Abstract
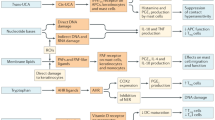
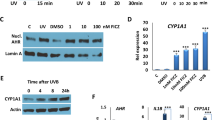
The recurrent interaction of skin with sunlight is an intrinsic constituent of human life, and exhibits both beneficial and detrimental effects. The apparent robust architectural framework of skin conceals remarkable mechanisms that operate at the interface between the surface and environment. In this Review, we discuss three distinct protective mechanisms and response pathways that safeguard skin from deleterious effects of ultraviolet (UV) radiation. The unique stratified epithelial architecture of human skin along with the antioxidant-response pathways constitutes the important defense mechanisms against UV radiation. The intricate pigmentary system and its intersection with the immune-system cytokine axis delicately balance tissue homeostasis. We discuss the relationship among these networks in the context of an unusual depigmenting disorder, vitiligo. The elaborate tunable mechanisms, elegant multilayered architecture and evolutionary selection pressures involved in skin and sunlight interaction makes this a compelling model to understand biological complexity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Dyck, J. Structure and colour-production of the blue barbs of Agapornis roseicollis and Cotinga maynana. Z. Zellforsch. Mikrosk. Anat. 115, 17–29 (1971).
Pathak, M.A., Riley, F.J., Fitzpatrick, T.B. & Curwen, W.L. Melanin formation in human skin induced by long-wave ultra-violet and visible light. Nature 193, 148–150 (1962).
Bogh, M.K., Schmedes, A.V., Philipsen, P.A., Thieden, E. & Wulf, H.C. Vitamin D production after UVB exposure depends on baseline vitamin D and total cholesterol but not on skin pigmentation. J. Invest. Dermatol. 130, 546–553 (2010).
Hart, P.H., Gorman, S. & Finlay-Jones, J.J. Modulation of the immune system by UV radiation: more than just the effects of vitamin D? Nat. Rev. Immunol. 11, 584–596 (2011).
Janich, P. et al. Human epidermal stem cell function is regulated by circadian oscillations. Cell Stem Cell 13, 745–753 (2013).
Bowden, G.T. Prevention of non-melanoma skin cancer by targeting ultraviolet-B-light signalling. Nat. Rev. Cancer 4, 23–35 (2004).
Rigel, D.S. Cutaneous ultraviolet exposure and its relationship to the development of skin cancer. J. Am. Acad. Dermatol. 58, S129–S132 (2008).
Bald, T. et al. Ultraviolet-radiation-induced inflammation promotes angiotropism and metastasis in melanoma. Nature 507, 109–113 (2014). Ancillary role of sunlight in facilitating metastasis of melanoma by the induction of angiotropic factor HMGB1 is deciphered and this study provides molecular explanation of sun exposure induced melanoma spread.
Wondrak, G.T., Jacobson, M.K. & Jacobson, E.L. Endogenous UVA-photosensitizers: mediators of skin photodamage and novel targets for skin photoprotection. Photochem. Photobiol. Sci. 5, 215–237 (2006).
Dalle Carbonare, M. & Pathak, M.A. Skin photosensitizing agents and the role of reactive oxygen species in photoaging. J. Photochem. Photobiol. B 14, 105–124 (1992).
Lechler, T. & Fuchs, E. Asymmetric cell divisions promote stratification and differentiation of mammalian skin. Nature 437, 275–280 (2005). Niche-dependent oriented cell divisions in the basal epidermal layer are described, and the crucial role of intercellular connections in the control of asymmetric cell divisions is highlighted.
Lin, J.Y. & Fisher, D.E. Melanocyte biology and skin pigmentation. Nature 445, 843–850 (2007).
Yamaguchi, Y., Brenner, M. & Hearing, V.J. The regulation of skin pigmentation. J. Biol. Chem. 282, 27557–27561 (2007). This is an excellent compilation of regulatory pathways that govern pigmentation during developmental and constitutive pigmentation in human epidermis.
Miyamura, Y. et al. Regulation of human skin pigmentation and responses to ultraviolet radiation. Pigment Cell Res. 20, 2–13 (2007).
Rawlings, A.V. & Harding, C.R. Moisturization and skin barrier function. Dermatol. Ther. 17 (suppl. 1): 43–48 (2004).
Wertz, P.W. Current understanding of skin biology pertinent to skin penetration: skin biochemistry. Skin Pharmacol. Physiol. 26, 217–226 (2013).
Del Rosso, J.Q. & Levin, J. Clinical relevance of maintaining the structural and functional integrity of the stratum corneum: why is it important to you? J. Drugs Dermatol. 10, s5–s12 (2011).
Koster, M.I. & Roop, D.R. Mechanisms regulating epithelial stratification. Annu. Rev. Cell Dev. Biol. 23, 93–113 (2007).
Elias, P.M. et al. Formation and functions of the corneocyte lipid envelope (CLE). Biochim. Biophys. Acta 1841, 314–318 (2014).
Proksch, E., Brandner, J.M. & Jensen, J.M. The skin: an indispensable barrier. Exp. Dermatol. 17, 1063–1072 (2008).
Feingold, K.R. Thematic review series: skin lipids. The role of epidermal lipids in cutaneous permeability barrier homeostasis. J. Lipid Res. 48, 2531–2546 (2007). This review summarizes the crucial role of lipids as cellular mortar for the barrier function of skin, and highlights their importance in inherited skin disorders.
Feingold, K.R. & Elias, P.M. Role of lipids in the formation and maintenance of the cutaneous permeability barrier. Biochim. Biophys. Acta 1841, 280–294 (2014).
Candi, E., Schmidt, R. & Melino, G. The cornified envelope: a model of cell death in the skin. Nat. Rev. Mol. Cell Biol. 6, 328–340 (2005).
van Smeden, J., Janssens, M., Gooris, G.S. & Bouwstra, J.A. The important role of stratum corneum lipids for the cutaneous barrier function. Biochim. Biophys. Acta 1841, 295–313 (2014).
Bragulla, H.H. & Homberger, D.G. Structure and functions of keratin proteins in simple, stratified, keratinized and cornified epithelia. J. Anat. 214, 516–559 (2009).
Feingold, K.R. The regulation and role of epidermal lipid synthesis. Adv. Lipid Res. 24, 57–82 (1991).
Ovaere, P., Lippens, S., Vandenabeele, P. & Declercq, W. The emerging roles of serine protease cascades in the epidermis. Trends Biochem. Sci. 34, 453–463 (2009).
Elias, P.M. et al. Basis for abnormal desquamation and permeability barrier dysfunction in RXLI. J. Invest. Dermatol. 122, 314–319 (2004).
Hansen, H.S. & Jensen, B. Essential function of linoleic acid esterified in acylglucosylceramide and acylceramide in maintaining the epidermal water permeability barrier. Evidence from feeding studies with oleate, linoleate, arachidonate, columbinate and alpha-linolenate. Biochim. Biophys. Acta 834, 357–363 (1985).
Wertz, P.W., Cho, E.S. & Downing, D.T. Effect of essential fatty acid deficiency on the epidermal sphingolipids of the rat. Biochim. Biophys. Acta 753, 350–355 (1983).
Elias, P.M. et al. Stratum corneum lipids in disorders of cornification. Steroid sulfatase and cholesterol sulfate in normal desquamation and the pathogenesis of recessive X-linked ichthyosis. J. Clin. Invest. 74, 1414–1421 (1984).
Schurer, N.Y. & Elias, P.M. The biochemistry and function of stratum corneum lipids. Adv. Lipid Res. 24, 27–56 (1991).
Zeeuwen, P.L. Epidermal differentiation: the role of proteases and their inhibitors. Eur. J. Cell Biol. 83, 761–773 (2004).
Madison, K.C. Barrier function of the skin: “la raison d'etre” of the epidermis. J. Invest. Dermatol. 121, 231–241 (2003).
Holleran, W.M. et al. Consequences of beta-glucocerebrosidase deficiency in epidermis. Ultrastructure and permeability barrier alterations in Gaucher disease. J. Clin. Invest. 93, 1756–1764 (1994).
Schmuth, M. et al. Permeability barrier disorder in Niemann-Pick disease: sphingomyelin-ceramide processing required for normal barrier homeostasis. J. Invest. Dermatol. 115, 459–466 (2000).
Mao-Qiang, M., Feingold, K.R., Jain, M. & Elias, P.M. Extracellular processing of phospholipids is required for permeability barrier homeostasis. J. Lipid Res. 36, 1925–1935 (1995).
Lopez-Pajares, V., Yan, K., Zarnegar, B.J., Jameson, K.L. & Khavari, P.A. Genetic pathways in disorders of epidermal differentiation. Trends Genet. 29, 31–40 (2013).
Elias, P.M., Williams, M.L., Holleran, W.M., Jiang, Y.J. & Schmuth, M. Pathogenesis of permeability barrier abnormalities in the ichthyoses: inherited disorders of lipid metabolism. J. Lipid Res. 49, 697–714 (2008).
Gupta, L.K. & Singhi, M.K. Wood's lamp. Indian J. Dermatol. Venereol. Leprol. 70, 131–135 (2004).
Schäfer, M. & Werner, S. Cancer as an overhealing wound: an old hypothesis revisited. Nat. Rev. Mol. Cell Biol. 9, 628–638 (2008).
Jensen, M.A., Wilkinson, J.E. & Krainer, A.R. Splicing factor SRSF6 promotes hyperplasia of sensitized skin. Nat. Struct. Mol. Biol. 21, 189–197 (2014).
Sundaram, G.M. et al. 'See-saw' expression of microRNA-198 and FSTL1 from a single transcript in wound healing. Nature 495, 103–106 (2013).
Rinn, J.L. et al. A systems biology approach to anatomic diversity of skin. J. Invest. Dermatol. 128, 776–782 (2008).
Driskell, R.R. et al. Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature 504, 277–281 (2013).
Bryan, N. et al. Reactive oxygen species (ROS)–a family of fate deciding molecules pivotal in constructive inflammation and wound healing. Eur. Cell. Mater. 24, 249–265 (2012).
Pillai, S., Oresajo, C. & Hayward, J. Ultraviolet radiation and skin aging: roles of reactive oxygen species, inflammation and protease activation, and strategies for prevention of inflammation-induced matrix degradation–a review. Int. J. Cosmet. Sci. 27, 17–34 (2005).
Takashima, A. & Bergstresser, P.R. Impact of UVB radiation on the epidermal cytokine network. Photochem. Photobiol. 63, 397–400 (1996).
Cadet, J., Douki, T., Ravanat, J.L. & Di Mascio, P. Sensitized formation of oxidatively generated damage to cellular DNA by UVA radiation. Photochem. Photobiol. Sci. 8, 903–911 (2009).
Walterscheid, J.P. et al. cis-Urocanic acid, a sunlight-induced immunosuppressive factor, activates immune suppression via the 5–HT2A receptor. Proc. Natl. Acad. Sci. USA 103, 17420–17425 (2006).
Denat, L., Kadekaro, A.L., Marrot, L., Leachman, S.A. & Abdel-Malek, Z.A. Melanocytes as Instigators and victims of oxidative stress. J. Invest. Dermatol. 134, 1512–1518 (2014). In this review of the current understanding of oxidative stress and mitigating pathways that are operational in skin, the role of ROS generation is discussed in the context of vitiligo and cancer.
Mitra, D. et al. An ultraviolet-radiation-independent pathway to melanoma carcinogenesis in the red hair/fair skin background. Nature 491, 449–453 (2012).
Berger, M.F. et al. Melanoma genome sequencing reveals frequent PREX2 mutations. Nature 485, 502–506 (2012).
Lai-Cheong, J.E. et al. Loss-of-function FERMT1 mutations in kindler syndrome implicate a role for fermitin family homolog-1 in integrin activation. Am. J. Pathol. 175, 1431–1441 (2009).
Bickers, D.R. & Athar, M. Oxidative stress in the pathogenesis of skin disease. J. Invest. Dermatol. 126, 2565–2575 (2006).
Natarajan, V.T. et al. Transcriptional upregulation of Nrf2-dependent phase II detoxification genes in the involved epidermis of vitiligo vulgaris. J. Invest. Dermatol. 130, 2781–2789 (2010). This study ascertains the role of oxidative stress in vitiligo and provides molecular understanding of differences in redox perturbations in the two epidermal cell types.
Cavarra, E. et al. UVA light stimulates the production of cathepsin G and elastase-like enzymes by dermal fibroblasts: a possible contribution to the remodeling of elastotic areas in sun-damaged skin. Biol. Chem. 383, 199–206 (2002).
Fagot, D., Asselineau, D. & Bernerd, F. Direct role of human dermal fibroblasts and indirect participation of epidermal keratinocytes in MMP-1 production after UV-B irradiation. Arch. Dermatol. Res. 293, 576–583 (2002).
Quan, T., He, T., Voorhees, J.J. & Fisher, G.J. Ultraviolet irradiation induces Smad7 via induction of transcription factor AP-1 in human skin fibroblasts. J. Biol. Chem. 280, 8079–8085 (2005).
Raposo, G. & Marks, M.S. Melanosomes—dark organelles enlighten endosomal membrane transport. Nat. Rev. Mol. Cell Biol. 8, 786–797 (2007).
Wasmeier, C., Hume, A.N., Bolasco, G. & Seabra, M.C. Melanosomes at a glance. J. Cell Sci. 121, 3995–3999 (2008).
Urabe, K. et al. The inherent cytotoxicity of melanin precursors: a revision. Biochim. Biophys. Acta 1221, 272–278 (1994).
Raposo, G. & Marks, M.S. The dark side of lysosome-related organelles: specialization of the endocytic pathway for melanosome biogenesis. Traffic 3, 237–248 (2002).
Fowler, D.M. et al. Functional amyloid formation within mammalian tissue. PLoS Biol. 4, e6 (2006).
Theos, A.C., Truschel, S.T., Raposo, G. & Marks, M.S. The Silver locus product Pmel17/gp100/Silv/ME20: controversial in name and in function. Pigment Cell Res. 18, 322–336 (2005).
Sturm, R.A., Box, N.F. & Ramsay, M. Human pigmentation genetics: the difference is only skin deep. Bioessays 20, 712–721 (1998).
Steingrímsson, E., Copeland, N.G. & Jenkins, N.A. Melanocytes and the microphthalmia transcription factor network. Annu. Rev. Genet. 38, 365–411 (2004).
Vachtenheim, J. & Borovansky, J. “Transcription physiology” of pigment formation in melanocytes: central role of MITF. Exp. Dermatol. 19, 617–627 (2010).
Borovanský, J. & Elleder, M. Melanosome degradation: fact or fiction. Pigment Cell Res. 16, 280–286 (2003).
Moan, J., Nielsen, K.P. & Juzeniene, A. Immediate pigment darkening: its evolutionary roles may include protection against folate photosensitization. FASEB J. 26, 971–975 (2012).
Wicks, N.L., Chan, J.W., Najera, J.A., Ciriello, J.M. & Oancea, E. UVA phototransduction drives early melanin synthesis in human melanocytes. Curr. Biol. 21, 1906–1911 (2011).
Bellono, N.W., Kammel, L.G., Zimmerman, A.L. & Oancea, E. UV light phototransduction activates transient receptor potential A1 ion channels in human melanocytes. Proc. Natl. Acad. Sci. USA 110, 2383–2388 (2013).
Yamaguchi, Y. & Hearing, V.J. Physiological factors that regulate skin pigmentation. Biofactors 35, 193–199 (2009).
Cui, R. et al. Central role of p53 in the suntan response and pathologic hyperpigmentation. Cell 128, 853–864 (2007). This milestone study revealed the central guardian of the genome, p53 to function as a sensor and effector for UV pigmentation through the transcriptional upregulation of POMC gene that codes for the central melanogenesis factor MSH.
Yang, G. et al. Inhibition of PAX3 by TGF-beta modulates melanocyte viability. Mol. Cell 32, 554–563 (2008).
Yamaguchi, Y. et al. Dickkopf 1 (DKK1) regulates skin pigmentation and thickness by affecting Wnt/beta-catenin signaling in keratinocytes. FASEB J. 22, 1009–1020 (2008).
Novák, B. & Tyson, J.J. Design principles of biochemical oscillators. Nat. Rev. Mol. Cell Biol. 9, 981–991 (2008).
Natarajan, V.T. et al. IFN-gamma signaling maintains skin pigmentation homeostasis through regulation of melanosome maturation. Proc. Natl. Acad. Sci. USA 111, 2301–2306 (2014). This recent study identified IFN-γ as a hypopigmenting factor by stalling melanosome maturation process and established a physiological and pathological relevance of this effect in multiple models.
Zaidi, M.R. et al. Interferon-gamma links ultraviolet radiation to melanomagenesis in mice. Nature 469, 548–553 (2011). This study unraveled the molecular basis to melanoma formation in response to UV light, and provides a link between the immune system and skin cancer formation.
Carroll, J.M., Crompton, T., Seery, J.P. & Watt, F.M. Transgenic mice expressing IFN-gamma in the epidermis have eczema, hair hypopigmentation, and hair loss. J. Invest. Dermatol. 108, 412–422 (1997).
Choi, H. et al. IL-4 inhibits the melanogenesis of normal human melanocytes through the JAK2-STAT6 signaling pathway. J. Invest. Dermatol. 133, 528–536 (2013).
Solano, F., Briganti, S., Picardo, M. & Ghanem, G. Hypopigmenting agents: an updated review on biological, chemical and clinical aspects. Pigment Cell Res. 19, 550–571 (2006).
Wang, C.Q. et al. Th17 cells and activated dendritic cells are increased in vitiligo lesions. PLoS ONE 6, e18907 (2011).
Rashighi, M. et al. CXCL10 is critical for the progression and maintenance of depigmentation in a mouse model of vitiligo. Sci. Transl. Med. 6, 223ra23 (2014).
Malhotra, N. & Dytoc, M. The pathogenesis of vitiligo. J. Cutan. Med. Surg. 17, 153–172 (2013).
Guerra, L., Dellambra, E., Brescia, S. & Raskovic, D. Vitiligo: pathogenetic hypotheses and targets for current therapies. Curr. Drug Metab. 11, 451–467 (2010).
Le Poole, I.C. & Luiten, R.M. Autoimmune etiology of generalized vitiligo. Curr. Dir. Autoimmun. 10, 227–243 (2008).
Singh, A. et al. HLA alleles and amino-acid signatures of the peptide-binding pockets of HLA molecules in vitiligo. J. Invest. Dermatol. 132, 124–134 (2012).
Westerhof, W., Manini, P., Napolitano, A. & d'Ischia, M. The haptenation theory of vitiligo and melanoma rejection: a close-up. Exp. Dermatol. 20, 92–96 (2011).
Schallreuter, K.U. et al. Butyrylcholinesterase is present in the human epidermis and is regulated by H2O2: more evidence for oxidative stress in vitiligo. Biochem. Biophys. Res. Commun. 349, 931–938 (2006).
Jin, Y. et al. Genome-wide association analyses identify 13 new susceptibility loci for generalized vitiligo. Nat. Genet. 44, 676–680 (2012).
Spritz, R.A. The genetics of generalized vitiligo. Curr. Dir. Autoimmun. 10, 244–257 (2008).
Ortonne, J.P. & Bose, S.K. Vitiligo: where do we stand? Pigment Cell Res. 6, 61–72 (1993).
Le Poole, I.C., Das, P.K., van den Wijngaard, R.M., Bos, J.D. & Westerhof, W. Review of the etiopathomechanism of vitiligo: a convergence theory. Exp. Dermatol. 2, 145–153 (1993).
Schallreuter, K.U. et al. Vitiligo pathogenesis: autoimmune disease, genetic defect, excessive reactive oxygen species, calcium imbalance, or what else? Exp. Dermatol. 17, 139–160 (2008). These two papers (refs. 94 and 95) summarize the caveats in interpreting the observations on vitiligo in light of one theory and propose interplay of several confounding factors in vitiligo etiopathogenesis.
Teulings, H.E. et al. Decreased risk of melanoma and nonmelanoma skin cancer in patients with vitiligo: a survey among 1307 patients and their partners. Br. J. Dermatol. 168, 162–171 (2013).
Kaidbey, K.H., Agin, P.P., Sayre, R.M. & Kligman, A.M. Photoprotection by melanin–a comparison of black and Caucasian skin. J. Am. Acad. Dermatol. 1, 249–260 (1979).
Kollias, N., Sayre, R.M., Zeise, L. & Chedekel, M.R. Photoprotection by melanin. J. Photochem. Photobiol. B 9, 135–160 (1991).
Chou, W.C. et al. Direct migration of follicular melanocyte stem cells to the epidermis after wounding or UVB irradiation is dependent on Mc1r signaling. Nat. Med. 19, 924–929 (2013).
Grimes, P.E. Psoralen photochemotherapy for vitiligo. Clin. Dermatol. 15, 921–926 (1997).
Acknowledgements
This work was supported by grant (TOUCH-BSC0302). P.G., A.R. and R.G. are supported by the Council for Scientific and Industrial Research (CSIR), India. We acknowledge CSIR and Department of Biotechnology for institutional support to CSIR-Institute of Genomics and Integrative Biology and National Institute of Immunology, respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
R.S.G. is the co-founder director on the board of Vyome Biosciences, a biopharmaceutical company in the area of dermatology.
Rights and permissions
About this article
Cite this article
Natarajan, V., Ganju, P., Ramkumar, A. et al. Multifaceted pathways protect human skin from UV radiation. Nat Chem Biol 10, 542–551 (2014). https://doi.org/10.1038/nchembio.1548
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1548
This article is cited by
-
Regulation of biological processes by intrinsically chiral engineered materials
Nature Reviews Materials (2023)
-
Metabolomics study of fibroblasts damaged by UVB and BaP
Scientific Reports (2021)
-
Protection against UVB deleterious skin effects in a mouse model: effect of a topical emulsion containing Cordia verbenacea extract
Photochemical & Photobiological Sciences (2021)
-
In vitro study of the antioxidant, photoprotective, anti-tyrosinase, and anti-urease effects of methanolic extracts from leaves of six Moroccan Lamiaceae
Journal of Food Measurement and Characterization (2021)
-
Distinct patterns of pigment development underlie convergent hyperpigmentation between nocturnal and diurnal geckos (Squamata: Gekkota)
BMC Evolutionary Biology (2020)