Abstract
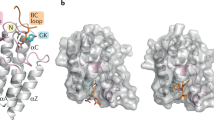
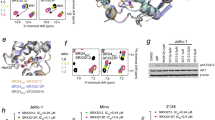
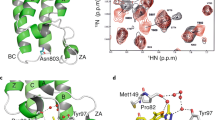
Cellular signaling is often propagated by multivalent interactions. Multivalency creates avidity, allowing stable biophysical recognition. Multivalency is an attractive strategy for achieving potent binding to protein targets, as the affinity of bivalent ligands is often greater than the sum of monovalent affinities. The bromodomain and extraterminal domain (BET) family of transcriptional coactivators features tandem bromodomains through which BET proteins bind acetylated histones and transcription factors. All reported antagonists of the BET protein BRD4 bind in a monovalent fashion. Here we describe, to our knowledge for the first time, a bivalent BET bromodomain inhibitor—MT1—which has unprecedented potency. Biophysical and biochemical studies suggest MT1 is an intramolecular bivalent BRD4 binder that is more than 100-fold more potent, in cellular assays, than the corresponding monovalent antagonist, JQ1. MT1 significantly (P < 0.05) delayed leukemia progression in mice, as compared to JQ1. These data qualify a powerful chemical probe for BET bromodomains and a rationale for further development of multidomain inhibitors of epigenetic reader proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mammen, M., Choi, S.-K. & Whitesides, G.M. Polyvalent interactions in biological systems: implications for design and use of multivalent ligands and inhibitors. Angew. Chem. Int. Ed. Engl. 37, 2754–2794 (1998).
Monsigny, M., Mayer, R. & Roche, A.C. Sugar-lectin interactions: sugar clusters, lectin multivalency and avidity. Carbohydr. Lett. 4, 35–52 (2000).
Kiessling, L.L., Gestwicki, J.E. & Strong, L.E. Synthetic multivalent ligands in the exploration of cell-surface interactions. Curr. Opin. Chem. Biol. 4, 696–703 (2000).
Bach, A. et al. Design and synthesis of highly potent and plasma-stable dimeric inhibitors of the PSD-95-NMDA receptor interaction. Angew. Chem. Int. Ed. Engl. 48, 9685–9689 (2009).
Illendula, A. et al. Chemical biology. A small-molecule inhibitor of the aberrant transcription factor CBFβ-SMMHC delays leukemia in mice. Science 347, 779–784 (2015).
Profit, A.A., Lee, T.R. & Lawrence, D.S. Bivalent inhibitors of protein tyrosine kinases. J. Am. Chem. Soc. 121, 280–283 (1999).
Sun, H. et al. Design, synthesis, and characterization of a potent, nonpeptide, cell-permeable, bivalent Smac mimetic that concurrently targets both the BIR2 and BIR3 domains in XIAP. J. Am. Chem. Soc. 129, 15279–15294 (2007).
Bach, A. et al. A high-affinity, dimeric inhibitor of PSD-95 bivalently interacts with PDZ1-2 and protects against ischemic brain damage. Proc. Natl. Acad. Sci. USA 109, 3317–3322 (2012).
Filippakopoulos, P. & Knapp, S. Targeting bromodomains: epigenetic readers of lysine acetylation. Nat. Rev. Drug Discov. 13, 337–356 (2014).
Zeng, L. & Zhou, M.M. Bromodomain: an acetyl-lysine binding domain. FEBS Lett. 513, 124–128 (2002).
Smith, S.G. & Zhou, M.M. The bromodomain: a new target in emerging epigenetic medicine. ACS Chem. Biol. 11, 598–608 (2016).
Zuber, J. et al. RNAi screen identifies Brd4 as a therapeutic target in acute myeloid leukaemia. Nature 478, 524–528 (2011).
Delmore, J.E. et al. BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 146, 904–917 (2011).
Yang, Z. et al. Recruitment of P-TEFb for stimulation of transcriptional elongation by the bromodomain protein Brd4. Mol. Cell 19, 535–545 (2005).
Krueger, B.J., Varzavand, K., Cooper, J.J. & Price, D.H. The mechanism of release of P-TEFb and HEXIM1 from the 7SK snRNP by viral and cellular activators includes a conformational change in 7SK. PLoS One 5, e12335 (2010).
Filippakopoulos, P. et al. Selective inhibition of BET bromodomains. Nature 468, 1067–1073 (2010).
Tanaka, M., Roberts, J.M., Qi, J. & Bradner, J.E. Inhibitors of emerging epigenetic targets for cancer therapy: a patent review (2010–2014). Pharm. Pat. Anal. 4, 261–284 (2015).
Schröder, S. et al. Two-pronged binding with bromodomain-containing protein 4 liberates positive transcription elongation factor b from inactive ribonucleoprotein complexes. J. Biol. Chem. 287, 1090–1099 (2012).
Baud, M.G. et al. Chemical biology. A bump-and-hole approach to engineer controlled selectivity of BET bromodomain chemical probes. Science 346, 638–641 (2014).
Picaud, S. et al. RVX-208, an inhibitor of BET transcriptional regulators with selectivity for the second bromodomain. Proc. Natl. Acad. Sci. USA 110, 19754–19759 (2013).
Zhang, G. et al. Down-regulation of NF-κB transcriptional activity in HIV-associated kidney disease by BRD4 inhibition. J. Biol. Chem. 287, 28840–28851 (2012).
McKeown, M.R. et al. Biased multicomponent reactions to develop novel bromodomain inhibitors. J. Med. Chem. 57, 9019–9027 (2014).
Roberts, J.M. & Bradner, J.E. A bead-based proximity assay for BRD4 ligand discovery. Curr. Protoc. Chem. Biol. 7, 263–278 (2015).
Dahlin, J.L. et al. PAINS in the assay: chemical mechanisms of assay interference and promiscuous enzymatic inhibition observed during a sulfhydryl-scavenging HTS. J. Med. Chem. 58, 2091–2113 (2015).
Dawson, M.A. et al. Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia. Nature 478, 529–533 (2011).
Chaidos, A. et al. Potent antimyeloma activity of the novel bromodomain inhibitors I-BET151 and I-BET762. Blood 123, 697–705 (2014).
Bartholomeeusen, K., Xiang, Y., Fujinaga, K. & Peterlin, B.M. Bromodomain and extra-terminal (BET) bromodomain inhibition activate transcription via transient release of positive transcription elongation factor b (P-TEFb) from 7SK small nuclear ribonucleoprotein. J. Biol. Chem. 287, 36609–36616 (2012).
Winter, G.E. et al. Drug development. Phthalimide conjugation as a strategy for in vivo target protein degradation. Science 348, 1376–1381 (2015).
Lu, J. et al. Hijacking the E3 ubiquitin ligase cereblon to efficiently target BRD4. Chem. Biol. 22, 755–763 (2015).
Zengerle, M., Chan, K.H. & Ciulli, A. Selective small molecule induced degradation of the BET bromodomain protein BRD4. ACS Chem. Biol. 10, 1770–1777 (2015).
Matzuk, M.M. et al. Small-molecule inhibition of BRDT for male contraception. Cell 150, 673–684 (2012).
Dutta Roy, R. & Stefan, M.I. Cooperative binding mitigates the high-dose hook effect. Preprint at. bioRxiv http://dx.doi.org/10.1101/021717 (2015).
Martinez Molina, D. et al. Monitoring drug target engagement in cells and tissues using the cellular thermal shift assay. Science 341, 84–87 (2013).
Arrowsmith, C.H. et al. The promise and peril of chemical probes. Nat. Chem. Biol. 11, 536–541 (2015).
Schultz, J., Copley, R.R., Doerks, T., Ponting, C.P. & Bork, P. SMART: a web-based tool for the study of genetically mobile domains. Nucleic Acids Res. 28, 231–234 (2000).
Sanchez, R., Meslamani, J. & Zhou, M.M. The bromodomain: from epigenome reader to druggable target. Biochim. Biophys. Acta 1839, 676–685 (2014).
Anonymous. Bromosporine. SGC http://www.thesgc.org/chemical-probes/Bromosporine (accessed 19 June 2016).
French, C.A. et al. Midline carcinoma of children and young adults with NUT rearrangement. J. Clin. Oncol. 22, 4135–4139 (2004).
Anders, L. et al. Genome-wide localization of small molecules. Nat. Biotechnol. 32, 92–96 (2014).
Brumatti, G., Sheridan, C. & Martin, S.J. Expression and purification of recombinant annexin V for the detection of membrane alterations on apoptotic cells. Methods 44, 235–240 (2008).
Kabsch, W. Integration, scaling, space-group assignment and post-refinement. Acta Crystallogr. D Biol. Crystallogr. 66, 133–144 (2010).
McCoy, A.J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Smart, O.S. et al. Exploiting structure similarity in refinement: automated NCS and target-structure restraints in BUSTER. Acta Crystallogr. D Biol. Crystallogr. 68, 368–380 (2012).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Wiseman, T., Williston, S., Brandts, J.F. & Lin, L.N. Rapid measurement of binding constants and heats of binding using a new titration calorimeter. Anal. Biochem. 179, 131–137 (1989).
Acknowledgements
We thank J.A. Perry for critical reading of the manuscript and discussions. NMC797 cells were a gift from C. French (Brigham and Women's Hospital). Crystallographic work was based on research conducted at the Advanced Photon Source on the Northeastern Collaborative Access Team beamlines. We thank Mitsubishi Tanabe Pharma Corporation for nonresearch support of M.T. This research was supported by the William Lawrence and Blanche Hughes Foundation (J.E.B.), the Leukemia and Lymphoma Society (J.E.B.) and the US National Institutes of Health (CA066996-01A1, J.E.B.).
Author information
Authors and Affiliations
Contributions
M.T. and J.M.R. designed and led the study under the supervision of J.E.B. M.T. designed bivalent molecules and performed organic chemistry and medicinal chemistry experiments. J.M.R. designed and performed biochemical, cellular, and in vivo experiments. J.P. performed BRD4(1) FP experiments. S.D.-P., H.-S.S. and S.L.D. performed crystallographic studies. H.S.S. performed SEC and ITC experiments. A.S. and T.G.S. performed in vivo studies. M.T., J.M.R. and J.E.B. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
M.T. is a visiting scientist from Mitsubishi Tanabe Pharma Corporation and is supported by the company for nonresearch funds. J.E.B. is a founder of Tensha Therapeutics, a biotechnology company that develops drug-like derivatives of JQ1 as investigational cancer therapies. Dana-Farber Cancer Institute and M.T., J.M.R. and J.E.B. have filed patent applications (62/259,797, 62/261,703 and 62/338,968) that include MT1 and the analogs described in this manuscript. J.E.B. is an employee of the Novartis Institutes of BioMedical Research (effective 1 January 2016).
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Figures 1–15 and Supplementary Tables 1–2. (PDF 3059 kb)
Supplementary Note
Synthetic Procedures. (PDF 950 kb)
Supplementary Data Set 1
Supplementary Figure 2 source data. (XLSX 14 kb)
Supplementary Data Set 2
Supplementary Figure 4 source data. (XLSX 13 kb)
Supplementary Data Set 3
Supplementary Figure 6 source data. (XLSX 12 kb)
Supplementary Data Set 4
Supplementary Figure 7 source data. (XLSX 12 kb)
Supplementary Data Set 5
Supplementary Figure 8 source data. (XLSX 63 kb)
Supplementary Data Set 6
Supplementary Figure 9 source data. (XLSX 22 kb)
Supplementary Data Set 7
Supplementary Figure 13 source data. (XLSX 15 kb)
Rights and permissions
About this article
Cite this article
Tanaka, M., Roberts, J., Seo, HS. et al. Design and characterization of bivalent BET inhibitors. Nat Chem Biol 12, 1089–1096 (2016). https://doi.org/10.1038/nchembio.2209
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2209
This article is cited by
-
Targeting bromodomain-containing proteins: research advances of drug discovery
Molecular Biomedicine (2023)
-
Phosphorylation-dependent BRD4 dimerization and implications for therapeutic inhibition of BET family proteins
Communications Biology (2021)
-
Trivalent PROTACs enhance protein degradation via combined avidity and cooperativity
Nature Chemical Biology (2021)
-
Clinical perspectives of BET inhibition in ovarian cancer
Cellular Oncology (2021)
-
Potent BRD4 inhibitor suppresses cancer cell-macrophage interaction
Nature Communications (2020)