Abstract
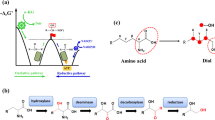
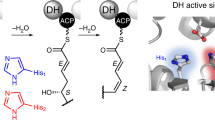
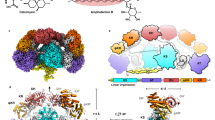
In this study, we engineered fatty acid synthases (FAS) for the biosynthesis of short-chain fatty acids and polyketides, guided by a combined in vitro and in silico approach. Along with exploring the synthetic capability of FAS, we aim to build a foundation for efficient protein engineering, with the specific goal of harnessing evolutionarily related megadalton-scale polyketide synthases (PKS) for the tailored production of bioactive natural compounds.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lynen, F. Methods Enzymol. 14, 17–33 (1969).
Maier, T., Leibundgut, M., Boehringer, D. & Ban, N. Q. Rev. Biophys. 43, 373–422 (2010).
Enderle, M., McCarthy, A., Paithankar, K.S. & Grininger, M. Acta Crystallogr. F Struct. Biol. Commun. 71, 1401–1407 (2015).
Zha, W., Shao, Z., Frost, J.W. & Zhao, H. J. Am. Chem. Soc. 126, 4534–4535 (2004).
Choi, Y.J. & Lee, S.Y. Nature 502, 571–574 (2013).
Peralta-Yahya, P.P., Zhang, F., del Cardayre, S.B. & Keasling, J.D. Nature 488, 320–328 (2012).
Chia, M., Schwartz, T.J., Shanks, B.H. & Dumesic, J.A. Green Chem. 14, 1850–1853 (2012).
Sumper, M., Oesterhelt, D., Riepertinger, C. & Lynen, F. Eur. J. Biochem. 10, 377–387 (1969).
Pirson, W., Schuhmann, L. & Lynen, F. Eur. J. Biochem. 36, 16–24 (1973).
Leibundgut, M., Jenni, S., Frick, C. & Ban, N. Science 316, 288–290 (2007).
Lomakin, I.B., Xiong, Y. & Steitz, T.A. Cell 129, 319–332 (2007).
Johansson, P. et al. Proc. Natl. Acad. Sci. USA. 105, 12803–12808 (2008).
Morisaki, N. et al. Eur. J. Biochem. 211, 111–115 (1993).
Aritomi, K. et al. Biosci. Biotechnol. Biochem. 68, 206–214 (2004).
Christensen, C.E., Kragelund, B.B., von Wettstein-Knowles, P. & Henriksen, A. Protein Sci. 16, 261–272 (2007).
Rangan, V.S. & Smith, S. J. Biol. Chem. 272, 11975–11978 (1997).
Bunkoczi, G. et al. Chem. Biol. 16, 667–675 (2009).
Gillespie, D.T. J. Phys. Chem. 81, 2340–2361 (1977).
Kresze, G.-B., Steber, L., Oesterhelt, D. & Lynen, F. Eur. J. Biochem. 79, 191–199 (1977).
Zhu, Z. et al. Nat. Chem. Biol. 13 http://dx.doi.org/10.1038/nchembio.2301 (2017).
Robbins, T., Liu, Y.-C., Cane, D.E. & Khosla, C. Curr. Opin. Struct. Biol. 41, 10–18 (2016).
Hertweck, C. Angew. Chem. Int. Ed. Engl. 48, 4688–4716 (2009).
Carter, G.T. Nat. Prod. Rep. 28, 1783–1789 (2011).
Cane, D.E., Walsh, C.T. & Khosla, C. Science 282, 63–68 (1998).
Menzella, H.G. et al. Nat. Biotechnol. 23, 1171–1176 (2005).
Zhang, X. et al. Synthesis 5, 749–753 (2007).
Hess, B., Kutzner, C., van der Spoel, D. & Lindahl, E. J. Chem. Theory Comput. 4, 435–447 (2008).
Seeliger, D., Buelens, F.P., Goette, M., de Groot, B.L. & Grubmüller, H. Nucleic Acids Res. 39, 8281–8290 (2011).
Buelens, F.P. & Grubmüller, H. J. Comput. Chem. 33, 25–33 (2012).
Lindorff-Larsen, K. et al. Proteins 78, 1950–1958 (2010).
Wang, J., Wolf, R.M., Caldwell, J.W., Kollman, P.A. & Case, D.A. J. Comput. Chem. 25, 1157–1174 (2004).
Boresch, S., Tettinger, F., Leitgeb, M. & Karplus, M. J. Phys. Chem. B 107, 9535–9551 (2003).
Olsen, J.G., Kadziola, A., von Wettstein-Knowles, P., Siggaard-Andersen, M. & Larsen, S. Structure 9, 233–243 (2001).
Oefner, C., Schulz, H., D'Arcy, A. & Dale, G.E. Acta Crystallogr. D Biol. Crystallogr. 62, 613–618 (2006).
Acknowledgements
We thank A. Matijczak and M. Enderle for starting the project with us by establishing purification of Corynebacterium ammoniagenes FAS and cloning first constructs. M.G. and H.G. thank D. Oesterhelt for continuous support over many years and for initiating the collaboration between H.G. and M.G. This work was supported by a Lichtenberg Grant of the Volkswagen Foundation to M.G. (grant number 85 701), the German Federal Ministry of Education and Research to F.B. (grant number 0315450I), the DFG excellence program CNMBP to F.B. (DFG-EXC 171), and the Max Planck Society (H.G., F.B.).
Author information
Authors and Affiliations
Contributions
J.G., S.S., M.J. and M.G. performed biochemical studies; J.G. and M.G. analyzed biochemical data; N.C. supported MS-based product analysis; F.B. performed kinetic and atomistic modeling; J.G., F.B., H.G. and M.G. designed research; J.G., F.B., H.G. and M.G. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.G. and M.G. are inventors of EP patent applications 15 174 342.4 (filed on June 26th, 2015) and 15 162 192.7 (filed on April 1st, 2015) on FAS mutations for short FA production.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Tables 1–3, Supplementary Figures 1–16 and Supplementary Notes 1–3. (PDF 8370 kb)
Supplementary Software
Modeling implementation for supplementary information. (TAR 10480 kb)
Rights and permissions
About this article
Cite this article
Gajewski, J., Buelens, F., Serdjukow, S. et al. Engineering fatty acid synthases for directed polyketide production. Nat Chem Biol 13, 363–365 (2017). https://doi.org/10.1038/nchembio.2314
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2314
This article is cited by
-
Structural insight into Pichia pastoris fatty acid synthase
Scientific Reports (2021)
-
Fusing α and β subunits of the fungal fatty acid synthase leads to improved production of fatty acids
Scientific Reports (2020)
-
Multidimensional engineering of Saccharomyces cerevisiae for efficient synthesis of medium-chain fatty acids
Nature Catalysis (2020)
-
Gating mechanism of elongating β-ketoacyl-ACP synthases
Nature Communications (2020)
-
Analysis of the co-translational assembly of the fungal fatty acid synthase (FAS)
Scientific Reports (2020)