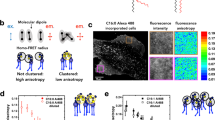
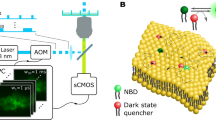
Abstract
Gangliosides, glycosphingolipids containing one or more sialic acid(s) in the glyco-chain, are involved in various important physiological and pathological processes in the plasma membrane. However, their exact functions are poorly understood, primarily because of the scarcity of suitable fluorescent ganglioside analogs. Here, we developed methods for systematically synthesizing analogs that behave like their native counterparts in regard to partitioning into raft-related membrane domains or preparations. Single-fluorescent-molecule imaging in the live-cell plasma membrane revealed the clear but transient colocalization and codiffusion of fluorescent ganglioside analogs with a fluorescently labeled glycosylphosphatidylinisotol (GPI)-anchored protein, human CD59, with lifetimes of 12 ms for CD59 monomers, 40 ms for CD59's transient homodimer rafts in quiescent cells, and 48 ms for engaged-CD59-cluster rafts, in cholesterol- and GPI-anchoring-dependent manners. The ganglioside molecules were always mobile in quiescent cells. These results show that gangliosides continually and dynamically exchange between raft domains and the bulk domain, indicating that raft domains are dynamic entities.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sampaio, J.L. et al. Membrane lipidome of an epithelial cell line. Proc. Natl. Acad. Sci. USA 108, 1903–1907 (2011).
Smith, D.C., Lord, J.M., Roberts, L.M. & Johannes, L. Glycosphingolipids as toxin receptors. Semin. Cell Dev. Biol. 15, 397–408 (2004).
Fleming, F.E. et al. Relative roles of GM1 ganglioside, N-acylneuraminic acids, and α2β1 integrin in mediating rotavirus infection. J. Virol. 88, 4558–4571 (2014).
Coskun, Ü., Grzybek, M., Drechsel, D. & Simons, K. Regulation of human EGF receptor by lipids. Proc. Natl. Acad. Sci. USA 108, 9044–9048 (2011).
Simons, K. & Gerl, M.J. Revitalizing membrane rafts: new tools and insights. Nat. Rev. Mol. Cell Biol. 11, 688–699 (2010).
Lingwood, D. et al. Cholesterol modulates glycolipid conformation and receptor activity. Nat. Chem. Biol. 7, 260–262 (2011).
Fujita, A. et al. Gangliosides GM1 and GM3 in the living cell membrane form clusters susceptible to cholesterol depletion and chilling. Mol. Biol. Cell 18, 2112–2122 (2007).
Schwarzmann, G., Wendeler, M. & Sandhoff, K. Synthesis of novel NBD-GM1 and NBD-GM2 for the transfer activity of GM2-activator protein by a FRET-based assay system. Glycobiology 15, 1302–1311 (2005).
Larsson, E.A. et al. Synthesis of reference standards to enable single cell metabolomic studies of tetramethylrhodamine-labeled ganglioside GM1. Carbohydr. Res. 342, 482–489 (2007).
Polyakova, O. et al. Proteolysis of prion protein by cathepsin S generates a soluble beta-structured intermediate oligomeric form, with potential implications for neurotoxic mechanisms. Eur. Biophys. J. 38, 209–218 (2009).
Marushchak, D., Gretskaya, N., Mikhalyov, I. & Johansson, L.B. Self-aggregation—an intrinsic property of G(M1) in lipid bilayers. Mol. Membr. Biol. 24, 102–112 (2007).
Eggeling, C. et al. Direct observation of the nanoscale dynamics of membrane lipids in a living cell. Nature 457, 1159–1162 (2009).
Polyakova, S.M. et al. New GM1 ganglioside derivatives for selective single and double labelling of the natural glycosphingolipid skeleton. Eur. J. Org. 30, 5162–5177 (2009).
Chinnapen, D.J. et al. Lipid sorting by ceramide structure from plasma membrane to ER for the cholera toxin receptor ganglioside GM1. Dev. Cell 23, 573–586 (2012).
Sezgin, E. et al. Partitioning, diffusion, and ligand binding of raft lipid analogs in model and cellular plasma membranes. Biochim. Biophys. Acta 1818, 1777–1784 (2012).
Hammond, A.T. et al. Crosslinking a lipid raft component triggers liquid ordered-liquid disordered phase separation in model plasma membranes. Proc. Natl. Acad. Sci. USA 102, 6320–6325 (2005).
Lingwood, D., Ries, J., Schwille, P. & Simons, K. Plasma membranes are poised for activation of raft phase coalescence at physiological temperature. Proc. Natl. Acad. Sci. USA 105, 10005–10010 (2008).
Kaiser, H.-J. et al. Order of lipid phases in model and plasma membranes. Proc. Natl. Acad. Sci. USA 106, 16645–16650 (2009).
Ando, H., Koike, Y., Ishida, H. & Kiso, M. Extending the possibility of an N-Troc-protected sialic acid donor toward variant sialo-glycoside synthesis. Tetrahedr. Lett. 44, 6883–6886 (2003).
Ando, H., Koike, Y., Koizumi, S., Ishida, H. & Kiso, M. 1,5-Lactamized sialyl acceptors for various disialoside syntheses: novel method for the synthesis of glycan portions of Hp-s6 and HLG-2 gangliosides. Angew. Chem. Int. Edn. Engl. 44, 6759–6763 (2005).
Imamura, A., Ando, H., Ishida, H. & Kiso, M. Ganglioside GQ1b: efficient total synthesis and the expansion to synthetic derivatives to elucidate its biological roles. J. Org. Chem. 74, 3009–3023 (2009).
Iwayama, Y., Ando, H., Ishida, H. & Kiso, M. A first total synthesis of ganglioside HLG-2. Chemistry 15, 4637–4648 (2009).
Tamai, H. et al. The total synthesis of the neurogenic ganglioside LLG-3 isolated from the starfish Linckia laevigata. Angew. Chem. Int. Edn. Engl. 50, 2330–2333 (2011).
Fujikawa, K. et al. The first total synthesis of ganglioside GalNAc-GD1a, a target molecule for autoantibodies in Guillain-Barré syndrome. Chemistry 17, 5641–5651 (2011).
Nakashima, S. et al. Efficiently synthesizing lacto-ganglio-series gangliosides by using a glucosyl ceramide cassette approach: the total synthesis of ganglioside X2. Chem. Asian J. 7, 1041–1051 (2012).
Hiramoto-Yamaki, N. et al. Ultrafast diffusion of a fluorescent cholesterol analog in compartmentalized plasma membranes. Traffic 15, 583–612 (2014).
Suzuki, K.G.N. et al. GPI-anchored receptor clusters transiently recruit Lyn and G α for temporary cluster immobilization and Lyn activation: single-molecule tracking study 1. J. Cell Biol. 177, 717–730 (2007a).
Suzuki, K.G.N., Fujiwara, T.K., Edidin, M. & Kusumi, A. Dynamic recruitment of phospholipase C γ at transiently immobilized GPI-anchored receptor clusters induces IP3-Ca2+ signaling: single-molecule tracking study 2. J. Cell Biol. 177, 731–742 (2007b).
Suzuki, K.G.N. et al. Transient GPI-anchored protein homodimers are units for raft organization and function. Nat. Chem. Biol. 8, 774–783 (2012).
Taylor, D.R., Watt, N.T., Perera, W.S. & Hooper, N.M. Assigning functions to distinct regions of the N-terminus of the prion protein that are involved in its copper-stimulated, clathrin-dependent endocytosis. J. Cell Sci. 118, 5141–5153 (2005).
Lenne, P.F. et al. Dynamic molecular confinement in the plasma membrane by microdomains and the cytoskeleton meshwork. EMBO J. 25, 3245–3256 (2006).
Jones, R. et al. Tracking diffusion of GM1 gangliosides and zona pellucida binding molecules in sperm plasma membranes following cholesterol efflux. Dev. Biol. 339, 398–406 (2010).
Svistounov, D. et al. The relationship between fenestrations, sieve plates and rafts in liver sinusoidal endothelial cells. PLoS One 7, e46134 (2012).
Baumgart, T. et al. Large-scale fluid/fluid phase separation of proteins and lipids in giant plasma membrane vesicles. Proc. Natl. Acad. Sci. USA 104, 3165–3170 (2007).
Levental, I., Grzybek, M. & Simons, K. Raft domains of variable properties and compositions in plasma membrane vesicles. Proc. Natl. Acad. Sci. USA 108, 11411–11416 (2011).
Harder, T., Scheiffele, P., Verkade, P. & Simons, K. Lipid domain structure of the plasma membrane revealed by patching of membrane components. J. Cell Biol. 141, 929–942 (1998).
Raghupathy, R. et al. Transbilayer lipid interactions mediate nanoclustering of lipid-anchored proteins. Cell 161, 581–594 (2015).
Mayor, S., Rothberg, K.G. & Maxfield, F.R. Sequestration of GPI-anchored proteins in caveolae triggered by cross-linking. Science 264, 1948–1951 (1994).
Tanaka, K.A.K. et al. Membrane molecules mobile even after chemical fixation. Nat. Methods 7, 865–866 (2010).
Kusumi, A. & Suzuki, K. Toward understanding the dynamics of membrane-raft-based molecular interactions. Biochim. Biophys. Acta 1746, 234–251 (2005).
Huang, H., Simsek, M.F., Jin, W. & Pralle, A. Effect of receptor dimerization on membrane lipid raft structure continuously quantified on single cells by camera based fluorescence correlation spectroscopy. PLoS One 10, e0121777 (2015).
Sahl, S.J., Leutenegger, M., Hilbert, M., Hell, S.W. & Eggeling, C. Fast molecular tracking maps nanoscale dynamics of plasma membrane lipids. Proc. Natl. Acad. Sci. USA 107, 6829–6834 (2010).
Honigmann, A. et al. Scanning STED-FCS reveals spatiotemporal heterogeneity of lipid interaction in the plasma membrane of living cells. Nat. Commun. 5, 5412 (2014).
Koyama-Honda, I. et al. Fluorescence imaging for monitoring the colocalization of two single molecules in living cells. Biophys. J. 88, 2126–2136 (2005).
Murase, K. et al. Ultrafine membrane compartments for molecular diffusion as revealed by single molecule techniques. Biophys. J. 86, 4075–4093 (2004).
Tsurifune, T. et al. Alteration of tumor phenotypes of B16 melanoma after genetic remodeling of the ganglioside profile. Int. J. Oncol. 17, 159–165 (2000).
Pagano, R.E. & Sleight, R.G. Defining lipid transport pathways in animal cells. Science 229, 1051–1057 (1985).
Schwarzmann, G., Hofmann, P., Pütz, U. & Albrecht, B. Demonstration of direct glycosylation of nondegradable glucosylceramide analogs in cultured cells. J. Biol. Chem. 270, 21271–21276 (1995).
Yamashiro, S. et al. Substrate specificity of beta1,4-N-acetylgalactosaminyltransferase in vitro and in cDNA-transfected cells. J. Biol. Chem. 270, 6149–6155 (1995).
Kenworthy, A.K. et al. Dynamics of putative raft-associated proteins at the cell surface. J. Cell Biol. 165, 735–746 (2004).
Takamiya, K. et al. Complex gangliosides are essential in spermatogenesis of mice: possible roles in the transport of testosterone. Proc. Natl. Sci. Acad. USA 95, 12147–12152 (1998).
Anderson, K., Li, S.C. & Li, Y.T. Diphenylamine-aniline-phosphoric acid reagent, a versatile spray reagent for revealing glycoconjugates on thin-layer chromatography plates. Anal. Biochem. 287, 337–339 (2000).
Sternberg, R.S. Biomedical image processing. IEEE Computer 16, 22–34 (1983).
Chen, Y. et al. Genome-wide functional annotation of dual-specificity protein- and lipid-binding modules that regulate protein interactions. Mol. Cell 46, 226–237 (2012).
Man, H.Y. et al. Regulation of AMPA receptor-mediated synaptic transmission by clathrin-dependent receptor internalization. Neuron 25, 649–662 (2000).
Fujiwara, T., Ritchie, K., Murakoshi, H., Jacobson, K. & Kusumi, A. Phospholipids undergo hop diffusion in compartmentalized cell membrane. J. Cell Biol. 157, 1071–1081 (2002).
Kasai, R.S. et al. Full characterization of GPCR monomer-dimer dynamic equilibrium by single molecule imaging. J. Cell Biol. 192, 463–480 (2011).
Dunne, P.D. et al. DySCo: quantitating associations of membrane proteins using two-color single-molecule tracking. Biophys. J. 97, L5–L7 (2009).
Otsu, N. A threshold selection method from gray-level histograms. IEEE Trans. Syst. Man Cybern. 9, 62–66 (1979).
Simson, R., Sheets, E.D. & Jacobson, K. Detection of temporary lateral confinement of membrane proteins using single-particle tracking analysis. Biophys. J. 69, 989–993 (1995).
Acknowledgements
We thank M. Sokabe of Nagoya University, B. Baird of Cornell University, S. Ohnishi of Kyoto University, and V. Horejsi of Academy of Sciences of the Czech Republic for their kind gifts of human epithelial T24 cells, RBL-2H3 cells, NRK cells, and a mouse hybridoma cell line MEM43/5 which produces anti CD59 monoclonal antibody, respectively, as well as R.S. Kasai, K.A.K. Tanaka, and K.M. Hirosawa for setting up the single-molecule tracking station and useful discussions. This work was supported in part by Grants-in-Aid for scientific research from the Japan Society for the Promotion of Science (Wakate A to H.A. (23688014), Kiban B to K.G.N.S. (24370055 and 15H04351), H.A. (15H04495), K.F. (24390078), and M.K. (22380067), and Kiban A to A.K. (24247029)); by Grants-in-Aid for Innovative Areas (Deciphering sugar chain-based signals regulating integrative neuronal functions) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan to H.A. (24110505 and 26110704), K.G.N.S. (23110001), K.F. (23110008), and T.K.F. (25113515 and 26115707); by a grant from Core Research for Evolutional Science and Technology (CREST project of “Creation of Fundamental Technologies for Understanding and Control of Biosystem Dynamics) of Japan Science and Technology Agency (JST) to A.K.; and by a grant awarded by the NIH to W.C. (GM68849). WPI-iCeMS of Kyoto University is supported by the World Premiere Research Center Initiative (WPI) of the MEXT.
Author information
Authors and Affiliations
Contributions
K.G.N.S., H.A., A.K., and M. Kiso conceived this research and designed experiments; N.K., H.A., M. Konishi, M. Koikeda, A.I., and M. Kiso developed organic synthesis methods and performed organic synthesis; R.S. and W.C. performed SPR experiments and analysis; K.G.N.S. examined ganglioside partitioning into DRM and Lo/Ld domains, studied colocalizations of CD59 patches with ganglioside analogues using epifluorescence, and performed all of the single-molecule tracking experiments; R.C. conducted most of the Lo/Ld partitioning measurements; T.K.F. set up the single-molecule tracking instrument and developed software for single-molecule colocalization detection; H.T. examined colocalizations of CD59 patches with ganglioside analogues using a confocal fluorescence microscope; F. Koichi, F. Keiko, and Y.Y. prepared GM3-depleted cell lines; K.G.N.S., H.A., A.K., and M. Kiso wrote the paper; and all authors participated in the revisions of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Tables 1–5 and Supplementary Figures 1–6. (PDF 2068 kb)
Supplementary Note
Synthetic Procedures (PDF 452 kb)
41589_2016_BFnchembio2059_MOESM69_ESM.mov
A representative simultaneous two-color single fluorescent-molecule imaging video clip of a 594-GM1 molecule (red) diffusing in the PM containing CD59 patches (green) (the original video for the still image in Fig. 3a, left). (MOV 1393 kb)
Rights and permissions
About this article
Cite this article
Komura, N., Suzuki, K., Ando, H. et al. Raft-based interactions of gangliosides with a GPI-anchored receptor. Nat Chem Biol 12, 402–410 (2016). https://doi.org/10.1038/nchembio.2059
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2059
This article is cited by
-
Single-molecule localization microscopy reveals STING clustering at the trans-Golgi network through palmitoylation-dependent accumulation of cholesterol
Nature Communications (2024)
-
Regulation of membrane protein structure and function by their lipid nano-environment
Nature Reviews Molecular Cell Biology (2023)
-
Recently developed glycosphingolipid probes and their dynamic behavior in cell plasma membranes as revealed by single-molecule imaging
Glycoconjugate Journal (2023)
-
Synthesis and biological activity of ganglioside GM3 analogues with a (S)-CHF-Sialoside linkage and an alkyne tag
Glycoconjugate Journal (2023)
-
Cell density-dependent membrane distribution of ganglioside GM3 in melanoma cells
Cellular and Molecular Life Sciences (2023)