Abstract
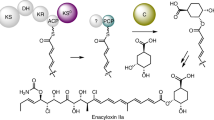
Nonribosomal peptide synthetases are versatile engines of bioactive natural product biosynthesis that function according to the multiple carrier thiotemplate mechanism. C-terminal thioesterase (TE) domains of these giant modular proteins typically catalyze product release by hydrolysis or macrocyclization. We now report an unprecedented, dual-function TE that is involved in the biosynthesis of nocardicin A, which is the paradigm monocyclic β-lactam antibiotic. Contrary to our expectation, a stereodefined series of potential peptide substrates for the nocardicin TE domain failed to undergo hydrolysis. The stringent discrimination against peptide intermediates was overcome by prior monocyclic β-lactam formation at an L-seryl site. Kinetic data are interpreted such that the TE domain acts as a gatekeeper to hold the assembling peptide on an upstream domain until β-lactam formation takes place and then rapidly catalyzes epimerization, which has not been observed previously as a TE catalytic function, and thioesterase cleavage to discharge a fully fledged pentapeptide β-lactam harboring nocardicin G, the universal precursor of the nocardicins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Aoki, H., Sakai, H., Kohsaka, M., Konomi, T. & Hosoda, J. Nocardicin A, a new monocyclic β-lactam antibiotic. I. Discovery, isolation and characterization. J. Antibiot. (Tokyo) 29, 492–500 (1976).
Townsend, C.A. & Wilson, B.A. The role of nocardicin G in nocardicin A biosynthesis. J. Am. Chem. Soc. 110, 3320–3321 (1988).
Townsend, C.A. & Brown, A.M. Nocardicin A: Biosynthetic experiments with amino acid precursors. J. Am. Chem. Soc. 105, 913–918 (1983).
Townsend, C.A., Brown, A.M. & Nguyen, L.T. Nocardicin A: Stereochemical and biomimetic studies of monocyclic β-lactam formation. J. Am. Chem. Soc. 105, 919–927 (1983).
Fawcett, P.A. et al. Synthesis of δ-(α-aminoadipyl)cysteinylvaline and its role in penicillin biosynthesis. Biochem. J. 157, 651–660 (1976).
Samson, S.M. et al. Isolation, sequence determination and expression in Escherichia coli of the isopenicillin N synthetase gene from Cephalosporium acremonium. Nature 318, 191–194 (1985).
Banko, G., Demain, A.L. & Wolfe, S. δ-(l-α-aminoadipyl)-L-cysteinyl-D-valine synthetase (ACV synthetase): a multifunctional enzyme with broad substrate specificity for the synthesis of penicillin and cephalosporin precursors. J. Am. Chem. Soc. 109, 2858–2860 (1987).
Roach, P.L. et al. Structure of isopenicillin N synthase complexed with substrate and the mechanism of penicillin formation. Nature 387, 827–830 (1997).
Townsend, C.A. & Brown, A.M. Nocardicin A biosynthesis: stereochemical course of monocyclic β-lactam formation. J. Am. Chem. Soc. 104, 1748–1750 (1982).
Gunsior, M. et al. The biosynthetic gene cluster for a monocyclic β-lactam antibiotic, nocardicin A. Chem. Biol. 11, 927–938 (2004).
Reeve, A.M., Breazeale, S.D. & Townsend, C.A. Purification, characterization, and cloning of an S-adenosylmethionine–dependent 3-amino-3-carboxypropyltransferase in nocardicin biosynthesis. J. Biol. Chem. 273, 30695–30703 (1998).
Kelly, W.L. & Townsend, C.A. Role of the cytochrome P450 NocL in nocardicin A biosynthesis. J. Am. Chem. Soc. 124, 8186–8187 (2002).
Kelly, W.L. Mutational analysis and characterization of nocardicin C-9′ epimerase. J. Biol. Chem. 279, 38220–38227 (2004).
Kelly, W.L. & Townsend, C.A. Mutational analysis of nocK and nocL in the nocardicin A producer Nocardia uniformis. J. Bacteriol. 187, 739–746 (2005).
Davidsen, J.M. & Townsend, C.A. Identification and characterization of NocR as a positive transcriptional regulator of the β-lactam nocardicin A in Nocardia uniformis. J. Bacteriol. 191, 1066–1077 (2009).
Davidsen, J.M. & Townsend, C.A. In vivo characterization of nonribosomal peptide synthetases NocA and NocB in the biosynthesis of nocardicin A. Chem. Biol. 19, 297–306 (2012).
Lambalot, R.H. et al. A new enzyme superfamily—the phosphopantetheinyl transferases. Chem. Biol. 3, 923–936 (1996).
Stachelhaus, T., Hüser, A. & Marahiel, M.A. Biochemical characterization of peptidyl carrier protein (PCP), the thiolation domain of multifunctional peptide synthetases. Chem. Biol. 3, 913–921 (1996).
Meier, J.L. & Burkart, M.D. The chemical biology of modular biosynthetic enzymes. Chem. Soc. Rev. 38, 2012–2045 (2009).
Marahiel, M.A., Stachelhaus, T. & Mootz, H.D. Modular peptide synthetases involved in nonribosomal peptide synthesis. Chem. Rev. 97, 2651–2674 (1997).
Walsh, C.T. et al. Tailoring enzymes that modify nonribosomal peptides during and after chain elongation on NRPS assembly lines. Curr. Opin. Chem. Biol. 5, 525–534 (2001).
Sattely, E.S., Fischbach, M.A. & Walsh, C.T. Total biosynthesis: in vitro reconstitution of polyketide and nonribosomal peptide pathways. Nat. Prod. Rep. 25, 757 (2008).
Du, L. & Lou, L. PKS and NRPS release mechanisms. Nat. Prod. Rep. 27, 255–278 (2010).
Baltz, R.H. Function of MbtH homologs in nonribosomal peptide biosynthesis and applications in secondary metabolite discovery. J. Ind. Microbiol. Biotechnol. 38, 1747–1760 (2011).
Davidsen, J.M., Bartley, D.M. & Townsend, C.A. Non-ribosomal propeptide precursor in nocardicin A biosynthesis predicted from adenylation domain specificity dependent on the MbtH family protein NocI. J. Am. Chem. Soc. 135, 1749–1759 (2013).
Gaudelli, N.M. & Townsend, C.A. Stereocontrolled syntheses of peptide thioesters containing modified seryl residues as probes of antibiotic biosynthesis. J. Org. Chem. 78, 6412–6426 (2013).
Trauger, J.W., Kohli, R.M., Mootz, H.D., Marahiel, M.A. & Walsh, C.T. Peptide cyclization catalysed by the thioesterase domain of tyrocidine synthetase. Nature 407, 215–218 (2000).
Ehmann, D.E., Trauger, J.W., Stachelhaus, T. & Walsh, C.T. Aminoacyl-SNACs as small-molecule substrates for the condensation domains of nonribosomal peptide synthetases. Chem. Biol. 7, 765–772 (2000).
Hamed, R.B. et al. The enzymes of β-lactam biosynthesis. Nat. Prod. Rep. 30, 21–107 (2013).
Sieber, S.A., Walsh, C.T. & Marahiel, M.A. Loading peptidyl-coenzyme A onto peptidyl carrier proteins: a novel approach in characterizing macrocyclization by thioesterase domains. J. Am. Chem. Soc. 125, 10862–10866 (2003).
Gokhale, R.S., Hunziker, D., Cane, D.E. & Khosla, C. Mechanism and specificity of the terminal thioesterase domain from the erythromycin polyketide synthase. Chem. Biol. 6, 117–125 (1999).
Lu, H., Tsai, S.C., Khosla, C. & Cane, D.E. Expression, site-directed mutagenesis, and steady state kinetic analysis of the terminal thioesterase domain of the methymycin/picromycin polyketide synthase. Biochemistry 41, 12590–12597 (2002).
Stachelhaus, T. & Walsh, C.T. Mutational analysis of the epimerization domain in the initiation module PheATE of gramicidin S synthetase. Biochemistry 39, 5775–5787 (2000).
Linne, U. & Marahiel, M.A. Control of directionality in nonribosomal peptide synthesis: role of the condensation domain in preventing misinitiation and timing of epimerization. Biochemistry 39, 10439–10447 (2000).
Amyes, T.L. & Richard, J.P. Generation and stability of a simple thiol ester enolate in aqueous solution. J. Am. Chem. Soc. 114, 10297–10302 (1992).
Bordwell, F.G. Equilibrium acidities in dimethyl sulfoxide solution. Acc. Chem. Res. 21, 456–463 (1988).
Balibar, C.J., Vaillancourt, F.H. & Walsh, C.T. Generation of D amino acid residues in assembly of arthrofactin by dual condensation/epimerization domains. Chem. Biol. 12, 1189–1200 (2005).
Patel, H.M., Tao, J. & Walsh, C.T. Epimerization of an L-cysteinyl to a D-cysteinyl residue during thiazoline ring formation in siderophore chain elongation by pyochelin synthetase from Pseudomonas aeruginosa. Biochemistry 42, 10514–10527 (2003).
Kopp, F. & Marahiel, M.A. Macrocyclization strategies in polyketide and nonribosomal peptide biosynthesis. Nat. Prod. Rep. 24, 735–749 (2007).
Giraldes, J.W. et al. Structural and mechanistic insights into polyketide macrolactonization from polyketide-based affinity labels. Nat. Chem. Biol. 2, 531–536 (2006).
Smith, S. The animal fatty acid synthase: one gene, one polypeptide, seven enzymes. FASEB J. 8, 1248–1259 (1994).
Tsai, S.C. et al. Crystal structure of the macrocycle-forming thioesterase domain of the erythromycin polyketide synthase: versatility from a unique substrate channel. Proc. Natl. Acad. Sci. USA 98, 14808–14813 (2001).
Korman, T.P. et al. Structure and function of an iterative polyketide synthase thioesterase domain catalyzing Claisen cyclization in aflatoxin biosynthesis. Proc. Natl. Acad. Sci. USA 107, 6246–6251 (2010).
Newman, A.G., Vagstad, A.L., Belecki, K., Scheerer, J.R. & Townsend, C.A. Analysis of the cercosporin polyketide synthase CTB1 reveals a new fungal thioesterase function. Chem. Commun. (Camb.) 48, 11772–11774 (2012).
Vagstad, A.L., Hill, E.A., Labonte, J.W. & Townsend, C.A. Characterization of a fungal thioesterase having Claisen cyclase and deacetylase activities in melanin biosynthesis. Chem. Biol. 19, 1525–1534 (2012).
Vaillancourt, F.H., Yeh, E., Vosburg, D.A., O'Connor, S.E. & Walsh, C.T. Cryptic chlorination by a non-haem iron enzyme during cyclopropyl amino acid biosynthesis. Nature 436, 1191–1194 (2005).
Salituro, G.M. & Townsend, C.A. Total syntheses of (−)-nocardicins A–G: a biogenetic approach. J. Am. Chem. Soc. 112, 760–770 (1990).
Ho, S.N., Hunt, H.D., Horton, R.M., Pullen, J.K. & Pease, L.R. Site-directed mutagenesis by overlap extension using the polymerase chain reaction. Gene 77, 51–59 (1989).
Collier, H.B. Letter: a note on the molar absorptivity of reduced Ellman's reagent, 3-carboxylato-4-nitrothiophenolate. Anal. Biochem. 56, 310–311 (1973).
Acknowledgements
This work was supported by US National Institutes of Health grant AI014937. We thank K.A. Moshos, C.J. Hastings and J.W. Li for their helpful discussion, chemical advice and encouragement and R.F. Li for guidance with molecular biology. We also thank K. Belecki and I.P. Mortimer for high-resolution MS data and C.T. Walsh (Harvard University) for providing the pET29-Sfp expression plasmid. A. Majumdar is graciously acknowledged for his assistance with the 1H-NMR arrayed D2O-exchange experiment.
Author information
Authors and Affiliations
Contributions
C.A.T. and N.M.G. developed the hypothesis and designed the study. N.M.G. performed all of the syntheses and experiments reported. Both authors analyzed and discussed the results. N.M.G. and C.A.T. prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Note and Supplementary Figures 1–9. (PDF 20234 kb)
Rights and permissions
About this article
Cite this article
Gaudelli, N., Townsend, C. Epimerization and substrate gating by a TE domain in β-lactam antibiotic biosynthesis. Nat Chem Biol 10, 251–258 (2014). https://doi.org/10.1038/nchembio.1456
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1456
This article is cited by
-
Exploring the Molecular Basis of Substrate and Product Selectivities of Nocardicin Bifunctional Thioesterase
Interdisciplinary Sciences: Computational Life Sciences (2022)
-
Thioesterase-mediated side chain transesterification generates potent Gq signaling inhibitor FR900359
Nature Communications (2021)
-
Structure of a bound peptide phosphonate reveals the mechanism of nocardicin bifunctional thioesterase epimerase-hydrolase half-reactions
Nature Communications (2019)
-
The hidden enzymology of bacterial natural product biosynthesis
Nature Reviews Chemistry (2019)
-
Monobactam formation in sulfazecin by a nonribosomal peptide synthetase thioesterase
Nature Chemical Biology (2018)