors and Affiliations
Contributions
C.B., M. Yoshida, C.L.M., J.S.P. and S.C.L. conceived the project. J.S.P., S.C.L., R.D., and S.W.S. designed the chemical genomic screens. J.S.P., S.C.L., J.M.B., R.O., M. Yoshimura, Y.Y., E. D.-A. performed the chemical genomic experiments. C.L.M., R.D., S.W.S., J.N., H.S., and E.W. designed the analysis software and performed analysis. G.W.B., N.P.T., and S.C.L. performed cell cycle experiments. J.P., K.A., M.A.L., and A.B. designed the sensitized yeast. Y.O., H.O., A.A.G., K.K. performed validation of cell wall targeting compounds. R.D. and M.C. designed the diagnostic mutant collection. H.O. and H.H. provided and curated NPDepo compounds. K.S. and B.A. provided analysis. J.v.L. edited the figures of the manuscript. The manuscript was written by J.S.P., S.C.L., C.L.M., and C.B. with input and editing from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Figures 1–15 and Supplementary Tables 1–3 (PDF 2375 kb)
Supplementary Note
Supplementary Note (PDF 638 kb)
Supplementary Dataset 1
Number of compounds in each collection causing >20% inhibition of growth against the yeast screening pool. (XLSX 18 kb)
Supplementary Dataset 2
Diagnostic screening pool deletion mutant strain members. (XLSX 160 kb)
Supplementary Dataset 3
768 multiplex tag primers. (XLS 149 kb)
Supplementary Dataset 4
Names, structures, doses, and chemical properties of screened compounds. (XLSX 4002 kb)
Supplementary Dataset 5
Average chemical genomic profile of 11,983 compounds. (XLSX 33280 kb)
Supplementary Dataset 6
Average chemical genomic profile of 1,522 high-confidence compounds. (XLSX 4207 kb)
Supplementary Dataset 7
Number of positive (CG score ≥+2.5), negative (CG score ≤−2.5), and all chemical genetic interactions per strain for all screened compounds. (XLSX 21 kb)
Supplementary Dataset 8
Compounds passing our false discovery rate filter (high-confidence set) with top 3 GO process predictions, bioprocess predictions, and driver genes. (XLSX 892 kb)
Supplementary Dataset 9
Interaction degree (positive and negative interactions) of the 275 strains from the RIKEN subset of the data also present in the S. cerevisiae genetic interaction array. Strains classified as high degree were removed to obtain “no-high-degree” process-level predictions, as were strains classified as low degree to obtain “no-low-degree” predictions. (XLSX 60 kb)
Supplementary Dataset 10
Comparison of the compounds, and their predictions, discovered with high confidence using all, only negative, and only positive chemical-genetic interactions from the RIKEN subset of the data. Each “Negative-all” and “Positive-all” profile contained all interactions with scores below or above zero, respectively, while the pair of “Negative-equal” and “Positive-equal” profiles for each compound contained an equal number of negative and positive interactions, respectively, above a score magnitude cutoff of 1. (XLSX 61 kb)
Supplementary Dataset 11
Growth inhibition by 24 bioactive compounds (10 μg/mL) of the screening pool versus the gtr1Δ deletion mutant (MATα pdr1Δ::natMX pdr3Δ::KI.URA3 snq2Δ::KI.LEU2 gtr1Δ::kanMX can1Δ::STE2pr-Sp_his5 lyp1Δ his3Δ1 leu2Δ0 ura3Δ0 met15Δ0). (XLSX 9 kb)
Supplementary Dataset 12
Bioprocesses and associated GO annotation for compounds of the NIH-CC. (XLSX 27 kb)
Supplementary Dataset 13
Functional enrichments and known targets of the GSK kinase inhibitor collection. (XLS 58 kb)
Supplementary Dataset 14
Cellular compartments with enrichment or depletion of targets across compound collections. (XLSX 33 kb)
Supplementary Dataset 15
Functional enrichments among substructures. “All compounds with substructure” shows the compounds containing the substructure for that given row, and “hit compounds with substructure” shows the subset of those compounds that is annotated to any of the GO terms in that row. Substructures can be visualized directly at mosaic.cs.umn.edu/RIKEN_substructures.html or indirectly by copy-and-pasting the semicolon-delimited compound sets into the search bar at mosaic.cs.umn.edu. (XLSX 3503 kb)
Supplementary Dataset 16
Validation of predicted biological process targets of 67 compounds using cell cycle analysis. (XLSX 13 kb)
Supplementary Dataset 17
Validation of cell wall–targeting compounds. (XLSX 14 kb)
Supplementary Dataset 18
Prioritized functional diversity set from public collections screened. (XLSX 15 kb)
Supplementary Dataset 19
Prioritized compound sets for 17 bioprocesses. Each high-confidence compound was placed into the group corresponding to its top bioprocess prediction in Supplementary Table 7. (XLSX 542 kb)
Supplementary Dataset 20
Number of high confidence compounds per bioprocess, and enrichments/depletions across the tested compound collections. (XLSX 16 kb)
Supplementary Dataset 21
Proportional differences in GO annotations versus background expectation for each library/group of compounds. (XLSX 641 kb)
Supplementary Dataset 22
Mapping of Lee et al. major chemical-genetic signatures to bioprocesses in this study. Pearson correlation coefficients (PCCs) > 0.3 between Lee et al. compounds and high confidence compounds (this study) were mapped to major chemical-genetic signatures (Lee et al.) or bioprocesses (this study) to generate a mapping from each major chemical-genetic signature to the bioprocesses that contained similar compounds. Only the most confident mapping, as determined by relative confidence (the product of the fractions of compounds in each set that contribute to the similarities) is shown. (XLSX 59 kb)
Rights and permissions
About this article
Cite this article
Piotrowski, J., Li, S., Deshpande, R. et al. Functional annotation of chemical libraries across diverse biological processes. Nat Chem Biol 13, 982–993 (2017). https://doi.org/10.1038/nchembio.2436
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2436
This article is cited by
-
From beer to breadboards: yeast as a force for biological innovation
Genome Biology (2024)
-
Mitochondrial genome structure and composition in 70 fishes: a key resource for fisheries management in the South Atlantic
BMC Genomics (2024)
-
A scalable platform for efficient CRISPR-Cas9 chemical-genetic screens of DNA damage-inducing compounds
Scientific Reports (2024)
-
BIONIC: discovering new biology through deep learning-based network integration
Nature Methods (2022)
-
High-throughput platform for yeast morphological profiling predicts the targets of bioactive compounds
npj Systems Biology and Applications (2022)