Abstract
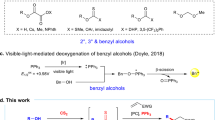
The development of new means of activating molecules and bonds for chemical reactions is a fundamental objective for chemists. In this regard, visible-light photoredox catalysis has emerged as a powerful technique for chemoselective activation of chemical bonds under mild reaction conditions. Here, we report a visible-light-mediated photocatalytic alcohol activation, which we use to convert alcohols to the corresponding bromides and iodides in good yields, with exceptional functional group tolerance. In this fundamentally useful reaction, the design and operation of the process is simple, the reaction is highly efficient, and the formation of stoichiometric waste products is minimized.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yoon, T. P., Ischay, M. A. & Du, J. Visible light photocatalysis as a greener approach to photochemical synthesis. Nature Chem. 2, 527–532 (2010).
Narayanam, J. M. R. & Stephenson, C. R. J. Visible light photoredox catalysis: applications in organic synthesis. Chem. Soc. Rev. doi:10.1039/b913880n (2010).
Hoffmann, N. Photochemical reactions as key steps in organic synthesis. Chem. Rev. 108, 1052–1103 (2008).
Fagnoni, M., Dondi, D., Ravelli, D. & Albini, A. Photocatalysis for the formation of the C–C bond. Chem. Rev. 107, 2725–2756 (2007).
Juris, A. et al. Ruthenium(II) polypyridine complexes: photophysics, photochemistry, electrochemistry, and chemiluminescence. Coord. Chem. Rev. 84, 85–277 (1988).
Nicewicz, D. A. & MacMillan, D. W. C. Merging photoredox catalysis with organocatalysis: the direct asymmetric alkylation of aldehydes. Science 322, 77–80 (2008).
Ischay, M. A., Anzovino, M. E., Du, J. & Yoon, T. P. Efficient visible light photocatalysis of [2+2] enone cycloadditions. J. Am. Chem. Soc. 130, 12886–12887 (2008).
Nicolaou, K. C. & Snyder, S. A. The essence of total synthesis. Proc. Natl Acad. Sci. USA 101, 11929–11936 (2004).
Taylor, M. S. & Jacobsen, E. N. Asymmetric catalysis in complex target synthesis. Proc. Natl Acad. Sci. USA 101, 5368–5373 (2004).
Beeler, A. B., Su, S., Singleton, C. A. & Porco, J. A. Jr. Discovery of chemical reactions through multidimensional screening. J. Am. Chem. Soc. 129, 1413–1419 (2007).
Beeson, T. D., Mastracchio, A., Hong, J–B., Ashton, K. & Macmillan, D. W. C. Enantioselective organocatalysis using SOMO activation. Science 316, 582–585 (2007).
Doyle, A. G. & Jacobsen, E. N. Small–molecule H-bond donors in asymmetric catalysis. Chem. Rev. 107, 5713–5743 (2007).
Zhang, Z. & Schreiner, P. R. (Thio)urea organocatalysis—what can be learnt from anion recognition? Chem. Soc. Rev. 38, 1187–1198 (2009).
Witham, C. A. et al. Converting homogeneous to heterogeneous in electrophilic catalysis using monodisperse metal nanoparticles. Nature Chem. 2, 36–41 (2010).
Cong, H., Becker, C. F., Elliott, S. J., Grinstaff, M. W. & Porco, J. A. Jr. Silver nanoparticle-catalyzed Diels–Alder cycloadditions of 2′-hydroxychalcones. J. Am. Chem. Soc. 132, 7514–7518 (2010).
Young, I. S. & Baran, P. S. Protecting group-free synthesis as an opportunity for invention. Nature Chem. 1, 193–205 (2009).
Burns, N. Z., Baran, P. S. & Hoffmann, R. W. Redox economy in organic synthesis. Angew. Chem. Int. Ed. 48, 2854–2867 (2009).
Newhouse, T., Baran, P. S. & Hoffmann, R. W. The economies of synthesis. Chem. Soc. Rev. 38, 3010–3021 (2009).
Nagib, D. A., Scott, M. E. & MacMillan, D. W. C. Enantioselective α-trifluoromethylation of aldehydes via photoredox organocatalysis. J. Am. Chem. Soc. 131, 10875–10877 (2009).
Du, J. & Yoon, T. P. Crossed intermolecular [2+2] cycloadditions of acyclic enones via visible light photocatalysis. J. Am. Chem. Soc. 131, 14604–14605 (2009).
Narayanam, J. M. R., Tucker, J. W. & Stephenson, C. R. J. Electron-transfer photoredox catalysis: development of a tin-free reductive dehalogenation reaction. J. Am. Chem. Soc. 131, 8756–8757 (2009).
Tucker, J. W., Narayanam, J. M. R., Krabbe, S. W. & Stephenson, C. R. J. Electron transfer photoredox catalysis: intramolecular radical addition to indoles and pyrroles. Org. Lett. 12, 368–371 (2010).
Tucker, J. W., Nguyen, J. D., Narayanam, J. M. R., Krabbe, S. W. & Stephenson, C. R. J. Tin-free radical cyclization reactions initiated by visible light photoredox catalysis. Chem. Commun. 46, 4985–4987 (2010).
Furst, L., Matsuura, B. S., Narayanam, J. M. R., Tucker, J. W. & Stephenson, C. R. J. Visible light-mediated intermolecular C–H functionalization of electron-rich heterocycles with malonates. Org. Lett. 12, 3104–3107 (2010).
Condie, A. G., Gonzalez–Gomez, J. C. & Stephenson, C. R. J. Visible-light photoredox catalysis: aza–Henry reactions via C–H functionalization. J. Am. Chem. Soc. 132, 1464–1465 (2010).
Larock, R. C. Comprehensive Organic Transformations 2nd edn, 689 (John Wiley & Sons, 1999).
Weiss, R. G. & Snyder, E. I. Stereochemistry of chloride formation from alcohols and thiols by use of triphenylphosphine and carbon tetrachloride. J. Chem. Soc. Chem. Commun. 21, 1358–1359 (1968).
Weiss, R. G. & Snyder, E. I. Stereochemistry of displacement reactions at the neopentyl carbon. Further observations on the triphenylphosphine–polyhalomethane–alcohol reaction. J. Org. Chem. 36, 403–406 (1972).
Fujisawa, T., Iida, S. & Sato, T. A convenient method for the transformation of alcohols to alkyl chlorides using N,N-diphenylchlorophenylmethyleniminium chloride. Chem. Lett. 13, 1173–1174 (1984).
Mukaiyama, T., Shoda, S.-I. & Watanabe, Y. A new synthetic method for the transformation of alcohols to alkyl chlorides using 2-chlorobenzoxazolium salt. Chem. Lett. 4, 383–386 (1977).
Benazza, T., Uzan, R., Beaupere, D. & Demailly, G. Direct regioselective chlorination of unprotected hexitols and pentitols by Vilsmeier and Haack's salt. Tetrahedron Lett. 33, 4901–4904 (1992).
Benazza, T., Uzan, R., Beaupere, D. & Demailly, G. Direct regioselective chlorination of unprotected hexitols and pentitols by Viehe's Salt. Tetrahedron Lett. 33, 3129–3132 (1992).
Kelly, B. D. & Lambert, T. H. Aromatic cation activation of alcohols: conversion to alkyl chlorides using dichlorodiphenylcyclopropene. J. Am. Chem. Soc. 131, 13930–13931 (2009).
Trost, B. M. The atom economy—a search for synthetic efficiency. Science 254, 1471–1477 (1991).
Arstad, E., Barrett, A. G. M., Hopkins, B. T. & Kobberling, J. ROMPgel-supported triphenylphosphine with potential application in parallel synthesis. Org. Lett. 4, 1975–1977 (2002).
Li, C.-J. & Trost, B. M. Green chemistry for chemical synthesis. Proc. Natl Acad. Sci. USA 105, 13197–13202 (2008).
Canoyelo, H. & Deronzier, A. Photo-oxidation of tris(2,2′-bipyridine)ruthenium(II) by para-substituted benzene diazonium salts in acetonitrile. Two-compartment photoelectrochemical cell applications. J. Chem. Soc. Faraday Trans. I 80, 3011–3019 (1984).
Canoyelo, H. & Deronzier, A. Photo-oxidation of some carbinols by the Ru(II) polypyridyl complex–aryl diazonium salt system. Tetrahedron Lett. 25, 5517–5520 (1984).
Zen, J. M., Liou, S. L., Kumar, A. S. & Hsia, M. S. An efficient and selective photocatalytic system for the oxidation of sulfides to sulfoxides. Angew. Chem. Int. Ed. 42, 577–579 (2003).
Ischay, M. A., Lu, Z. & Yoon, T. P. [2+2] Cycloadditions by oxidative visible light photocatalysis. J. Am. Chem. Soc. 132, 8572–8574 (2010).
Léonel, E., Paugam, J. P. & Nédélec, J.-Y. A new preparative route to organic halides from alcohols via the reduction of polyhalomethanes. J. Org. Chem. 62, 7061–7064 (1997).
Fukui, K., Morokuma, K., Kato, H. & Yonezawa, T. The polarographic reduction and electronic structures of organic halides. Bull. Chem. Soc. Jpn 36, 217–222 (1963).
Léonel, E., Lejaye, M., Oudeyer, S., Paugam, J. P. & Nédélec, J.-Y. gem-Dihalocyclopropane formation by iron/copper activation of tetrahalomethanes in the presence of nucleophilic olefins. Evidence for a carbene pathway. Tetrahedron Lett. 45, 2635–2638 (2004).
Kharasch, M. S., Jensen, E. V. & Urry, W. H. Addition of carbon tetrabromide and bromoform to olefins. J. Am. Chem. Soc. 68, 154–155 (1946).
Hepburn, D. R. & Hudson, H. R. Factors in the formation of isomerically and optically pure alkyl halides. Part XI. Vilsmeier reagents for the replacement of a hydroxy-group by chlorine or bromine. J. Chem. Soc. Perkin Trans. I, 754–757 (1976).
Acknowledgements
This work was supported by the donors of the American Chemical Society Petroleum Research Fund (48479-G1) and Boston University. Nuclear magnetic resonance (CHE-0619339) and mass spectrometry (CHE-0443618) facilities at Boston University are supported by the National Science Foundation. The authors are grateful to A. Phillips, J. Porco and P. Wipf for helpful suggestions regarding this manuscript, and to F. Meschini for preliminary studies.
Author information
Authors and Affiliations
Contributions
C.D. performed the experiments. All authors conceived and designed the experiments, analysed the data, contributed to discussions and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 2036 kb)
Rights and permissions
About this article
Cite this article
Dai, C., Narayanam, J. & Stephenson, C. Visible-light-mediated conversion of alcohols to halides. Nature Chem 3, 140–145 (2011). https://doi.org/10.1038/nchem.949
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.949